Abstract
Survivin, a member of the inhibitor of apoptosis protein family, is overexpressed in a variety of human neoplasms. The prognostic significance of survivin expression in diffuse large B-cell lymphoma patients treated with rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP) is unclear. We used standard immunohistochemistry methods to quantify survivin expression in 463 patients with de novo diffuse large B-cell lymphoma who received the R-CHOP. Of the 463 patients, 269 (58%) had survivin overexpression with a cutoff of >25%, associated with an International Prognostic Index score of >2 (P=0.015), disease in ≥2 extranodal sites (P=0.011), and a high Ki-67 index (P<0.0001). Among patients with activated B cell-like disease, the overall survival rate of survivin-positive patients was significantly lower than that of survivin-negative patients (P=0.033); multivariate analysis confirmed that in these patients, survivin overexpression was an independent prognostic factor for survival. Among patients with wild-type p53 overexpression, the overall survival and progression-free survival rates of the survivin-positive group were significantly lower than those of the survivin-negative group (P=0.035 and P=0.04 respectively). In STAT3-positive patients, survivin overexpression was associated with significantly better survival. Among patients with activated B cell-like disease, survivin-positive compared with survivin-negative groups had significantly different gene expression signatures, including genes involved in mitosis or tumor cell proliferation. Our results indicate that survivin is an independent prognostic factor for poor outcome in patients with activated B cell-like disease treated with the R-CHOP regimen, and patients with survivin-positive activated B cell-like diffuse large B-cell lymphoma seem to benefit less from this treatment and may require additional novel agents.
Similar content being viewed by others
Main
Diffuse large B-cell lymphoma, the most common B-cell lymphoma, comprises ∼31% of all lymphomas in Western countries and 37% of B-cell tumors worldwide.1, 2, 3 In the 1970s, a subset of patients with diffuse large B-cell lymphoma was first cured with anthracycline-based combination chemotherapy regimens.4 Subsequently, the cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) regimen became the standard treatment for diffuse large B-cell lymphoma patients because of its equivalent response and survival rates and low toxicity.5 With the development of anti-CD20 monoclonal antibody rituximab, CHOP plus rituximab (R-CHOP) regimen replaced CHOP, based on the improved complete remission rate and event-free survival rate of R-CHOP, currently the standard treatment for diffuse large B-cell lymphoma.6, 7, 8, 9 However, despite many breakthroughs in the treatment of diffuse large B-cell lymphoma, up to one-third of diffuse large B-cell lymphoma patients still suffer from relapse or primary refractory disease.10
The variable clinical presentations of diffuse large B-cell lymphoma reflect the disease’s molecular heterogeneity. Gene expression profiling studies have identified two molecularly distinct forms of diffuse large B-cell lymphoma: germinal center B cell-like diffuse large B-cell lymphoma and activated B cell-like diffuse large B-cell lymphoma.11 Patients with germinal center B cell-like diffuse large B-cell lymphoma have significantly better overall survival than those with activated B cell-like diffuse large B-cell lymphoma.11 A refined gene expression profiling-based classification based on B cell-associated gene signatures from the normal B-cell hierarchy12 and presence of MYC and BCL2 rearrangements by fluorescent in situ hybridization (FISH) are additional promising prognostic factors.13, 14 However, the clinical International Prognostic Index is still the gold standard in clinic for prognostic stratification of diffuse large B-cell lymphoma.15
Survivin, which is encoded by BIRC5 (ref. 16) and a member of the inhibitor of apoptosis protein family, was identified in 1997.17 Experimental evidence suggests that survivin is involved in multiple signaling pathways in tumors and might globally affect multiple signaling pathways and surpass the heterogeneity or complexity of tumors. Survivin lacks a caspase-associated recruiting domain that is critical for binding and inactivating caspases.18 However, survivin inhibits caspase 9 by binding to and stabilizing the X-linked inhibitor of apoptosis,19 and this inhibitory effect is negatively regulated by second mitochondria-derived activator of caspases.20 Survivin also interacts with hepatitis B X-interacting protein that binds to caspase 9 and inhibits mitochondrial-driven apoptosis.21
In addition to its functions in apoptosis, survivin is also involved in cell division. Survivin is a chromosomal passenger protein22 and interacts with other proteins in the chromosomal passenger complex, such as aurora kinase B, inner centromere protein, and borealin.23 Signal transducer and activator of transcription 3 (STAT3) and p53 proteins regulate survivin expression.24, 25 Cyclin-dependent kinase1 and heat shock protein 90 participate in posttranslational modifications of survivin by regulating survivin’s stability.26, 27
Survivin is expressed during fetal development but is undetectable in normal terminally differentiated adult tissues. Previous studies have shown that survivin is highly expressed in most human solid cancers, such as carcinomas of lung, colon, rectum, breast, and bladder, and neuroblastoma,17, 28, 29 and survivin expression is relevant to the prognosis or therapeutic response of these cancers.28, 29, 30, 31, 32 Survivin is also expressed in hematological malignancies33, 34 and survivin expression has been identified as a prognostic factor in diffuse large B-cell lymphoma patients treated with the CHOP regimen.35 However, in the R-CHOP era, studies on the prognostic significance of survivin in diffuse large B-cell lymphoma reported in the literature are conflicting.36, 37 In this study, we investigated survivin expression and its prognostic impact in 463 patients with de novo diffuse large B-cell lymphoma who were treated with the standard R-CHOP regimen.
Materials and methods
Patient Selection
We reviewed the cases of 463 patients with de novo diffuse large B-cell lymphoma who were diagnosed between 2000 and 2010 and treated with R-CHOP regimen. These cases were organized as a part of the International diffuse large B-cell lymphoma rituximab-CHOP Consortium Program study. All cases were diagnosed according to World Health Organization classification criteria and reviewed by a group of hematopathologists. Patients with transformation from low-grade B-cell lymphoma, primary cutaneous diffuse large B-cell lymphoma, primary central nervous system diffuse large B-cell lymphoma, or primary mediastinal large B-cell lymphoma were excluded, as were patients with AIDS/HIV. Patients were classified as either germinal center B cell-like or activated B cell-like subtypes mainly according to gene expression profiling and/or by the immunohistochemical Visco–Young algorithm.38 The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of each participating center. The overall collaboration was approved by the Institutional Review Board of the University of Texas MD Anderson Cancer Center in Houston, TX, USA.
Tissue Microarray and Immunohistochemistry Studies
Hematoxylin and eosin-stained slides from each of the 463 diffuse large B-cell lymphoma patients were reviewed. Tumor cell-rich areas were selected for tissue microarray analysis and prepared using a tissue microarrayer (Beecher Instruments). Immunohistochemistry studies using antibodies for survivin (EP2880Y, Epitomics), p53 (DO-7, DAKO), pSTAT3 (EP2147Y, Abcam), Myc (clone Y69, Epitomics), Bcl-2 (Clone-124, DAKO), Ki-67 (MIB-1, DAKO), Blimp-1 (EPR16655, Epitomics), pAKT (726E11, CST), CD10 (56C6, Vantana), Bcl-6 (PG-B6p, DAKO), MUM1 (MUM1P, DAKO), and FOXP1 (EPR4113, Abcam) were performed using a streptavidin-biotin complex technique on 4 μm tissue microarray sections in all cases.38, 39, 40, 41, 42, 43, 44 A list of antibodies and methods used for immunostaining are summarized in Supplementary Table S1. The formalin-fixed, paraffin-embedded tissue slides underwent deparaffinization and heat-induced antigen retrieval techniques. An endogenous biotin-blocking kit (Ventana) was used to decrease background staining. Following antigen retrieval and primary antibody incubation, the reaction was completed in a Ventana ES instrument using a diaminobenzidine immunoperoxidase detection kit (Ventana). The immunohistochemical stains were scored with 5% increments independently by two pathologists (ZL and KHY), and disagreements were resolved by joint review at a multiheaded microscope. A cutoff value for each marker’s overexpression was established from an analysis of receiver-operating characteristic (ROC) curves and/or X-Tile analysis as described previously to achieve maximum specificity and sensitivity. When an optimal cutoff could not be determined by ROC curve and X-Tile analyses, a conventional cutoff value for individual markers was decided on the basis of previous reports in the literature or mean values. The optimal cutoff thresholds were determined using X-Tile statistical software (http://www.tissuearray.org/rimmlab), based on the association of the biomarker with the optimal χ2 value for overall survival. The distribution of specific biomarker IHC data could produce a cut point that corresponds to the highest χ2 value by X-Tile. By using X-Tile, the optimal threshold could be independently determined for many biomarkers. However, no cutoff value could be obtained for rare biomarker, such as Ki-67 in X-Tile, because none of the cutoff value is associated with overall survival or progression-free survival. Thus, 70% threshold value was used for Ki-67. The cutoff value for survivin overexpression was defined as 25% positive tumor cells. The cutoff values for p53, pSTAT3, Ki-67, and cell-of-origin markers have been described previously: 20% for p53, 50% for pSTAT3, 30% for CD10, Bcl-6, and MUM1, 60% for FOXP1, and 70% for Ki-67 (Supplementary Table S1).38, 39, 41
Gene Expression Profiling
Total RNAs were extracted from formalin-fixed, paraffin-embedded tissue specimens of 425 patients. Gene expression profiling data sets were acquired using Affymetrix GeneChip Human Genome U133 Plus 2.0 as previously described.38 The CEL files are deposited in the National Center for Biotechnology Information Gene Expression Omnibus repository (GSE#31312).38 The parameters of percent, scaling factor, and background (average, min, max) were used to assess the quality of the arrays. During the quality control analysis, the samples with present calls <20% were filtered from the data set, leaving 384 samples. The CEL data were normalized using robust multiarray analysis and patients were assigned a molecular subtype (germinal center B-cell type, activated B-cell type, or unclassifiable) according to the expression of classifier genes reported previously.11, 45, 46 An analysis of differential expression gene expression was performed using t-tests between the contrasts of interest. The contrasts were visualized with heatmaps using significant probe sets.
Statistical Analysis
Different diffuse large B-cell lymphoma subgroups’ clinical and laboratory features at the time of presentation were compared using the χ2 test. The overall survival duration was calculated from the date of diagnosis to the date of last follow-up or death. The progression-free survival duration was calculated from the date of diagnosis to the time of progression or death. Different groups’ overall survival and progression-free survival curves were analyzed by using the Kaplan–Meier method that was performed with GraphPad Prism 6 software. Between-group differences in overall survival and progression-free survival were compared using the log-rank (Mantel–Cox) test. Univariate and multivariate analyses for survival were performed using the Cox proportional hazards regression model; IBM SPSS statistics V22 was used to conduct the analyses. Unpaired t-tests were used to compare different groups’ expressions of various protein and BIRC5 mRNA levels. All differences with P-values <0.05 were considered to be statistically significant.
Results
Patient Characteristics
The main clinical characteristics of the patients in the study group are summarized in Table 1 according to survivin expression. The median age of patients in the study was 64 years (range, 16–92 years); 273 patients (59%) were >60 years old and 268 (58%) were men. A total of 227 patients (49%) were classified as germinal center B cell-like subtype and 233 patients (51%) were classified as activated B cell-like subtype.
Survivin Expression and Clinical Parameters
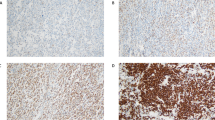
The immunohistochemical results (Figure 1a–j) and the frequency of survivin expression in the activated B cell-like diffuse large B-cell lymphoma and germinal center B cell-like diffuse large B-cell lymphoma subgroups are shown (Figure 1k–m). The staining pattern of survivin is predominantly nuclear. Equivocal cytoplasmic staining may be present in ∼10% cases, but very weak staining precludes meaningful analysis. Survivin overexpression was demonstrated in 194 of 463 (42%) diffuse large B-cell lymphoma patients (Table 1). The BIRC5 mRNA level of survivin-positive diffuse large B-cell lymphoma group was significantly higher than that of the survivin-negative diffuse large B-cell lymphoma group (Figure 1n), and similar results were found in the activated B cell-like diffuse large B-cell lymphoma and germinal center B cell-like diffuse large B-cell lymphoma subgroups (Figure 1o). The differential BIRC5 mRNA expression between survivin-positive and -negative diffuse large B-cell lymphoma samples supported the cutoff value of survivin overexpression by immunohistochemistry. In addition, the difference in survivin expression between the germinal center B cell-like diffuse large B-cell lymphoma and activated B-cell-like diffuse large B-cell lymphoma subgroups was not significant (P=0.19; Figure 1p). Consistent with this result, the difference in BIRC5 mRNA level (P=0.24) between the germinal center B cell-like diffuse large B-cell lymphoma and activated B cell-like diffuse large B-cell lymphoma subgroups was not significant (Figure 1q).
Representative examples of immunohistochemical staining in diffuse large B-cell lymphoma (magnification × 40). (a–e) Representative example of germinal center B cell-like diffuse large B-cell lymphoma positive for survivin (a), cluster of differentiation 10 (CD10; b), and B-cell lymphoma 6 (Bcl-6; c), and negative for multiple myeloma 1 (MUM1; d) and forkhead box P1 (FOXP1; e). (f–j) Representative example of active B cell-like diffuse large B-cell lymphoma positive for survivin (f), Bcl-6 (h), MUM1 (i), and FOXP1 (j), and negative for CD10 (g). Frequency distribution of survivin expression in diffuse large B-cell lymphoma (k), activated B cell-like diffuse large B-cell lymphoma (l), and germinal center B cell-like diffuse large B-cell lymphoma (m). (n and o) Relationship between BIRC5 mRNA level and survivin expression in diffuse large B-cell lymphoma (n) and in germinal center B cell-like diffuse large B-cell lymphoma or activated B cell-like diffuse large B-cell lymphoma (o). (p and q) Expression of survivin (p) and BIRC5 mRNA (q) in germinal center B cell-like diffuse large B-cell lymphoma and activated B cell-like diffuse large B-cell lymphoma.
In diffuse large B-cell lymphoma, univariate analysis demonstrated a significant association between survivin overexpression and ≥2 extranodal disease sites (P=0.011), International Prognostic Index score >2 (P=0.015), high Ki-67 index (P<0.0001), and TP53 mutation (P=0.023; Table 1). In activated B cell-like diffuse large B-cell lymphoma, survivin overexpression was associated with therapy response (P=0.037), high Ki-67 index (P<0.0001), and p53 overexpression (P=0.0048); in germinal center B cell-like diffuse large B-cell lymphoma, survivin overexpression was associated with ≥2 extranodal disease sites (P=0.0026), International Prognostic Index score >2 (P=0.0392), and high Ki-67 index (P<0.0001; Table 1).
Relationship of Survivin Expression and Other Tumor Markers
We compared the expression of a number of tumor markers in the survivin-positive versus -negative diffuse large B-cell lymphoma groups. We found significant differences between the survivin-negative and survivin-positive groups in the expression of p53 (P=0.0024), Myc (P<0.0001), B lymphocyte-induced maturation protein-1 (Blimp-1; P=0.034), Ki-67 (P<0.0001), phosphorylated protein kinase B (pAKT; P=0.0038), and B-cell lymphoma 2 (Bcl-2; P=0.049; Figure 2a–f). We found similar results for Myc, Ki-67, and pAKT expression after stratifying patients into activated B cell-like diffuse large B-cell lymphoma and germinal center B cell-like diffuse large B-cell lymphoma subgroups (Figure 2h, j, and k). p53 and Blimp-1 expression was significantly different only between survivin-positive and survivin-negative groups of activated B cell-like diffuse large B-cell lymphoma patients (P=0.0006, P=0.021; Figure 2g and i). In both activated B cell-like diffuse large B-cell lymphoma and germinal center B cell-like diffuse large B-cell lymphoma patients, the difference in Bcl-2 expression between the survivin-positive and survivin-negative groups was not significant (P=0.10, P=0.32; Figure 2l).
The relationship between survivin expression and the expression of p53 (a), Myc (b), B lymphocyte-induced maturation protein-1 (Blimp-1; c), Ki-67 (d), phosphorylated protein kinase B (pAKT; e), and B-cell lymphoma 2 (Bcl-2; f) in diffuse large B-cell lymphoma. (g–l) The relationship between survivin expression and the expression of p53 (g), Myc (h), Blimp-1 (i), Ki-67 (j), pAKT (k), and Bcl-2 (l) in germinal center B cell-like diffuse large B-cell lymphoma and activated B cell-like diffuse large B-cell lymphoma.
Prognostic Significance of Survivin Overexpression
The median follow-up time was 44 months. The overall survival and progression-free survival rates of patients with survivin-positive diffuse large B-cell lymphoma did not differ significantly from those of patients with survivin-negative diffuse large B-cell lymphoma (P=0.08, P=0.35; Figure 3a and b). Among germinal center B cell-like diffuse large B-cell lymphoma patients, there was no significant difference between the survivin-positive and survivin-negative groups (P=0.69, P=0.74; Figure 3c and d). However, among the activated B cell-like diffuse large B-cell lymphoma patients, the overall survival rate was significantly lower in patients with survivin-positive diffuse large B-cell lymphoma than it was in patients with survivin-negative diffuse large B-cell lymphoma (P=0.033; Figure 3e and f).
Impact of survivin expression on the overall survival and progression-free survival of patients with diffuse large B-cell lymphoma (a and b), patients with germinal center B cell-like diffuse large B-cell lymphoma (c and d), and patients with activated B cell-like diffuse large B-cell lymphoma (e and f).
Furthermore, survivin overexpression affected overall survival in patients with stage III or IV disease (P=0.03) but not stage I or II disease (Supplementary Figure S1A–D). Among patients stratified according to other clinical characteristics, including International Prognostic Index score, lactate dehydrogenase level, size of largest tumor, and age, the overall survival and progression-free survival of patients with survivin-positive diffuse large B-cell lymphoma did not differ significantly from those of patients with survivin-negative diffuse large B-cell lymphoma (Supplementary Figure S1E–T).
There was a significant difference in BIRC5 mRNA level between the wild-type p53 (WT-p53) and p53 mutant (MUT-p53) subgroup of diffuse large B-cell lymphoma (P=0.0025; Figure 4a). Survivin immunoexpression results between the WT-p53 and MUT-p53 subgroups of diffuse large B-cell lymphoma were consistent with the BIRC5 mRNA results (P=0.041; Figure 4b). In the WT-p53 subgroup of diffuse large B-cell lymphoma, survivin overexpression was associated with ≥2 extranodal disease sites (P=0.0083), International Prognostic Index score >2 (P=0.041), and high Ki-67 index (P<0.0001); survivin overexpression was associated with high Ki-67 index (P<0.0001) in the MUT-p53 subgroup of diffuse large B-cell lymphoma (Table 2).
BIRC5 mRNA and survivin expression in wild-type p53 (WT-p53) and p53 mutant (MUT-p53) groups and impact of survivin expression on overall survival and progression-free survival in diffuse large B-cell lymphoma patients with different p53 expression statuses. The expression of BIRC5 mRNA and survivin in the WT-p53 subgroup was significantly different from that in the MUT-p53 group (a and b). In patients with WT-p53, survivin overexpression was associated with worse overall survival (c and d). Among patients with MUT-p53, differences in overall survival and progression-free survival between the survivin-positive and survivin-negative groups were not significant (e and f).
In diffuse large B-cell lymphoma patients with WT-p53, the overall survival but not progression-free survival of the survivin-positive group was significantly lower than that of the survivin-negative group (P=0.048, P=0.15; Figure 4c and Supplementary Figure S2A). In diffuse large B-cell lymphoma patients with MUT-p53, the overall survival and progression-free survival of the survivin-positive and survivin-negative groups did not differ significantly (P=0.98, P=0.82; Figure 4d and Supplementary Figure S2B). Of the 463 patients in the study, 82 (18%) had WT-p53 overexpression. In this patients subset, WT-p53/survivin-positive diffuse large B-cell lymphoma was associated with inferior overall survival (P=0.04) and progression-free survival (P=0.035) compared with WT-p53-positive survivin-negative diffuse large B-cell lymphoma (Figure 4e and Supplementary Figure S2C).
In the STAT3-positive diffuse large B-cell lymphoma subgroup, survivin overexpression was associated with B symptoms (P=0.036) and high Ki-67 index (P=0.03); in the STAT3-negative subgroup, survivin overexpression was associated with International Prognostic Index score >2 (P=0.016) and high Ki-67 index (P<0.0001; Table 3). Interestingly, in the 64 patients with STAT3-positive diffuse large B-cell lymphoma (14%), the coexpression of STAT3 and survivin was associated with significantly better overall survival (P=0.015) and progression-free survival (P=0.019; Figure 5a and b). We found no significant prognostic effect of survivin overexpression in the WT-p53-negative (P=0.4 and P=0.94 for overall- and progression-free survival respectively; Figure 4f and Supplementary Figure S2D) or STAT3-negative diffuse large B-cell lymphoma subgroups (marginal P=0.068 for overall survival and P=0.28 for progression-free survival; Figure 5c and d).
Impact of survivin expression on overall survival and progression-free survival in diffuse large B-cell lymphoma patients with or without signal transducer and activator of transcription 3 (STAT3) overexpression. In patients with STAT3 overexpression, survivin expression was associated with better overall survival and progression-free survival (a and b). Among patients without STAT3 expression, overall survival and progression-free survival did not differ significantly between the survivin-positive and survivin-negative groups (c and d).
Multivariate analysis confirmed the prognostic significance of International Prognostic Index score and TP53 mutation in diffuse large B-cell lymphoma (Table 4) and showed that survivin overexpression is an independent prognostic factor for the survival of patients with activated B cell-like diffuse large B-cell lymphoma (P=0.026, P=0.033; Table 4).
Differential Gene Expression
To clarify the molecular basis underlying the aggressive clinical course of survivin-positive activated B cell-like diffuse large B-cell lymphoma, we compared the gene expression profiling results of the survivin-positive activated B cell-like diffuse large B-cell lymphoma patients with those of the survivin-negative activated B cell-like diffuse large B-cell lymphoma patients. A total of 86 genes were differentially expressed; 61 genes were upregulated in survivin-positive group and 25 genes were upregulated in the survivin-negative group (FDR=0.1; Figure 6a and Supplementary Table S2). Of the 61 upregulated genes in the survivin-positive group, 18 (30%) were involved in DNA replication and repair, mitosis, cell cycle regulation, and/or proliferation. In contrast, only one upregulated gene in the survivin-negative group was involved in these processes (Table 5). In addition, among the upregulated genes in the survivin-positive group, nine were involved in metabolism and nine were involved in transcription and translation regulation. Interestingly, TRIM35, which may play a role as a tumor suppressor, was upregulated in the survivin-positive activated B cell-like diffuse large B-cell lymphoma group (Table 5).
Gene expression profiles of diffuse large B-cell lymphoma subgroups and the network of survivin’s functions. Gene expression profiles of survivin-positive and survivin-negative activated B cell-like diffuse large B-cell lymphoma (a). A total of 86 genes corresponding to 101 probe sets were differentially expressed between these groups (FDR=0.1). Gene expression profiles of survivin-positive and survivin-negative germinal center B cell-like diffuse large B-cell lymphoma is shown in (b). A total of 58 genes corresponding to 75 probe sets were differentially expressed between these groups (FDR=0.1). A brief network of survivin’s functions generated using published data. AKT, protein kinase B; AURKB, aurora kinase B; Bcl-2, B-cell lymphoma 2; CDK1, cyclin-dependent kinase 1; CENPA, centromere protein A; CPC, chromosomal passenger complex; HBXIP, hepatitis B virus X-interacting protein; HSP90, heat shock protein 90; INCENP, inner centromere protein; PI3K, phosphoinositide 3-kinase; SMAC, small mitochondria-derived activator of caspase; STAT3, signal transducer and activator of transcription 3; TOP2A, topoisomerase IIα; TUBB1, tubulin β1 chain; XIAP, X-linked inhibitor of apoptosis protein (c).
We also compared the gene expression profiling results of subgroups of germinal center B cell-like diffuse large B-cell lymphoma patients, in whom survivin was not associated with poor prognosis. Interestingly, we found that the gene expression profiling of the germinal center B cell-like diffuse large B-cell lymphoma group was very similar to that of the activated B cell-like diffuse large B-cell lymphoma group (Figure 6b and Table 5). Between the survivin-positive germinal center B cell-like diffuse large B-cell lymphoma patients and survivin-negative germinal center B cell-like diffuse large B-cell lymphoma patients, 58 genes were differentially expressed; 49 (84%) were upregulated in the survivin-positive group and 9 (16%) were upregulated in the survivin-negative group (Supplementary Table S3). Six genes—CEP55, RAD51, TOP2A, H2AFX, H2AFZ, and TAF5—were upregulated in both the survivin-positive activated B cell-like diffuse large B-cell lymphoma and survivin-positive germinal center B cell-like diffuse large B-cell lymphoma groups (Table 5). As in the survivin-positive activated B cell-like diffuse large B-cell lymphoma group, most of the other genes upregulated in the survivin-positive germinal center B cell-like diffuse large B-cell lymphoma group were involved in mitosis, cell cycle regulation, gene transcription, translation, and/or metabolism (Table 5). Comparing the survivin positivity gene expression profiling signatures of the germinal center B cell-like and activated B cell-like diffuse large B-cell lymphoma subgroups, the most distinctive feature was a group of eight genes, involved in growth factor, receptor, and signal transduction, upregulated in survivin-negative activated B cell-like diffuse large B-cell lymphoma.
Discussion
Earlier studies have shown that survivin expression is significantly associated with poor overall survival in diffuse large B-cell lymphoma patients treated with the CHOP chemotherapy regimen.35, 47, 48 However, the significant improvement in diffuse large B-cell lymphoma patients’ survival following the addition of rituximab to CHOP warranted a re-evaluation of such prognostic factors.49, 51, 52 The results of studies investigating these factors have been conflicting. Mitrovic et al37 reported that survivin had no prognostic significance in 57 diffuse large B-cell lymphoma patients treated with the R-CHOP regimen. In contrast, Markovic et al36 demonstrated that survivin was an unfavorable factor for therapy response and associated with shorter survival time in 56 diffuse large B-cell lymphoma patients treated with R-CHOP. Given such uncertainty, we conducted the present international multicenter study in a large cohort of diffuse large B-cell lymphoma patients to clarify the prognostic significance of survivin in diffuse large B-cell lymphoma patients who have received R-CHOP. In this study, we found no significant difference in survivin expression between germinal center B cell-like diffuse large B-cell lymphoma and activated B cell-like diffuse large B-cell lymphoma patients, as Markovic et al36 demonstrated, but we did find that survivin expression was associated with higher International Prognostic Index score, higher number of extranodal disease sites, and higher Ki-67 index. Survivin expression predicted shorter overall survival only in the activated B cell-like diffuse large B-cell lymphoma patients and was an independent prognostic factor for activated B cell-like diffuse large B-cell lymphoma.
The subcellular localization of survivin helps determine its function; cytoplasmic survivin is involved in apoptosis, whereas nuclear survivin is involved in cell division.53 However, the findings of studies that have investigated the prognostic significance of survivin in these subcellular pools are inconsistent.54 Some investigations have shown that nuclear survivin was associated with unfavorable prognosis, whereas other research has shown that cytoplasmic survivin was associated with unfavorable prognosis. In diffuse large B-cell lymphoma, Mitrovic et al37 and Markovic et al36 separately detected the expression of survivin in different subcellular pools. Although both groups found three staining patterns in diffuse large B-cell lymphoma, including cytoplasmic, nuclear, and mixed staining, neither group found a significant difference between any type of survivin expression and patient survival.36, 37 Variations reported from different studies may have resulted from low sensitive immunohistochemical method, low concentration of survivin in the cytoplasm, and different surviving antibodies used in the study. In this study, we only observed predominant nuclear staining pattern of survivin that correlated with poorer survival in activated B cell-like diffuse large B-cell lymphoma and in patients with wild-type p53 overexpression. However, ∼10% patients may have very weak cytoplasmic staining but it is technically challenging to be certain and biological analysis based on equivocal cytoplasmic staining is less reliable.
To identify the mechanisms that may play a role in determining the difference in survival between the survivin-positive and survivin-negative activated B cell-like diffuse large B-cell lymphoma groups, we analyzed these two groups by gene expression profiling analysis. Among activated B cell-like diffuse large B-cell lymphoma patients, the gene expression profiling results of the survivin-positive group were significantly different from those of the survivin-negative group. Remarkably, the majority of differentially expressed genes in the survivin-positive group were related to mitosis, cell cycle, and/or metabolism. This gene expression signature may help explain why survivin-positive activated B cell-like diffuse large B-cell lymphoma patients have worse outcomes than survivin-negative activated B cell-like diffuse large B-cell lymphoma patients. Interestingly, we found that the survivin gene expression signature in germinal center B cell-like diffuse large B-cell lymphoma was similar to that in activated B cell-like diffuse large B-cell lymphoma. Although survivin has been identified as an apoptosis inhibitor, it also plays a role in cell division.55, 56, 57 One study demonstrated that the deletion of the survivin gene resulted in the absence of mitotic spindle, suggesting that survivin regulates chromosome segregation and cytokinesis.58 In another study, an anti-survivin antibody application caused the premature separation of sister chromatids and the dysregulation of spindle-checkpoint activation.57 Considering the function of survivin in mitosis, we presumed that the upregulation of genes involved in the cell cycle and in mitosis in survivin-positive activated B cell-like diffuse large B-cell lymphoma is due in part to survivin overexpression but not the cause of survivin overexpression, and underlied the worse survival of survivin-positive activated B cell-like diffuse large B-cell lymphoma patients.
Genes involved in metabolism were also identified in survivin-positive activated B cell-like diffuse large B-cell lymphoma. The transaldolase 1 gene TALDO1 is a key enzyme of the pentose phosphate pathway, providing ribose-5-phosphate for nucleic acid synthesis and nicotinamide adenine dinucleotide phosphate for lipid biosynthesis.59 The phosphoglucomutase gene PGM2 catalyzes the conversion of the nucleoside breakdown products ribose-1-phosphate and deoxyribose-1-phosphate to the corresponding 5-phosphopentoses and may catalyze the interconversion of glucose-1-phosphate and glucose-6-phosphate.60 The pentose phosphate pathway and glucose-6-phosphate both contribute to the Warburg effect in cancer cell metabolism.61 However, whether the Warburg effect plays an etiological role in cancer remains unclear.61, 62 Therefore, we cannot conclude whether the genes involved in metabolism that were upregulated in survivin-positive activated B cell-like diffuse large B-cell lymphoma patients should be considered to be a result or the cause of survivin overexpression. Additional studies are needed to clarify the mechanisms underlying the difference in survival between survivin-positive and survivin-negative activated B cell-like diffuse large B-cell lymphoma patients.
We also found that survivin overexpression was associated with inferior overall survival and progression-free survival in patients with WT-p53 overexpression. The relationship between survivin and WT-p53 may account for this association. Human p53, which is encoded by the TP53 gene, is a tumor suppressor that can block cell progression by regulating the cell cycle and/or inducing apoptosis. WT-p53, but not MUT-p53, represses survivin expression at both the mRNA and protein levels.24 When a survivin expression plasmid is transfected into cells, the resulting survivin overexpression rescues the cells from p53-induced apoptosis, thereby enabling their proliferation and promoting tumor growth.24
The relationship between survivin and STAT3 is somewhat more complicated than that between survivin and p53. STAT3 directly binds to and regulates the survivin promoter,63 and STAT3 activation induces survivin expression and rescues cells from apoptosis.25, 63 However, survivin can also bind to STAT3 dimers and repress STAT3 transactivation of target gene promoters.64 Thus, clarifying the mechanism underlying the association of STAT3–survivin coexpression with better overall survival and progression-free survival is difficult.
Our study suggests that activated B cell-like diffuse large B-cell lymphoma patients derive less benefit from R-CHOP than germinal center B cell-like diffuse large B-cell lymphoma patients and therefore require additional effective treatments.65, 66 Agents targeting survivin may provide encouragement for such treatments. The small-molecule YM155, which has shown promising growth inhibitory effects and potent antitumor activities in cell lines and xenograft models,67, 68 is the most well-studied survivin inhibitor. Although the drug’s safety and tolerability have been confirmed, single-agent YM155, as monotherapy regimen, demonstrated only limited activity in patients with refractory diffuse large B-cell lymphoma in a phase II clinical study.69 In contrast, preclinical studies have demonstrated that YM155 plus rituximab, bendamustine, or a STAT3 inhibitor has obvious inhibitory effects on diffuse large B-cell lymphoma.70, 71 Taking into account the negative prognostic impact of survivin expression in activated B cell-like diffuse large B-cell lymphoma patients, assessment of clinical benefits should include stratification into different subgroups when evaluating survivin suppressants, especially when it combines with rituximab.72, 73
Besides the agents that target survivin, survivin-derived peptide cancer vaccines are another promising choice. Survivin-derived peptide vaccines can elicit immune response through increasing survivin-specific CD8+ cytotoxic T lymphocytes. Survivin-2B80-88 (refs. 74, 75, 76) and Survivin-2B77 have shown the safety and therapeutic potential in cancers, such as breast cancer, colorectal cancer, and gastric cancer. SVN53-67/M57 (SurVaxM) has demonstrated its clinical potential in the treatment of gliomas and, more importantly, this vaccine contains multiple HLA epitopes that may be applicable to a large patient population.78, 79, 80 DPX-Survivac vaccine, which has been proved to be well tolerated and shows the clinical benefits in ovarian cancer in phase I clinical trial,81 is currently being examined in diffuse large B-cell lymphoma patients with survivin expression in a phase II clinical trial.82 Furthermore, incomplete Freund’s adjuvant and type-I interferon (IFNα) are two adjuvants that can enhance survivin-specific antitumor immunity.77, 83, 84
In summary, we identified survivin as an independent prognostic factor for poor outcome in activated B cell-like diffuse large B-cell lymphoma patients treated with R-CHOP. Our findings suggest that survivin affects the survival of activated B cell-like diffuse large B-cell lymphoma patients by influencing the mitosis and/or proliferation of tumor cells and is a promising therapeutic target in diffuse large B-cell lymphoma and its subgroup patients.
References
Young KH, Medeiros LJ, Chan WC . Diffuse large B-cell lymphoma. In: Orazi A, Weiss LM, Foucar K, Knowles DM (eds). Neoplastic Hematopathology. Lippincott Williams & Wilkins: Philadelphia, PA, 2014, pp 502–565.
Testoni M, Zucca E, Young KH et al. Genetic lesions in diffuse large B-cell lymphomas. Ann Oncol 2015;26:1069–1080.
Stein H, Warnke RA, Chan WC et al. Diffuse large B-cell lymphoma, not otherwise specified. In: Swerdlow SH, Campo E, Harris NL et al. (eds). WHO Classification of Tumours of Haematopoetic and Lymphoid Tissues 4th (edn.) International Agency for Research on Cancer (IARC): Lyon, France, 2008, pp 233–261.
DeVita VT Jr, Canellos GP, Chabner B et al. Advanced diffuse histiocytic lymphoma, a potentially curable disease. Lancet 1975;1:248–250.
Fisher RI, Gaynor ER, Dahlberg S et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin's lymphoma. N Engl J Med 1993;328:1002–1006.
Coiffier B, Lepage E, Briere J et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002;346:235–242.
Pfreundschuh M, Trumper L, Osterborg A et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol 2006;7:379–391.
Coiffier B, Thieblemont C, Van Den Neste E et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the Groupe d'Etudes des Lymphomes de l'Adulte. Blood 2010;116:2040–2045.
Pfreundschuh M, Kuhnt E, Trumper L et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group. Lancet Oncol 2011;12:1013–1022.
Friedberg JW . Relapsed/refractory diffuse large B-cell lymphoma. Hematology Am Soc Hematol Educ Program 2011;2011:498–505.
Alizadeh AA, Eisen MB, Davis RE et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000;403:503–511.
Dybkaer K, Bogsted M, Falgreen S et al. Diffuse large B-cell lymphoma classification system that associates normal B-cell subset phenotypes with prognosis. J Clin Oncol 2015;33:1379–1388.
Savage KJ, Johnson NA, Ben-Neriah S et al. MYC gene rearrangements are associated with a poor prognosis in diffuse large B-cell lymphoma patients treated with R-CHOP chemotherapy. Blood 2009;114:3533–3537.
Vaidya R, Witzig TE . Prognostic factors for diffuse large B-cell lymphoma in the R(X)CHOP era. Ann Oncol 2014;25:2124–2133.
A predictive model for aggressive non-Hodgkin's lymphoma. The International Non-Hodgkin's Lymphoma Prognostic Factors Project. N Engl J Med 1993;329:987–994.
Altieri DC . The case for survivin as a regulator of microtubule dynamics and cell-death decisions. Curr Opin Cell Biol 2006;18:609–615.
Ambrosini G, Adida C, Altieri DC . A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med 1997;3:917–921.
Schimmer AD . Inhibitor of apoptosis proteins: translating basic knowledge into clinical practice. Cancer Res 2004;64:7183–7190.
Dohi T, Beltrami E, Wall NR et al. Mitochondrial survivin inhibits apoptosis and promotes tumorigenesis. J Clin Invest 2004;114:1117–1127.
Sun C, Nettesheim D, Liu Z et al. Solution structure of human survivin and its binding interface with Smac/Diablo. Biochemistry 2005;44:11–17.
Marusawa H, Matsuzawa S, Welsh K et al. HBXIP functions as a cofactor of survivin in apoptosis suppression. EMBO J 2003;22:2729–2740.
Adams RR, Carmena M, Earnshaw WC . Chromosomal passengers and the (aurora) ABCs of mitosis. Trends Cell Biol 2001;11:49–54.
Jeyaprakash AA, Klein UR, Lindner D et al. Structure of a Survivin-Borealin-INCENP core complex reveals how chromosomal passengers travel together. Cell 2007;131:271–285.
Mirza A, McGuirk M, Hockenberry TN et al. Human survivin is negatively regulated by wild-type p53 and participates in p53-dependent apoptotic pathway. Oncogene 2002;21:2613–2622.
Aoki Y, Feldman GM, Tosato G . Inhibition of STAT3 signaling induces apoptosis and decreases survivin expression in primary effusion lymphoma. Blood 2003;101:1535–1542.
O'Connor DS, Grossman D, Plescia J et al. Regulation of apoptosis at cell division by p34cdc2 phosphorylation of survivin. Proc Natl Acad Sci USA 2000;97:13103–13107.
Fortugno P, Beltrami E, Plescia J et al. Regulation of survivin function by Hsp90. Proc Natl Acad Sci USA 2003;100:13791–13796.
Swana HS, Grossman D, Anthony JN et al. Tumor content of the antiapoptosis molecule survivin and recurrence of bladder cancer. N Engl J Med 1999;341:452–453.
Adida C, Berrebi D, Peuchmaur M et al. Anti-apoptosis gene, survivin, and prognosis of neuroblastoma. Lancet 1998;351:882–883.
Kawasaki H, Altieri DC, Lu CD et al. Inhibition of apoptosis by survivin predicts shorter survival rates in colorectal cancer. Cancer Res 1998;58:5071–5074.
Monzo M, Rosell R, Felip E et al. A novel anti-apoptosis gene: Re-expression of survivin messenger RNA as a prognosis marker in non-small-cell lung cancers. J Clin Oncol 1999;17:2100–2104.
Tanaka K, Iwamoto S, Gon G et al. Expression of survivin and its relationship to loss of apoptosis in breast carcinomas. Clin Cancer Res 2000;6:127–134.
Adida C, Recher C, Raffoux E et al. Expression and prognostic significance of survivin in de novo acute myeloid leukaemia. Br J Haematol 2000;111:196–203.
Shinozawa I, Inokuchi K, Wakabayashi I et al. Disturbed expression of the anti-apoptosis gene, survivin, and EPR-1 in hematological malignancies. Leuk Res 2000;24:965–970.
Adida C, Haioun C, Gaulard P et al. Prognostic significance of survivin expression in diffuse large B-cell lymphomas. Blood 2000;96:1921–1925.
Markovic O, Marisavljevic D, Cemerikic-Martinovic V et al. Survivin expression in patients with newly diagnosed nodal diffuse large B cell lymphoma (DLBCL). Med Oncol 2012;29:3515–3521.
Mitrovic Z, Ilic I, Aurer I et al. Prognostic significance of survivin and caspase-3 immunohistochemical expression in patients with diffuse large B-cell lymphoma treated with rituximab and CHOP. Pathol Oncol Res 2011;17:243–247.
Visco C, Li Y, Xu-Monette ZY et al. Comprehensive gene expression profiling and immunohistochemical studies support application of immunophenotypic algorithm for molecular subtype classification in diffuse large B-cell lymphoma: a report from the International DLBCL Rituximab-CHOP Consortium Program Study. Leukemia 2012;26:2103–2113.
Xu-Monette ZY, Wu L, Visco C et al. Mutational profile and prognostic significance of TP53 in diffuse large B-cell lymphoma patients treated with R-CHOP: report from an International DLBCL Rituximab-CHOP Consortium Program Study. Blood 2012;120:3986–3996.
Xu-Monette ZY, Moller MB, Tzankov A et al. MDM2 phenotypic and genotypic profiling, respective to TP53 genetic status, in diffuse large B-cell lymphoma patients treated with rituximab-CHOP immunochemotherapy: a report from the International DLBCL Rituximab-CHOP Consortium Program. Blood 2013;122:2630–2640.
Ok CY, Chen J, Xu-Monette ZY et al. Clinical implications of phosphorylated STAT3 expression in de novo diffuse large B-cell lymphoma. Clin Cancer Res 2014;20:5113–5123.
Hu S, Xu-Monette ZY, Tzankov A et al. MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: a report from The International DLBCL Rituximab-CHOP Consortium Program. Blood 2013;121:4021–4031.
Tzankov A, Xu-Monette ZY, Gerhard M et al. Rearrangements of MYC gene facilitate risk stratification in diffuse large B-cell lymphoma patients treated with rituximab-CHOP. Mod Pathol 2014;27:958–971.
Visco C, Tzankov A, Xu-Monette ZY et al. Patients with diffuse large B-cell lymphoma of germinal center origin with BCL2 translocations have poor outcome, irrespective of MYC status: a report from an International DLBCL rituximab-CHOP Consortium Program Study. Haematologica 2013;98:255–263.
Dave SS, Fu K, Wright GW et al. Molecular diagnosis of Burkitt's lymphoma. N Engl J Med 2006;354:2431–2442.
Irizarry RA, Bolstad BM, Collin F et al. Summaries of Affymetrix GeneChip probe level data. Nucleic Acids Res 2003;31:e15.
Sung J-Y, Lim S-J, Kim YW et al. Prognostic significance of pSTAT3 and Survivin expression in diffuse large B-cell lymphoma. Basic Appl Pathol 2010;3:7–13.
Mainou-Fowler T, Overman LM, Dignum H et al. A new subtype-specific monoclonal antibody for IAP-survivin identifies high-risk patients with diffuse large B-cell lymphoma and improves the prognostic value of bcl-2. Int J Oncol 2008;32:59–68.
Seki R, Ohshima K, Fujisaki T et al. Prognostic impact of immunohistochemical biomarkers in diffuse large B-cell lymphoma in the rituximab era. Cancer Sci 2009;100:1842–1847.
Ngo L, Hee SW, Lim LC et al. Prognostic factors in patients with diffuse large B cell lymphoma: before and after the introduction of rituximab. Leuk Lymphoma 2008;49:462–469.
Huang YC, Liu CY, Lu HJ et al. Comparison of prognostic models for patients with diffuse large B-cell lymphoma in the rituximab era. Ann Hematol 2013;92:1513–1520.
Gaudio F, Giordano A, Perrone T et al. High Ki67 index and bulky disease remain significant adverse prognostic factors in patients with diffuse large B cell lymphoma before and after the introduction of rituximab. Acta Haematol 2011;126:44–51.
Stauber RH, Mann W, Knauer SK . Nuclear and cytoplasmic survivin: molecular mechanism, prognostic, and therapeutic potential. Cancer Res 2007;67:5999–6002.
Li F, Yang J, Ramnath N et al. Nuclear or cytoplasmic expression of survivin: what is the significance? Int J Cancer 2005;114:509–512.
Altieri DC . Validating survivin as a cancer therapeutic target. Nat Rev Cancer 2003;3:46–54.
Li F, Ambrosini G, Chu EY et al. Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 1998;396:580–584.
Kallio MJ, Nieminen M, Eriksson JE . Human inhibitor of apoptosis protein (IAP) survivin participates in regulation of chromosome segregation and mitotic exit. FASEB J 2001;15:2721–2723.
Uren AG, Wong L, Pakusch M et al. Survivin and the inner centromere protein INCENP show similar cell-cycle localization and gene knockout phenotype. Curr Biol 2000;10:1319–1328.
Basta PV, Bensen JT, Tse CK et al. Genetic variation in Transaldolase 1 and risk of squamous cell carcinoma of the head and neck. Cancer Detect Prev 2008;32:200–208.
Maliekal P, Sokolova T, Vertommen D et al. Molecular identification of mammalian phosphopentomutase and glucose-1,6-bisphosphate synthase, two members of the alpha-D-phosphohexomutase family. J Biol Chem 2007;282:31844–31851.
Upadhyay M, Samal J, Kandpal M et al. The Warburg effect: insights from the past decade. Pharmacol Ther 2013;137:318–330.
Hsu PP, Sabatini DM . Cancer cell metabolism: Warburg and beyond. Cell 2008;134:703–707.
Gritsko T, Williams A, Turkson J et al. Persistent activation of stat3 signaling induces survivin gene expression and confers resistance to apoptosis in human breast cancer cells. Clin Cancer Res 2006;12:11–19.
Wang H, Holloway MP, Ma L et al. Acetylation directs survivin nuclear localization to repress STAT3 oncogenic activity. J Biol Chem 2010;285:36129–36137.
Chen JY, Xu-Monette ZY, Deng L et al. Dysregulated CXCR4 expression promotes lymphoma cell survival and independently predicts disease progression in germinal center B-cell-like diffuse large B-cell lymphoma. Oncotarget 2015;6:5597–5614.
Li L, Xu-Monette ZY, Ok CY et al. Prognostic impact of c-Rel nuclear expression and REL amplification and crosstalk between c-Rel and the p53 pathway in diffuse large B-cell lymphoma. Oncotarget 2015; [E-pub ahead of print].
Nakahara T, Kita A, Yamanaka K et al. Broad spectrum and potent antitumor activities of YM155, a novel small-molecule survivin suppressant, in a wide variety of human cancer cell lines and xenograft models. Cancer Sci 2011;102:614–621.
Kita A, Nakahara T, Yamanaka K et al. Antitumor effects of YM155, a novel survivin suppressant, against human aggressive non-Hodgkin lymphoma. Leuk Res 2011;35:787–792.
Cheson BD, Bartlett NL, Vose JM et al. A phase II study of the survivin suppressant YM155 in patients with refractory diffuse large B-cell lymphoma. Cancer 2012;118:3128–3134.
Kaneko N, Kita A, Yamanaka K et al. Combination of YM155, a survivin suppressant with a STAT3 inhibitor: a new strategy to treat diffuse large B-cell lymphoma. Leuk Res 2013;37:1156–1161.
Kaneko N, Mitsuoka K, Amino N et al. Combination of YM155, a survivin suppressant, with bendamustine and rituximab: a new combination therapy to treat relapsed/refractory diffuse large B-cell lymphoma. Clin Cancer Res 2014;20:1814–1822.
Ok CY, Ye Q, Manyam GC et al. Age cutoff for Epstein-Barr virus-positive diffuse large B-cell lymphoma – is it necessary? Oncotarget 2015;6:13935–13946.
Ok CY, Xu-Monette ZY, Li L et al. Evaluation of NF-κB subunit expression and signaling pathway activation demonstrates that p52 expression confers better outcome in germinal center B-cell-like diffuse large B-cell lymphoma in association with CD30 and BCL2 functions. Mod Pathol 2015; [E-pub ahead of print].
Tsuruma T, Hata F, Torigoe T et al. Phase I clinical study of anti-apoptosis protein, survivin-derived peptide vaccine therapy for patients with advanced or recurrent colorectal cancer. J Transl Med 2004;2:19.
Miyazaki A, Kobayashi J, Torigoe T et al. Phase I clinical trial of survivin-derived peptide vaccine therapy for patients with advanced or recurrent oral cancer. Cancer Sci 2011;102:324–329.
Idenoue S, Hirohashi Y, Torigoe T et al. A potent immunogenic general cancer vaccine that targets survivin, an inhibitor of apoptosis proteins. Clin Cancer Res 2005;11:1474–1482.
Tsuruma T, Iwayama Y, Ohmura T et al. Clinical and immunological evaluation of anti-apoptosis protein, survivin-derived peptide vaccine in phase I clinical study for patients with advanced or recurrent breast cancer. J Transl Med 2008;6:24.
Ciesielski MJ, Ahluwalia MS, Munich SA et al. Antitumor cytotoxic T-cell response induced by a survivin peptide mimic. Cancer Immunol Immunother 2010;59:1211–1221.
Shen L, Ciesielski M, Ramakrishnan S et al. Class I histone deacetylase inhibitor entinostat suppresses regulatory T cells and enhances immunotherapies in renal and prostate cancer models. PLoS One 2012;7:e30815.
Fenstermaker RA, Ciesielski MJ . Challenges in the development of a survivin vaccine (SurVaxM) for malignant glioma. Expert Rev Vaccines 2014;13:377–385.
Riedmann EM . Ovarian cancer vaccine candidate DPX-Survivac: positive interim results from phase 1. Hum Vaccin Immunother 2012;8:1743.
Mraz M, Malinova K, Kotaskova J et al. miR-34a, miR-29c and miR-17-5p are downregulated in CLL patients with TP53 abnormalities. Leukemia 2009;23:1159–1163.
Kameshima H, Tsuruma T, Kutomi G et al. Immunotherapeutic benefit of alpha-interferon (IFNalpha) in survivin2B-derived peptide vaccination for advanced pancreatic cancer patients. Cancer Sci 2013;104:124–129.
Kameshima H, Tsuruma T, Torigoe T et al. Immunogenic enhancement and clinical effect by type-I interferon of anti-apoptotic protein, survivin-derived peptide vaccine, in advanced colorectal cancer patients. Cancer Sci 2011;102:1181–1187.
Acknowledgements
This study was supported by the National Cancer Institute/National Institutes of Health (R01CA138688 and 1RC1CA146299 to KHY). ZL is the recipient of a scholarship research award; GCM is supported by a grant from the Michael and Susan Dell Foundation; ZYX-M is the recipient of the Harold C and Mary L. Daily Endowment Fellowship and the Shannon Timmins Fellowship for Leukemia Research Award. KHY is supported by The University of Texas MD Anderson Cancer Center Institutional Research and Development Fund, an Institutional Research Grant Award, an MD Anderson Cancer Center Lymphoma Specialized Programs on Research Excellence (SPORE) Research Development Program Award, an MD Anderson Cancer Center Myeloma SPORE Research Development Program Award, a Gundersen Lutheran Medical Foundation Award, and MD Anderson Cancer Center Collaborative Funds with Roche Molecular System, Gilead Pharmaceutical, Dai Sanyo Pharmaceutical, Adaptive Biotechnology, and HTG Molecular Diagnostics, and partially supported by the National Cancer Institute/National Institutes of Health (P50CA136411 and P50CA142509). This work was also partially supported by National Cancer Institute and National Institutes of Health grants P50CA136411 and P50CA142509, and by MD Anderson’s Cancer Center Support Grant CA016672.
Author contributions
Conception and design: ZL, ZYX-M, and KHY; research performance: ZL, ZYX-M, and KHY; provision of study materials, key reagents, and technology: ZL, ZYX-M, XC, GCM, XW, AT, YX, XL, CV, LZ, SM-M, KD, AC, AO, YZ, GB, KLR, EDH, WWLC, JH, MP, AJMF, BEP, MBM, MAP, JNW, LJM, and KHY; collection and assembly of data under approved IRB and MTA: ZYXM, AT, CV, SM-M, KD, AC, AO, YZ, GB, KLR, EDH, WWLC, JH, MP, AJMF, BMP, MBM, MAP, JNW, and KHY; data analysis and interpretation: ZL, ZYX-M, LJM, and KHY; manuscript writing: ZL, ZYX-M, LJM, and KHY; final approval of manuscript: all authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Modern Pathology website
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, Z., Xu-Monette, Z., Cao, X. et al. Prognostic and biological significance of survivin expression in patients with diffuse large B-cell lymphoma treated with rituximab-CHOP therapy. Mod Pathol 28, 1297–1314 (2015). https://doi.org/10.1038/modpathol.2015.94
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2015.94
This article is cited by
-
Diagnostic and predictive biomarkers for lymphoma diagnosis and treatment in the era of precision medicine
Modern Pathology (2016)
-
New developments in the pathology of malignant lymphoma: a review of the literature published from May 2015–September 2015
Journal of Hematopathology (2015)