Abstract
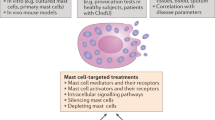
Mast cells are known to be the primary responders in allergic reactions, orchestrating strong responses to minute amounts of allergens. Several recent observations indicate that they may also have a key role in coordinating the early phases of autoimmune diseases, particularly those involving auto-antibodies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Galli, S. J., Maurer, M. & Lantz, C. S. Mast cells as sentinels of innate immunity. Curr. Opin. Immunol. 11, 53–59 (1999).
Mekori, Y. A. & Metcalfe, D. D. Mast cells in innate immunity. Immunol. Rev. 173, 131–140 (2000).
Galli, S. J., Zsebo, K. M. & Geissler, E. N. The kit ligand, stem cell factor. Adv. Immunol. 55, 1–96 (1994).
Young, J. D., Liu, C. C., Butler, G., Cohn, Z. A. & Galli, S. J. Identification, purification, and characterization of a mast cell-associated cytolytic factor related to tumor necrosis factor. Proc. Natl Acad. Sci. USA 84, 9175–9179 (1987).
Williams, C. M. & Galli, S. J. The diverse potential effector and immunoregulatory roles of mast cells in allergic disease. J. Allergy Clin. Immunol. 105, 847–859 (2000).
Turner, H. & Kinet, J. P. Signalling through the high-affinity IgE receptor FcɛRI. Nature 402, B24–B30 (1999).
Daeron, M., Malbec, O., Latour, S., Arock, M. & Fridman, W. H. Regulation of high-affinity IgE receptor-mediated mast cell activation by murine low-affinity IgG receptors. J. Clin. Invest. 95, 577–585 (1995).
Sylvestre, D. L. & Ravetech, J. V. A dominant role for mast cell Fc receptors in the arthus reaction. Immunity 5, 387–390 (1996).
Okayama, Y., Hagaman, D. D. & Metcalfe, D. D. A comparison of mediators released or generated by IFN-γ-treated human mast cells following aggregation of Fc gamma RI or Fc epsilon RI. J. Immunol. 166, 4705–4712 (2001).
Austen, K. F. & Becker, E. L. Mechanisms of immunologic injury of rat peritoneal mast cells. II. Complement requirement and phosphonate ester inhibition of release of histamine by rabbit anti-rat γ-globulin. J. Exp. Med. 124, 397–416 (1966).
Prodeus, A. P., Zhou, X., Maurer, M., Galli, S. J. & Carroll, M. C. Impaired mast cell-dependent natural immunity in complement C3-deficient mice. Nature 390, 172–175 (1997).
Supajatura, V. et al. Protective roles of mast cells against enterobacterial infection are mediated by Toll-like receptor 4. J. Immunol. 167, 2250–2256 (2001).
Applequist, S. E., Wallin, R. P. & Ljunggren, H. G. Variable expression of Toll-like receptor in murine innate and adaptive immune cell lines. Int. Immunol. 14, 1065–1074 (2002).
Schmitt, E., Huls, C., Nagel, B. & Rude, E. Characterization of a T-cell-derived mast cell costimulatory activity (MCA) that acts synergistically with interleukin 3 and interleukin 4 on the growth of murine mast cells. Cytokine 2, 407–415 (1990).
Mekori, Y. A. & Metcalfe, D. D. Mast cell-T cell interactions. J. Allergy Clin. Immunol. 104, 517–523 (1999).
Steinman, L. Multiple sclerosis: a two-stage disease. Nature Immunol. 2, 762–764 (2001).
Brown, M., Tanzola, M. & Robbie-Ryan, M. Mechanisms underlying mast cell influence on EAE disease course. Mol. Immunol. 38, 1373 (2002).
Brenner, T., Soffer, D., Shalit, M. & Levi-Schaffer, F. Mast cells in experimental allergic encephalomyelitis: characterization, distribution in the CNS and in vitro activation by myelin basic protein and neuropeptides. J. Neurol. Sci. 122, 210–213 (1994).
Rozniecki, J. J., Hauser, S. L., Stein, M., Lincoln, R. & Theoharides, T. C. Elevated mast cell tryptase in cerebrospinal fluid of multiple sclerosis patients. Ann Neurol. 37, 63–66 (1995).
Brosnan, C. F. & Tansey, F. A. Delayed onset of experimental allergic neuritis in rats treated with reserpine. J. Neuropathol. Exp. Neurol. 43, 84–93 (1984).
Dietsch, G. N. & Hinrichs, D. J. The role of mast cells in the elicitation of experimental allergic encephalomyelitis. J. Immunol. 142, 1476–1481 (1989).
Seeldrayers, P. A., Yasui, D., Weiner, H. L. & Johnson, D. Treatment of experimental allergic neuritis with nedocromil sodium. J. Neuroimmunol. 25, 221–226 (1989).
Secor, V. H., Secor, W. E., Gutekunst, C. A. & Brown, M. A. Mast cells are essential for early onset and severe disease in a murine model of multiple sclerosis. J. Exp. Med. 191, 813–822 (2000).
Lock, C. et al. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nature Med. 8, 500–508 (2002).
Chabas, D. et al. The influence of the proinflammatory cytokine, osteopontin, on autoimmune demyelinating disease. Science 294, 1731–1735 (2001).
Lafaille, J. J. et al. Myelin basic protein-specific T helper 2 (Th2) cells cause experimental autoimmune encephalomyelitis in immunodeficient hosts rather than protect them from the disease. J. Exp. Med. 186, 307–312 (1997).
Kouskoff, V. et al. Organ-specific disease provoked by systemic autoreactivity. Cell 87, 811–822 (1996).
Matsumoto, I., Staub, A., Benoist, C. & Mathis, D. Arthritis provoked by linked T and B cell recognition of a glycolytic enzyme. Science 286, 1732–1735 (1999).
Ji, H. et al. Arthritis critically dependent on innate immune system players. Immunity 16, 157–168 (2002).
Wipke, B. T. & Allen, P. M. Essential role of neutrophils in the initiation and progression of a murine model of rheumatoid arthritis. J. Immunol. 167, 1601–1608 (2001).
Ji, H. et al. Critical roles for interleukin-1 and tumor necrosis factor-α in antibody-induced arthritis. J. Exp. Med. 196, 77–85 (2002).
Lee, D. M. et al. Mast cells: a cellular link between autoantibodies and inflammatory arthritis. Science 297, 1689–1692 (2002).
Malfait, A. M. et al. The beta2-adrenergic agonist salbutamol is a potent suppressor of established collagen-induced arthritis: mechanisms of action. J. Immunol. 162, 6278–6283 (1999).
van den Broek, M. F., van den Berg,W. B. & Van de Putte, L. B. The role of mast cells in antigen induced arthritis in mice. J. Rheumatol. 15, 544–551 (1988).
Woolley, D. E. & Tetlow, L. C. Mast cell activation and its relation to proinflammatory cytokine production in the rheumatoid lesion. Arthritis Res. 2, 65–74 (2000).
Crisp, A. J., Chapman, C. M., Kirkham, S. E., Schiller, A. L. & Krane, S. M. Articular mastocytosis in rheumatoid arthritis. Arthritis Rheum. 27, 845–851 (1984).
Olsson, N., Ulfgren, A. K. & Nilsson, G. Demonstration of mast cell chemotactic activity in synovial fluid from rheumatoid patients. Ann. Rheum. Dis. 60, 187–193 (2001).
Yamada, T. et al. Localization of vascular endothelial growth factor in synovial membrane mast cells: examination with “multi-labelling subtraction immunostaining”. Virchows Arch. 433, 567–570 (1998).
Stanley, J. R. in Fitzpatrick's Dermatology in General Medicine (eds Freedberg, I. M. et al.) 666–671 (McGraw-Hill, New York, 1999).
Liu, Z. et al. A passive transfer model of the organ-specific autoimmune disease, bullous pemphigoid, using antibodies generated against the hemidesmosomal antigen, BP180. J. Clin. Invest. 92, 2480–2488 (1993).
Liu, Z. et al. The role of complement in experimental bullous pemphigoid. J. Clin. Invest. 95, 1539–1544 (1995).
Liu, Z. et al. A major role for neutrophils in experimental bullous pemphigoid. J. Clin. Invest. 100, 1256–1263 (1997).
Chen, R. et al. Mast cells play a key role in neutrophil recruitment in experimental bullous pemphigoid. J. Clin. Invest. 108, 1151–1158 (2001).
Wintroub, B. U., Mihm, M. C., Jr, Goetzl, E. J., Soter, N. A. & Austen, K. F. Morphologic and functional evidence for release of mast-cell products in bullous pemphigoid. N. Engl. J. Med. 298, 417–421 (1978).
Baba, T. et al. An eosinophil chemotactic factor present in blister fluids of bullous pemphigoid patients. J. Immunol. 116, 112–116 (1976).
Katayama, I., Doi, T. & Nishioka, K. High histamine level in the blister fluid of bullous pemphigoid. Arch. Dermatol. Res. 276, 126–127 (1984).
Konttinen, Y. T. et al. Mast cell derangement in salivary glands in patients with Sjogren's syndrome. Rheumatol. Int. 19, 141–147 (2000).
Napoli, D. C. & Freeman, T. M. Autoimmunity in chronic urticaria and urticarial vasculitis. Curr Allergy Asthma Rep. 1, 329–336 (2001).
Ludgate, M. & Baker, G. Unlocking the immunological mechanisms of orbital inflammation in thyroid eye disease. Clin. Exp. Immunol. 127, 193–198 (2002).
Kiely, P. D., Pecht, I. & Oliveira, D. B. Mercuric chloride-induced vasculitis in the Brown Norway rat: αβ T cell-dependent and -independent phases: role of the mast cell. J. Immunol. 159, 5100–5106 (1997).
Zhang, Y., Ramos, B. F. & Jakschik, B. A. Neutrophil recruitment by tumor necrosis factor from mast cells in immune complex peritonitis. Science 258, 1957–1959 (1992).
Malaviya, R., Ikeda, T., Ross, E. & Abraham, S. N. Mast cell modulation of neutrophil influx and bacterial clearance at sites of infection through TNF-α. Nature 381, 77–80 (1996).
Pedotti, R. et al. An unexpected version of horror autotoxicus: anaphylactic shock to a self-peptide. Nature Immunol. 2, 216–222 (2001).
Bielekova, B. et al. Encephalitogenic potential of the myelin basic protein peptide (amino acids 83–99) in multiple sclerosis: results of a phase II clinical trial with an altered peptide ligand. Nature Med. 6, 1167–1175 (2000).
Kappos, L. et al. Induction of a non-encephalitogenic type 2 T helper-cell autoimmune response in multiple sclerosis after administration of an altered peptide ligand in a placebo-controlled, randomized phase II trial. The Altered Peptide Ligand in Relapsing MS Study Group. Nature Med. 6, 1176–1182 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benoist, C., Mathis, D. Mast cells in autoimmune disease. Nature 420, 875–878 (2002). https://doi.org/10.1038/nature01324
Issue Date:
DOI: https://doi.org/10.1038/nature01324
This article is cited by
-
Analysis of immune cell components and immune-related gene expression profiles in peripheral blood of patients with type 1 diabetes mellitus
Journal of Translational Medicine (2021)
-
Functional testing of topical skin formulations using an optimised ex vivo skin organ culture model
Archives of Dermatological Research (2016)
-
Neuroendocrinology of the skin
Reviews in Endocrine and Metabolic Disorders (2016)
-
Heart–kidney crosstalk and role of humoral signaling in critical illness
Critical Care (2014)
-
Protective and pathological roles of mast cells and basophils
Nature Reviews Immunology (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.