Abstract
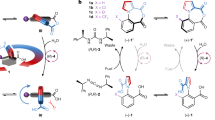
Molecular motor proteins are ubiquitous in nature1 and have inspired attempts to create artificial machines2 that mimic their ability to produce controlled motion on the molecular level. A recent example of an artificial molecular rotor is a molecule undergoing a unidirectional 120° intramolecular rotation around a single bond3,4; another is a molecule capable of repetitive unimolecular rotation driven by multiple and successive isomerization of its central double bond5,6,7,8. Here we show that sequential and unidirectional rotation can also be induced in mechanically interlocked assemblies comprised of one or two small rings moving around one larger ring. The small rings in these [2]- and [3]catenanes9 move in discrete steps between different binding sites located on the larger ring, with the movement driven by light, heat or chemical stimuli that change the relative affinity of the small rings for the different binding sites10,11,12. We find that the small ring in the [2]catenane moves with high positional integrity but without control over its direction of motion, while the two rings in the [3]catenane mutually block each other's movement to ensure an overall stimuli-induced unidirectional motion around the larger ring.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Schliwa, M. (ed.) Molecular Motors (VCH-Wiley, Weinheim, 2003)
Balzani, V., Credi, A., Raymo, F. M. & Stoddart, J. F. Artificial molecular machines. Angew. Chem. Int. Edn 39, 3348–3391 (2000)
Kelly, T. R., De Silva, H. & Silva, R. A. Unidirectional rotary motion in a molecular system. Nature 401, 150–152 (1999)
Kelly, T. R., Silva, R. A., De Silva, H., Jasmin, S. & Zhao, Y. A rationally designed prototype of a molecular motor. J. Am. Chem. Soc. 122, 6935–6949 (2000)
Koumura, N., Zijlstra, R. W. J., van Delden, R. A., Harada, N. & Feringa, B. L. Light-driven monodirectional molecular rotor. Nature 401, 152–155 (1999)
Feringa, B. L., Koumura, N., van Delden, R. A. & ter Wiel, M. K. J. Light-driven molecular switches and motors. Appl. Phys. A 75, 301–308 (2002)
Koumura, N. L., Geertsema, E. M., van Gelder, M. B., Meetsma, A. & Feringa, B. L. Second generation light-driven molecular motors. Unidirectional rotation controlled by a single stereogenic center with near-perfect photoequilibria and acceleration of the speed of rotation by structural modification. J. Am. Chem. Soc. 124, 5037–5051 (2002)
van Delden, R. A., Koumura, N., Harada, N. & Feringa, B. L. Unidirectional rotary motion in a liquid crystalline environment: Color tuning by a molecular motor. Proc. Natl Acad. Sci. USA 99, 4945–4949 (2002)
Sauvage, J.-P. & Dietrich-Buchecker, C. (eds) Molecular Catenanes, Rotaxanes and Knots (VCH-Wiley, Weinheim, 1999)
Gatti, F. G. et al. Stiff, and sticky in the right places: The dramatic influence of preorganizing guest binding sites on the hydrogen bond-directed assembly of rotaxanes. J. Am. Chem. Soc. 123, 5983–5989 (2001)
Gatti, F. G. et al. Photoisomerization of a rotaxane hydrogen bonding template: Light-induced acceleration of a large amplitude rotational motion. Proc. Natl Acad. Sci. USA 100, 10–14 (2003)
Altieri, A. et al. Remarkable positional discrimination in bistable, light and heat-switchable, hydrogen bonded molecular shuttles. Angew. Chem. Int. Edn 42, 2296–2300 (2003)
Yasuda, R., Noji, H., Kinosita, K. Jr & Yoshida, M. F1-ATPase is a highly efficient molecular motor that rotates with discrete 120° steps. Cell 93, 1117–1124 (1998)
Frkanec, L., Jokić, M., Makarević, J., Wolsperger, K. & Žinić, M. Bis(PheOH) maleic acid amide-fumaric acid amide photoisomerization induces microsphere-to-gel fibre morphological transition: A photoinduced gelation system. J. Am. Chem. Soc. 124, 9716–9717 (2002)
Feynman, R., Leighton, R. B. & Sands, M. L. The Feynman Lectures on Physics Vol. 1, Ch. 46 (Addison-Wesley, Reading, MA, 1963)
Davis, A. P. Tilting at windmills? The second law survives. Angew. Chem. Int. Edn 37, 909–910 (1998)
Brouwer, A. M. et al. Photoinduction of fast, reversible translational motion in a hydrogen-bonded molecular shuttle. Science 291, 2124–2128 (2001)
Kelly, T. R. et al. A molecular brake. J. Am. Chem. Soc. 116, 3657–3658 (1994)
Trnka, T. M. & Grubbs, R. H. The development of L2X2Ru = CHR olefin metathesis catalysts: An organometallic success story. Acc. Chem. Res. 34, 18–29 (2001)
Acknowledgements
We thank E. M. Perez for developing the catalytic Br2 reaction conditions and P. J. Nash for several of the model [2]rotaxanes. This work was carried out through the support of the RTN network EMMMA and the FET MechMol. D.A.L. is an EPSRC Advanced Research Fellow.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Leigh, D., Wong, J., Dehez, F. et al. Unidirectional rotation in a mechanically interlocked molecular rotor. Nature 424, 174–179 (2003). https://doi.org/10.1038/nature01758
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01758
This article is cited by
-
Selective synthesis of tightly- and loosely-twisted metallomacrocycle isomers towards precise control of helicity inversion motion
Nature Communications (2023)
-
Catenated covalent organic frameworks constructed from polyhedra
Nature Synthesis (2023)
-
Ratcheting synthesis
Nature Reviews Chemistry (2023)
-
Thermally-induced atropisomerism promotes metal-organic cage construction
Nature Communications (2023)
-
An electric molecular motor
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.