Abstract
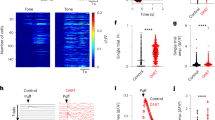
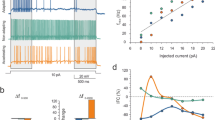
To understand the computations performed by the input layers of cortical structures, it is essential to determine the relationship between sensory-evoked synaptic input and the resulting pattern of output spikes. In the cerebellum, granule cells constitute the input layer, translating mossy fibre signals into parallel fibre input to Purkinje cells1. Until now, their small size and dense packing1,2 have precluded recordings from individual granule cells in vivo. Here we use whole-cell patch-clamp recordings to show the relationship between mossy fibre synaptic currents evoked by somatosensory stimulation and the resulting granule cell output patterns. Granule cells exhibited a low ongoing firing rate, due in part to dampening of excitability by a tonic inhibitory conductance mediated by GABAA (γ-aminobutyric acid type A) receptors. Sensory stimulation produced bursts of mossy fibre excitatory postsynaptic currents (EPSCs) that summate to trigger bursts of spikes. Notably, these spike bursts were evoked by only a few quantal EPSCs, and yet spontaneous mossy fibre inputs triggered spikes only when inhibition was reduced. Our results reveal that the input layer of the cerebellum balances exquisite sensitivity with a high signal-to-noise ratio. Granule cell bursts are optimally suited to trigger glutamate receptor activation3,4,5 and plasticity6,7,8 at parallel fibre synapses, providing a link between input representation and memory storage in the cerebellum.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eccles, J. C., Ito, M. & Szentagothai, J. The Cerebellum as a Neuronal Machine (Springer, Berlin, 1967)
Jakab, R. L. & Hamori, J. Quantitative morphology and synaptology of cerebellar glomeruli in the rat. Anat. Embryol. (Berl.) 179, 81–88 (1988)
Konnerth, A., Llano, I. & Armstrong, C. M. Synaptic currents in cerebellar Purkinje cells. Proc. Natl Acad. Sci. USA 87, 2662–2665 (1990)
Takechi, H., Eilers, J. & Konnerth, A. A new class of synaptic response involving calcium release in dendritic spines. Nature 396, 757–760 (1998)
Perkel, D. J., Hestrin, S., Sah, P. & Nicoll, R. A. Excitatory synaptic currents in Purkinje cells. Proc. R. Soc. Lond. B 241, 116–121 (1990)
Casado, M., Isope, P. & Ascher, P. Involvement of presynaptic N-methyl-d-aspartate receptors in cerebellar long-term depression. Neuron 33, 123–130 (2002)
Wang, S. S.-H., Denk, W. & Häusser, M. Coincidence detection in single dendritic spines mediated by calcium release. Nature Neurosci. 3, 1266–1273 (2000)
Brown, S. P., Brenowitz, S. D. & Regehr, W. G. Brief presynaptic bursts evoke synapse-specific retrograde inhibition mediated by endogenous cannabinoids. Nature Neurosci. 6, 1048–1057 (2003)
Silver, R. A., Traynelis, S. F. & Cull-Candy, S. G. Rapid-time-course miniature and evoked excitatory currents at cerebellar synapses in situ. Nature 355, 163–166 (1992)
Gabbiani, F., Midtgaard, J. & Knöpfel, T. Synaptic integration in a model of cerebellar granule cells. J. Neurophysiol. 72, 999–1009 (1994)
Shambes, G. M., Gibson, J. M. & Welker, W. Fractured somatotopy in granule cell tactile areas of rat cerebellar hemispheres revealed by micromapping. Brain Behav. Evol. 15, 94–140 (1978)
Bower, J. M. & Woolston, D. C. Congruence of spatial organization of tactile projections to granule cell and Purkinje cell layers of cerebellar hemispheres of the albino rat: vertical organization of cerebellar cortex. J. Neurophysiol. 49, 745–766 (1983)
Morissette, J. & Bower, J. M. Contribution of somatosensory cortex to responses in the rat cerebellar granule cell layer following peripheral tactile stimulation. Exp. Brain Res. 109, 240–250 (1996)
D'Angelo, E., De Filippi, G., Rossi, P. & Taglietti, V. Synaptic excitation of individual rat cerebellar granule cells in situ: evidence for the role of NMDA receptors. J. Physiol. (Lond.) 484, 397–413 (1995)
Brickley, S. G., Revilla, V., Cull-Candy, S. G., Wisden, W. & Farrant, M. Adaptive regulation of neuronal excitability by a voltage-independent potassium conductance. Nature 409, 88–92 (2001)
Brickley, S. G., Cull-Candy, S. G. & Farrant, M. Development of a tonic form of synaptic inhibition in rat cerebellar granule cells resulting from persistent activation of GABAA receptors. J. Physiol. (Lond.) 497, 753–759 (1996)
D'Angelo, E., De Filippi, G., Rossi, P. & Taglietti, V. Ionic mechanism of electroresponsiveness in cerebellar granule cells implicates the action of a persistent sodium current. J. Neurophysiol. 80, 493–503 (1998)
Wall, M. J. & Usowicz, M. M. Development of action potential-dependent and independent spontaneous GABAA receptor-mediated currents in granule cells of postnatal rat cerebellum. Eur. J. Neurosci. 9, 533–548 (1997)
Hamann, M., Rossi, D. J. & Attwell, D. Tonic and spillover inhibition of granule cells control information flow through cerebellar cortex. Neuron 33, 625–633 (2002)
Stell, B. M., Brickley, S. G., Tang, C. Y., Farrant, M. & Mody, I. Neuroactive steroids reduce neuronal excitability by selectively enhancing tonic inhibition mediated by δ subunit-containing GABAA receptors. Proc. Natl Acad. Sci. USA 100, 14439–14444 (2003)
Eccles, J. C., Faber, D. S., Murphy, J. T., Sabah, N. H. & Taborikova, H. Afferent volleys in limb nerves influencing impulse discharges in cerebellar cortex. I. In mossy fibers and granule cells. Exp. Brain Res. 13, 15–35 (1971)
Garwicz, M., Jörntell, H. & Ekerot, C. F. Cutaneous receptive fields and topography of mossy fibres and climbing fibres projecting to cat cerebellar C3 zone. J. Physiol. (Lond.) 512, 277–293 (1998)
Marr, D. A theory of cerebellar cortex. J. Physiol. (Lond.) 202, 437–470 (1969)
Albus, J. S. A theory of cerebellar function. Math. Biosci. 10, 25–61 (1971)
Wall, M. J. Endogenous nitric oxide modulates GABAergic transmission to granule cells in adult rat cerebellum. Eur. J. Neurosci. 18, 869–878 (2003)
Krahe, R. & Gabbiani, F. Burst firing in sensory systems. Nature Rev. Neurosci. 5, 13–23 (2004)
Lisman, J. E. Bursts as a unit of neural information: making unreliable synapses reliable. Trends Neurosci. 20, 38–43 (1997)
Hahnloser, R. H., Kozhevnikov, A. A. & Fee, M. S. An ultra-sparse code underlies the generation of neural sequences in a songbird. Nature 419, 65–70 (2002)
Margrie, T. W., Brecht, M. & Sakmann, B. In vivo, low-resistance, whole-cell recordings from neurons in the anaesthetized and awake mammalian brain. Pflügers Arch. 444, 491–498 (2002)
Llinás, R. in The Cerebellum: New Vistas (eds Palay, S. L. & Chan-Palay, V.) 189–194 (Springer, New York, 1982)
Acknowledgements
We thank B. Clark, J. Davie, M. Farrant and A. Roth for comments. This work was supported by grants from the European Union (M.H.), Wellcome Trust (M.H. and T.W.M.), Gatsby Foundation (M.H.), NHMRC (T.W.M.) and by a UCL Graduate School Research Scholarship (P.C.). T.W.M. acknowledges the MPI für medizinische Forschung, Heidelberg, for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
The relationship between bursting input and bursting output in cerebellar granule cells in vivo. (PDF 132 kb)
Supplementary Figure 2
Contribution of postsynaptic nonlinearities to granule cell integration in vivo. (PDF 151 kb)
Rights and permissions
About this article
Cite this article
Chadderton, P., Margrie, T. & Häusser, M. Integration of quanta in cerebellar granule cells during sensory processing. Nature 428, 856–860 (2004). https://doi.org/10.1038/nature02442
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02442
This article is cited by
-
Local synaptic inhibition mediates cerebellar granule cell pattern separation and enables learned sensorimotor associations
Nature Neuroscience (2024)
-
Optimal routing to cerebellum-like structures
Nature Neuroscience (2023)
-
Multi-target action of β-alanine protects cerebellar tissue from ischemic damage
Cell Death & Disease (2022)
-
Autoimmune obsessive-compulsive disorder with novel anti-CNS autoantibodies in cerebrospinal fluid
Molecular Psychiatry (2022)
-
Model simulations unveil the structure-function-dynamics relationship of the cerebellar cortical microcircuit
Communications Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.