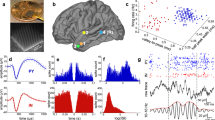
Abstract
Sensorimotor coordination emerges early in development. The maturation period is characterized by the establishment of somatotopic cortical maps1,2, the emergence of long-range cortical connections3, heightened experience-dependent plasticity4,5,6,7 and spontaneous uncoordinated skeletal movement8,9. How these various processes cooperate to allow the somatosensory system to form a three-dimensional representation of the body is not known. In the visual system, interactions between spontaneous network patterns and afferent activity have been suggested to be vital for normal development10,11. Although several intrinsic cortical patterns of correlated neuronal activity have been described in developing somatosensory cortex in vitro12,13,14, the in vivo patterns in the critical developmental period and the influence of physiological sensory inputs on these patterns remain unknown. We report here that in the intact somatosensory cortex of the newborn rat in vivo, spatially confined spindle bursts represent the first and only organized network pattern. The localized spindles are selectively triggered in a somatotopic manner by spontaneous muscle twitches8,9, motor patterns analogous to human fetal movements15,16. We suggest that the interaction between movement-triggered sensory feedback signals and self-organized spindle oscillations shapes the formation of cortical connections required for sensorimotor coordination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Killackey, H. P., Rhoades, R. W. & Bennettclarke, C. A. The formation of a cortical somatotopic map. Trends Neurosci. 18, 402–407 (1995)
Armstrong-James, M. A. Spontaneous and evoked single unit activity in 7-day rat cerebral cortex. J. Physiol. (Lond.) 208, 10P–11P (1970)
Lopez-Bendito, G. & Molnar, Z. Thalamocortical development: How are we going to get there? Nature Rev. Neurosci. 4, 276–289 (2003)
Fox, K. A critical period for experience-dependent synaptic plasticity in rat barrel cortex. J. Neurosci. 12, 1826–1838 (1992)
Fox, K. Anatomical pathways and molecular mechanisms for plasticity in the barrel cortex. Neuroscience 111, 799–814 (2002)
Feldman, D. E., Nicoll, R. A. & Malenka, R. C. Synaptic plasticity at thalamocortical synapses in developing rat somatosensory cortex: LTP, LTD, and silent synapses. J. Neurobiol. 41, 92–101 (1999)
Jain, N., Diener, P. S., Coq, J. O. & Kaas, J. H. Patterned activity via spinal dorsal quadrant inputs is necessary for the formation of organized somatosensory maps. J. Neurosci. 23, 10321–10330 (2003)
Gramsbergen, A., Schwartze, P. & Prechtl, H. F. The postnatal development of behavioral states in the rat. Dev. Psychobiol. 3, 267–280 (1970)
Petersson, P., Waldenstrom, A., Fahraeus, C. & Schouenborg, J. Spontaneous muscle twitches during sleep guide spinal self-organization. Nature 424, 72–75 (2003)
Katz, L. C. & Shatz, C. J. Synaptic activity and the construction of cortical circuits. Science 274, 1133–1138 (1996)
Katz, L. C. & Crowley, J. C. Development of cortical circuits: lessons from ocular dominance columns. Nature Rev. Neurosci. 3, 34–42 (2002)
Yuste, R., Peinado, A. & Katz, L. C. Neuronal domains in developing neocortex. Science 257, 665–669 (1992)
Yuste, R., Nelson, D. A., Rubin, W. W. & Katz, L. C. Neuronal domains in developing neocortex: mechanisms of coactivation. Neuron 14, 7–17 (1995)
Garaschuk, O., Linn, J., Eilers, J. & Konnerth, A. Large-scale oscillatory calcium waves in the immature cortex. Nature Neurosci. 3, 452–459 (2000)
de Vries, J. I., Visser, G. H. & Prechtl, H. F. The emergence of fetal behaviour. I. Qualitative aspects. Early Hum. Dev. 7, 301–322 (1982)
Clancy, B., Darlington, R. B. & Finlay, B. L. Translating developmental time across mammalian species. Neuroscience 105, 7–17 (2001)
Steriade, M. Impact of network activities on neuronal properties in corticothalamic systems. J. Neurophysiol. 86, 1–39 (2001)
Kidd, F. L. & Isaac, J. T. Developmental and activity-dependent regulation of kainate receptors at thalamocortical synapses. Nature 400, 569–573 (1999)
Ben Ari, Y. Developing networks play a similar melody. Trends Neurosci. 24, 353–360 (2001)
Ben-Ari, Y., Khazipov, R., Leinekugel, X., Caillard, O. & Gaiarsa, J. L. GABA-A, NMDA and AMPA receptors: a developmentally regulated ‘menage a trois’. Trends Neurosci. 20, 523–529 (1997)
Steriade, M., McCormick, D. A. & Sejnowski, T. J. Thalamocortical oscillations in the sleeping and aroused brain. Science 262, 679–685 (1993)
Destexhe, A. & Sejnowski, T. Thalamocortical Assemblies: How Ion Channels, Single Neurons and Large-Scale Networks Organize Sleep Oscillations (Oxford Univ. Press, New York, 2001)
Chatrian, G. E., Petersen, M. C. & Lazarte, J. A. The blocking of the rolandic wicket rhythm and some central changes related to movement. Electroencephalogr. Clin. Neurophysiol. 11(Suppl.), 497–510 (1959)
O'Donovan, M. J. The origin of spontaneous activity in developing networks of the vertebrate nervous system. Curr. Opin. Neurobiol. 9, 94–104 (1999)
Bureau, I., Shepherd, G. M. G. & Svoboda, K. Precise development of functional and anatomical columns in the neocortex. Neuron 42, 789–801 (2004)
Dreyfus-Brisac, C. & Larroche, J. C. Discontinuous electroencephalograms in the premature newborn and at term. Electro-anatomo-clinical correlations [in French]. Rev. Electroencephalogr. Neurophysiol. Clin. 1, 95–99 (1971)
Leinekugel, X. et al. Correlated bursts of activity in the neonatal hippocampus in vivo. Science 296, 2049–2052 (2002)
Margrie, T. W., Brecht, M. & Sakmann, B. In vivo, low-resistance, whole-cell recordings from neurons in the anaesthetized and awake mammalian brain. Pflugers Arch. 444, 491–498 (2002)
Karlsson, K. A. & Blumberg, M. S. Hippocampal theta in the newborn rat is revealed under conditions that promote REM sleep. J. Neurosci. 23, 1114–1118 (2003)
Bragin, A. et al. Gamma (40–100 Hz) oscillation in the hippocampus of the behaving rat. J. Neurosci. 15, 47–60 (1995)
Acknowledgements
We would like to thank T. Pankevych for helping with histological reconstruction, R. Cossart, D. Robbe, S. Montgomery and M. Zugaro for constructive comments. Supported by grants from the National Institutes of Health (G.B. and G.L.H.) and INSERM (R.K. and Y.B.-A.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Methods
This section provides detailed information about the methods used in experimental procedures in the paper. (PDF 47 kb)
Supplementary Figures S1–S6
This section contains Supplementary Figures S1–S6. Fig. S1 illustrates the persistence of spindle bursts under urethane anesthesia. Fig. S2 depicts the participation of thalamic VPL neurons in S1 spindle-bursts. Fig. S3 shows the slow spread of spindle activity across S1 cortex. Fig. S4 is a representation of body map in S1 cortex. Fig. S5 is a comparison of the latencies of movement-triggered sharp potentials and skin stimulation-evoked potentials. Fig. S6 is the properties of evoked S1 bursts under anesthesia. (PDF 1601 kb)
Supplementary Table
The Table contains quantitative data of the intracellular experiments, the parameters of spontaneous synaptic currents in neonatal rat S1 neurons. (PDF 27 kb)
Rights and permissions
About this article
Cite this article
Khazipov, R., Sirota, A., Leinekugel, X. et al. Early motor activity drives spindle bursts in the developing somatosensory cortex. Nature 432, 758–761 (2004). https://doi.org/10.1038/nature03132
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03132
This article is cited by
-
A Thalamocortical Perspective on Sleep Spindle Alterations in Neurodevelopmental Disorders
Current Sleep Medicine Reports (2024)
-
Prominent in vivo influence of single interneurons in the developing barrel cortex
Nature Neuroscience (2023)
-
Spontaneous activity in whisker-innervating region of neonatal mouse trigeminal ganglion
Scientific Reports (2022)
-
A novel somatosensory spatial navigation system outside the hippocampal formation
Cell Research (2021)
-
Developmental decrease of entorhinal-hippocampal communication in immune-challenged DISC1 knockdown mice
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.