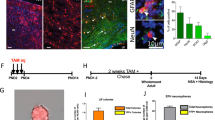
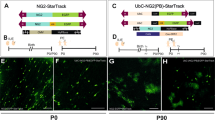
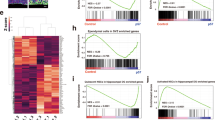
Abstract
During brain development, neurons and glia are generated from a germinal zone containing both neural stem cells (NSCs) and more limited intermediate neural progenitors (INPs)1,2,3. The signalling events that distinguish between these two proliferative neural cell types remain poorly understood. The Notch signalling pathway is known to maintain NSC character and to inhibit neurogenesis, although little is known about the role of Notch signalling in INPs. Here we show that both NSCs and INPs respond to Notch receptor activation, but that NSCs signal through the canonical Notch effector C-promoter binding factor 1 (CBF1), whereas INPs have attenuated CBF1 signalling. Furthermore, whereas knockdown of CBF1 promotes the conversion of NSCs to INPs, activation of CBF1 is insufficient to convert INPs back to NSCs. Using both transgenic and transient in vivo reporter assays we show that NSCs and INPs coexist in the telencephalic ventricular zone and that they can be prospectively separated on the basis of CBF1 activity. Furthermore, using in vivo transplantation we show that whereas NSCs generate neurons, astrocytes and oligodendrocytes at similar frequencies, INPs are predominantly neurogenic. Together with previous work on haematopoietic stem cells4, this study suggests that the use or blockade of the CBF1 cascade downstream of Notch is a general feature distinguishing stem cells from more limited progenitors in a variety of tissues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Temple, S. The development of neural stem cells. Nature 414, 112–117 (2001)
Gal, J. S. et al. Molecular and morphological heterogeneity of neural precursors in the mouse neocortical proliferative zones. J. Neurosci. 26, 1045–1056 (2006)
Merkle, F. T. & Alvarez-Buylla, A. Neural stem cells in mammalian development. Curr. Opin. Cell Biol. 18, 704–709 (2006)
Duncan, A. W. et al. Integration of Notch and Wnt signaling in hematopoietic stem cell maintenance. Nature Immunol. 6, 314–322 (2005)
Bolos, V., Grego-Bessa, J. & de la Pompa, J. L. Notch signaling in development and cancer. Endocr. Rev. 28, 339–363 (2007)
Yoon, K. & Gaiano, N. Notch signaling in the mammalian central nervous system: insights from mouse mutants. Nature Neurosci. 8, 709–715 (2005)
Chiba, S. Notch signaling in stem cell regulation. Stem Cells 24, 2437–2447 (2006)
Iso, T., Kedes, L. & Hamamori, Y. HES and HERP families: multiple effectors of the Notch signaling pathway. J. Cell. Physiol. 194, 237–255 (2003)
Yun, K. et al. Modulation of the notch signaling by Mash1 and Dlx1/2 regulates sequential specification and differentiation of progenitor cell types in the subcortical telencephalon. Development 129, 5029–5040 (2002)
Mason, H. A. et al. Notch signaling coordinates the patterning of striatal compartments. Development 132, 4247–4258 (2005)
Hsieh, J. J. et al. Truncated mammalian Notch1 activates CBF1/RBPJk-repressed genes by a mechanism resembling that of Epstein–Barr virus EBNA2. Mol. Cell. Biol. 16, 952–959 (1996)
Estrach, S., Ambler, C. A., Lo Celso, C., Hozumi, K. & Watt, F. M. Jagged 1 is a β-catenin target gene required for ectopic hair follicle formation in adult epidermis. Development 133, 4427–4438 (2006)
Hellstrom, M. et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776–780 (2007)
Lee, A. et al. Isolation of neural stem cells from the postnatal cerebellum. Nature Neurosci. 8, 723–729 (2005)
Tokunaga, A. et al. Mapping spatio-temporal activation of Notch signaling during neurogenesis and gliogenesis in the developing mouse brain. J. Neurochem. 90, 142–154 (2004)
Niwa, H., Yamamura, K. & Miyazaki, J. Efficient selection for high-expression transfectants with a novel eukaryotic vector. Gene 108, 193–199 (1991)
Waltzer, L., Bourillot, P. Y., Sergeant, A. & Manet, E. RBP-Jκ repression activity is mediated by a co-repressor and antagonized by the Epstein–Barr virus transcription factor EBNA2. Nucleic Acids Res. 23, 4939–4945 (1995)
Sawamoto, K. et al. Direct isolation of committed neuronal progenitor cells from transgenic mice coexpressing spectrally distinct fluorescent proteins regulated by stage-specific neural promoters. J. Neurosci. Res. 65, 220–227 (2001)
Ohtsuka, T. et al. Visualization of embryonic neural stem cells using Hes promoters in transgenic mice. Mol. Cell. Neurosci. 31, 109–122 (2006)
DePrimo, S. E., Stambrook, P. J. & Stringer, J. R. Human placental alkaline phosphatase as a histochemical marker of gene expression in transgenic mice. Transgenic Res. 5, 459–466 (1996)
Gaiano, N., Nye, J. S. & Fishell, G. Radial glial identity is promoted by Notch1 signaling in the murine forebrain. Neuron 26, 395–404 (2000)
Tanigaki, K. et al. Notch1 and Notch3 instructively restrict bFGF-responsive multipotent neural progenitor cells to an astroglial fate. Neuron 29, 45–55 (2001)
Gaiano, N. & Fishell, G. The role of notch in promoting glial and neural stem cell fates. Annu. Rev. Neurosci. 25, 471–490 (2002)
He, W., Ingraham, C., Rising, L., Goderie, S. & Temple, S. Multipotent stem cells from the mouse basal forebrain contribute GABAergic neurons and oligodendrocytes to the cerebral cortex during embryogenesis. J. Neurosci. 21, 8854–8862 (2001)
Battiste, J. et al. Ascl1 defines sequentially generated lineage-restricted neuronal and oligodendrocyte precursor cells in the spinal cord. Development 134, 285–293 (2007)
Mizutani, K. & Saito, T. Progenitors resume generating neurons after temporary inhibition of neurogenesis by Notch activation in the mammalian cerebral cortex. Development 132, 1295–1304 (2005)
Saito, T. & Nakatsuji, N. Efficient gene transfer into the embryonic mouse brain using in vivo electroporation. Dev. Biol. 240, 237–246 (2001)
Nakashima, K. et al. Synergistic signaling in fetal brain by STAT3–Smad1 complex bridged by p300. Science 284, 479–482 (1999)
Elbashir, S. M. et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411, 494–498 (2001)
Acknowledgements
We thank D. Hayward, D. Johns, E. Manet, T. Haydar, A. Ayoub and T. Ohtsuka for plasmids; J. Corbin for the PLAP mice; L. Blosser and A. Tam for cell sorting; R.-J. Zhao and J. Kim for technical assistance; and R. Wechsler-Reya, T. Reya, M. Starz-Gaiano, T. Haydar and S. Temple for discussions. K.M. was supported by a fellowship from the Japan Society for the Promotion of Science. This work was supported by grants from the Burroughs Wellcome Fund, the Sidney Kimmel Foundation for Cancer Research, and the National Institute of Neurological Disorders and Stroke (all to N.G.).
Author Contributions K.M. performed in utero electroporations, adherent and neurosphere cell culture experiments, quantitative RT–PCR analysis, γ-secretase inhibition and shRNA experiments. K.Y. generated and validated the TNR line, established flow cytometry staining protocols, performed γ-secretase inhibition and MEF experiments, and did the Nestin tissue staining. L.D. characterized the in vivo expression pattern of EGFP in the telencephalon of TNR embryos, performed in vivo cell transplantations, and did the CD133 tissue staining. A.T. performed clN1, EGFP and CD133 immunostainings. N.G. conceived of the TNR line, oversaw the project, and prepared the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
N.G. and K.Y. are listed by the Johns Hopkins Technology Transfer office as the inventors of the TNR mouse line; licensing of that line to for-profit entities could result in remuneration for the inventors.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1-S13 with Legends (PDF 4876 kb)
Rights and permissions
About this article
Cite this article
Mizutani, Ki., Yoon, K., Dang, L. et al. Differential Notch signalling distinguishes neural stem cells from intermediate progenitors. Nature 449, 351–355 (2007). https://doi.org/10.1038/nature06090
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06090
This article is cited by
-
Sel1l May Contributes to the Determinants of Neuronal Lineage and Neuronal Maturation Regardless of Hrd1 via Atf6-Sel1l Signaling
Neurochemical Research (2023)
-
Role of microRNA-34b-5p in cancer and injury: how does it work?
Cancer Cell International (2022)
-
Probing Notch1-Dll4 signaling in regulating osteogenic differentiation of human mesenchymal stem cells using single cell nanobiosensor
Scientific Reports (2022)
-
Methylglyoxal couples metabolic and translational control of Notch signalling in mammalian neural stem cells
Nature Communications (2020)
-
Temporal plasticity of apical progenitors in the developing mouse neocortex
Nature (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.