Abstract
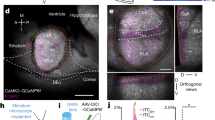
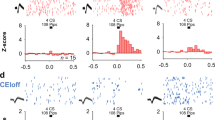
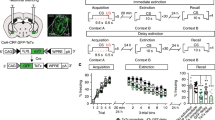
Congruent findings from studies of fear learning in animals and humans indicate that research on the circuits mediating fear constitutes our best hope of understanding human anxiety disorders1,2,3,4. In mammals, repeated presentations of a conditioned stimulus that was previously paired to a noxious stimulus leads to the gradual disappearance of conditioned fear responses. Although much evidence suggests that this extinction process depends on plastic events in the amygdala1,2,3,4,5,6,7, the underlying mechanisms remain unclear. Intercalated (ITC) amygdala neurons constitute probable mediators of extinction because they receive information about the conditioned stimulus from the basolateral amygdala (BLA)8,9, and contribute inhibitory projections to the central nucleus (CEA)10,11, the main output station of the amygdala for conditioned fear responses12. Thus, after extinction training, ITC cells could reduce the impact of conditioned-stimulus-related BLA inputs to the CEA by means of feed-forward inhibition. Here we test the hypothesis that ITC neurons mediate extinction by lesioning them with a toxin that selectively targets cells expressing µ-opioid receptors (µORs). Electron microscopic observations revealed that the incidence of µOR-immunoreactive synapses is much higher in ITC cell clusters than in the BLA or CEA and that µORs typically have a post-synaptic location in ITC cells. In keeping with this, bilateral infusions of the µOR agonist dermorphin conjugated to the toxin saporin in the vicinity of ITC neurons caused a 34% reduction in the number of ITC cells but no significant cell loss in surrounding nuclei. Moreover, ITC lesions caused a marked deficit in the expression of extinction that correlated negatively with the number of surviving ITC neurons but not CEA cells. Because ITC cells exhibit an unusual pattern of receptor expression, these findings open new avenues for the treatment of anxiety disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Myers, K. M. & Davis, M. Mechanisms of fear extinction. Mol. Psychiatry 12, 120–150 (2007)
Phelps, E. A. & LeDoux, J. E. Contributions of the amygdala to emotion processing: from animal models to human behavior. Neuron 48, 175–187 (2005)
Quirk, G. J. & Mueller, D. Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology 33, 56–72 (2008)
Ressler, K. J. & Mayberg, H. S. Targeting abnormal neural circuits in mood and anxiety disorders: from the laboratory to the clinic. Nature Neurosci. 10, 1116–1124 (2007)
Falls, W. A., Miserendino, M. J. D. & Davis, M. Extinction of fear-potentiated startle: blockade by infusion of an NMDA antagonist into the amygdala. J. Neurosci. 12, 854–863 (1992)
Sotres-Bayon, F., Bush, D. E. & LeDoux, J. E. Acquisition of fear extinction requires activation of NR2B-containing NMDA receptors in the lateral amygdala. Neuropsychopharmacology 32, 1929–1940 (2007)
Walker, D. L., Ressler, K. J., Lu, K. T. & Davis, M. Facilitation of conditioned fear extinction by systemic administration or intra-amygdala infusions of d-Cycloserine as assessed with fear-potentiated startle in rats. J. Neurosci. 22, 2343–2351 (2002)
Marowsky, A., Yanagawa, Y., Obata, K. & Vogt, K. E. A specialized subclass of interneurons mediates dopaminergic facilitation of amygdala function. Neuron 48, 1025–1037 (2005)
Royer, S., Martina, M. & Paré, D. An inhibitory interface gates impulse traffic between the input and output stations of the amygdala. J. Neurosci. 19, 10575–10583 (1999)
Paré, D. & Smith, Y. Distribution of GABA immunoreactivity in the amygdaloid complex of the cat. Neuroscience 57, 1061–1076 (1993)
Paré, D. & Smith, Y. The intercalated cell masses project to the central and medial nuclei of the amygdala in cats. Neuroscience 57, 1077–1090 (1993)
Davis, M. in The Amygdala: A Functional Analysis (ed. Aggleton, J. P.) 213–287 (Oxford Univ. Press, 2000)
Royer, S. & Paré, D. Bidirectional synaptic plasticity in intercalated amygdala neurons and the extinction of conditioned fear responses. Neuroscience 115, 455–462 (2002)
Jacobsen, K. X., Hoistad, M., Staines, W. A. & Fuxe, K. The distribution of dopamine D1 receptor and mu-opioid receptor 1 receptor immunoreactivities in the amygdala and interstitial nucleus of the posterior limb of the anterior commissure: relationships to tyrosine hydroxylase and opioid peptide terminal systems. Neuroscience 141, 2007–2018 (2006)
Poulin, J. F., Chevalier, B., Laforest, S. & Drolet, G. Enkephalinergic afferents of the centromedial amygdala in the rat. J. Comp. Neurol. 496, 859–876 (2006)
Wiley, R. G. & Lappi, D. A. Targeted toxins in pain. Adv. Drug Deliv. Rev. 55, 1043–1054 (2003)
Gaudriault, G., Nouel, D., Dal Farra, C., Beaudet, A. & Vincent, J. P. Receptor-induced internalization of selective peptidic mu and delta opioid ligands. J. Biol. Chem. 272, 2880–2888 (1997)
Harris, J. A. & Westbrook, R. F. Evidence that GABA transmission mediates context-specific extinction of learned fear. Psychopharmacology (Berl.) 140, 105–115 (1998)
Lee, H. & Kim, J. J. Amygdalar NMDA receptors are critical for new fear learning in previously fear-conditioned rats. J. Neurosci. 18, 8444–8454 (1998)
Hobin, J. A., Goosens, K. A. & Maren, S. Context-dependent neuronal activity in the lateral amygdala represents fear memories after extinction. J. Neurosci. 23, 8410–8416 (2003)
Repa, J. C. et al. Two different lateral amygdala cell populations contribute to the initiation and storage of memory. Nature Neurosci. 4, 724–731 (2001)
McDonald, A. J., Mascagni, F. & Guo, L. Projections of the medial and lateral prefrontal cortices to the amygdala: A Phaseolus vulgaris leucoagglutinin study in the rat. Neuroscience 71, 55–75 (1996)
Milad, M. R. & Quirk, G. J. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature 420, 70–74 (2002)
Quirk, G. J., Likhtik, E., Pelletier, J. G. & Paré, D. Stimulation of medial prefrontal cortex decreases the responsiveness of central amygdala output neurons. J. Neurosci. 23, 8800–8807 (2003)
Royer, S. & Paré, D. Conservation of total synaptic weight through balanced synaptic depression and potentiation. Nature 422, 518–522 (2003)
Burgos-Robles, A., Vidal-Gonzalez, I., Santini, E. & Quirk, G. J. Consolidation of fear extinction requires NMDA receptor-dependent bursting in the ventromedial prefrontal cortex. Neuron 53, 871–880 (2007)
Milad, M. R. et al. Presence and acquired origin of reduced recall for fear extinction in PTSD: Results of a twin study. J. Psychiatr. Res. 42, 515–520 (2008)
Bremner, J. D., Elzinga, B., Schmahl, C. & Vermetten, E. Structural and functional plasticity of the human brain in posttraumatic stress disorder. Prog. Brain Res. 167, 171–186 (2008)
Shin, L. M., Rauch, S. L. & Pitman, R. K. Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Ann. NY Acad. Sci. 1071, 67–79 (2006)
Fuxe, K. et al. The dopamine D1 receptor-rich main and paracapsular intercalated nerve cell groups of the rat amygdala: relationship to the dopamine innervation. Neuroscience 119, 733–746 (2003)
Acknowledgements
This work was supported by RO1 grant MH-073610 to D.P. and NRSA fellowship F31 MH76415 to E.L. from NIMH.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information 1
The file contains Supplementary Figures 1-3 with Legends. (PDF 602 kb)
Rights and permissions
About this article
Cite this article
Likhtik, E., Popa, D., Apergis-Schoute, J. et al. Amygdala intercalated neurons are required for expression of fear extinction. Nature 454, 642–645 (2008). https://doi.org/10.1038/nature07167
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07167
This article is cited by
-
Differential recruitment of brain circuits during fear extinction in non-stressed compared to stress resilient animals
Scientific Reports (2024)
-
Dynamic Changes of the Infralimbic Cortex and Its Regulation of the Prelimbic Cortex in Rats with Chronic Inflammatory Pain
Neuroscience Bulletin (2024)
-
Exploration driven by a medial preoptic circuit facilitates fear extinction in mice
Communications Biology (2023)
-
Prefrontal circuits encode both general danger and specific threat representations
Nature Neuroscience (2023)
-
Memory Trace for Fear Extinction: Fragile yet Reinforceable
Neuroscience Bulletin (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.