Abstract
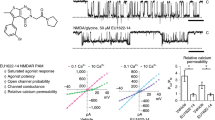
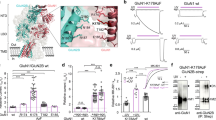
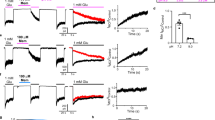
N-methyl-d-aspartate (NMDA) receptors (NMDARs) are a major class of excitatory neurotransmitter receptors in the central nervous system. They form glutamate-gated ion channels that are highly permeable to calcium and mediate activity-dependent synaptic plasticity1. NMDAR dysfunction is implicated in multiple brain disorders, including stroke, chronic pain and schizophrenia2. NMDARs exist as multiple subtypes with distinct pharmacological and biophysical properties that are largely determined by the type of NR2 subunit (NR2A to NR2D) incorporated in the heteromeric NR1/NR2 complex1,3,4. A fundamental difference between NMDAR subtypes is their channel maximal open probability (Po), which spans a 50-fold range from about 0.5 for NR2A-containing receptors to about 0.01 for receptors containing NR2C and NR2D; NR2B-containing receptors have an intermediate value (about 0.1)5,6,7,8,9. These differences in Po confer unique charge transfer capacities and signalling properties on each receptor subtype4,6,10,11. The molecular basis for this profound difference in activity between NMDAR subtypes is unknown. Here we show that the subunit-specific gating of NMDARs is controlled by the region formed by the NR2 amino-terminal domain (NTD), an extracellular clamshell-like domain previously shown to bind allosteric inhibitors12,13,14,15, and the short linker connecting the NTD to the agonist-binding domain (ABD). The subtype specificity of NMDAR Po largely reflects differences in the spontaneous (ligand-independent) equilibrium between open-cleft and closed-cleft conformations of the NR2-NTD. This NTD-driven gating control also affects pharmacological properties by setting the sensitivity to the endogenous inhibitors zinc and protons. Our results provide a proof of concept for a drug-based bidirectional control of NMDAR activity by using molecules acting either as NR2-NTD ‘closers’ or ‘openers’ promoting receptor inhibition or potentiation, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dingledine, R., Borges, K., Bowie, D. & Traynelis, S. F. The glutamate receptor ion channels. Pharmacol. Rev. 51, 7–61 (1999)
Kemp, J. A. & McKernan, R. M. NMDA receptor pathways as drug targets. Nature Neurosci. 5 (Suppl.). 1039–1042 (2002)
Paoletti, P. & Neyton, J. NMDA receptor subunits: function and pharmacology. Curr. Opin. Pharmacol. 7, 39–47 (2007)
Cull-Candy, S. G. & Leszkiewicz, D. N. Role of distinct NMDA receptor subtypes at central synapses. Sci. STKE 2004, re16 (2004)
Chen, N., Luo, T. & Raymond, L. A. Subtype-dependence of NMDA receptor channel open probability. J. Neurosci. 19, 6844–6854 (1999)
Erreger, K., Dravid, S. M., Banke, T. G., Wyllie, D. J. & Traynelis, S. F. Subunit-specific gating controls rat NR1/NR2A and NR1/NR2B NMDA channel kinetics and synaptic signalling profiles. J. Physiol. (Lond.) 563, 345–358 (2005)
Wyllie, D. J., Behe, P. & Colquhoun, D. Single-channel activations and concentration jumps: comparison of recombinant NR1a/NR2A and NR1a/NR2D NMDA receptors. J. Physiol. (Lond.) 510, 1–18 (1998)
Dravid, S. M., Prakash, A. & Traynelis, S. F. Activation of recombinant NR1/NR2C NMDA receptors. J. Physiol. (Lond.) 586, 4425–4439 (2008)
Popescu, G., Robert, A., Howe, J. R. & Auerbach, A. Reaction mechanism determines NMDA receptor response to repetitive stimulation. Nature 430, 790–793 (2004)
Liu, Y. et al. NMDA receptor subunits have differential roles in mediating excitotoxic neuronal death both in vitro and in vivo . J. Neurosci. 27, 2846–2857 (2007)
Liu, L. et al. Role of NMDA receptor subtypes in governing the direction of hippocampal synaptic plasticity. Science 304, 1021–1024 (2004)
Paoletti, P. et al. Molecular organization of a zinc binding N-terminal modulatory domain in a NMDA receptor subunit. Neuron 28, 911–925 (2000)
Low, C. M., Zheng, F., Lyuboslavsky, P. & Traynelis, S. F. Molecular determinants of coordinated proton and zinc inhibition of N-methyl-d-aspartate NR1/NR2A receptors. Proc. Natl Acad. Sci. USA 97, 11062–11067 (2000)
Choi, Y. B. & Lipton, S. A. Identification and mechanism of action of two histidine residues underlying high-affinity Zn2+ inhibition of the NMDA receptor. Neuron 23, 171–180 (1999)
Perin-Dureau, F., Rachline, J., Neyton, J. & Paoletti, P. Mapping the binding site of the neuroprotectant ifenprodil on NMDA receptors. J. Neurosci. 22, 5955–5965 (2002)
Jones, K. S., VanDongen, H. M. & VanDongen, A. M. The NMDA receptor M3 segment is a conserved transduction element coupling ligand binding to channel opening. J. Neurosci. 22, 2044–2053 (2002)
Yuan, H., Erreger, K., Dravid, S. M. & Traynelis, S. F. Conserved structural and functional control of N-methyl-d-aspartate receptor gating by transmembrane domain M3. J. Biol. Chem. 280, 29708–29716 (2005)
Blanke, M. L. & VanDongen, A. M. Constitutive activation of the N-methyl-d-aspartate receptor via cleft-spanning disulfide bonds. J. Biol. Chem. 283, 21519–21529 (2008)
Rachline, J., Perin-Dureau, F., Le Goff, A., Neyton, J. & Paoletti, P. The micromolar zinc-binding domain on the NMDA receptor subunit NR2B. J. Neurosci. 25, 308–317 (2005)
Gielen, M. et al. Structural rearrangements of NR1/NR2A NMDA receptors during allosteric inhibition. Neuron 57, 80–93 (2008)
Sun, Y. et al. Mechanism of glutamate receptor desensitization. Nature 417, 245–253 (2002)
Mayer, M. L. Glutamate receptors at atomic resolution. Nature 440, 456–462 (2006)
Tang, C., Schwieters, C. D. & Clore, G. M. Open-to-closed transition in apo maltose-binding protein observed by paramagnetic NMR. Nature 449, 1078–1082 (2007)
Kniazeff, J. et al. Locking the dimeric GABAB G-protein-coupled receptor in its active state. J. Neurosci. 24, 370–377 (2004)
Mony, L. et al. Structural basis of NR2B-selective antagonist recognition by N-methyl-d-aspartate receptors. Mol. Pharmacol. 75, 60–74 (2009)
Zheng, F. et al. Allosteric interaction between the amino terminal domain and the ligand binding domain of NR2A. Nature Neurosci. 4, 894–901 (2001)
Marvin, J. S. & Hellinga, H. W. Manipulation of ligand binding affinity by exploitation of conformational coupling. Nature Struct. Biol. 8, 795–798 (2001)
Low, C. M. et al. Molecular determinants of proton-sensitive N-methyl-d-aspartate receptor gating. Mol. Pharmacol. 63, 1212–1222 (2003)
Lisman, J. E. et al. Circuit-based framework for understanding neurotransmitter and risk gene interactions in schizophrenia. Trends Neurosci. 31, 234–242 (2008)
Furukawa, H., Singh, S. K., Mancusso, R. & Gouaux, E. Subunit arrangement and function in NMDA receptors. Nature 438, 185–192 (2005)
Paoletti, P., Ascher, P. & Neyton, J. High-affinity zinc inhibition of NMDA NR1-NR2A receptors. J. Neurosci. 17, 5711–5725 (1997)
Erreger, K. & Traynelis, S. F. Zinc inhibition of rat NR1/NR2A N-methyl-d-aspartate receptors. J. Physiol. (Lond.) 586, 763–778 (2008)
Schorge, S., Elenes, S. & Colquhoun, D. Maximum likelihood fitting of single channel NMDA activity with a mechanism composed of independent dimers of subunits. J. Physiol. (Lond.) 569, 395–418 (2005)
Colquhoun, D. & Sigworth, F. J. in Single-channel Recording (eds Sakmann, B. & Neher, E.) 483–587 (Plenum, 1995)
Sigworth, F. J. & Sine, S. M. Data transformations for improved display and fitting of single-channel dwell time histograms. Biophys. J. 52, 1047–1054 (1987)
Jackson, M. B., Wong, B. S., Morris, C. E., Lecar, H. & Christian, C. N. Successive openings of the same acetylcholine receptor channel are correlated in open time. Biophys. J. 42, 109–114 (1983)
Kunishima, N. et al. Structural basis of glutamate recognition by a dimeric metabotropic glutamate receptor. Nature 407, 971–977 (2000)
Doyle, D. A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998)
Acknowledgements
We thank B. Barbour, P.-J. Corringer, J. Neyton and D. Stroebel for comments on the manuscript, and S. Carvalho, M. Casado and M. Gendrel for experimental help. This work was supported by the Ministère de la Recherche (M.G., L.M.), the Université Pierre et Marie Curie (UPMC) and the Fondation pour la Recherche Médicale (FRM) (M.G.), NIH grant R01 MH045817 (J.W.J.), the Institut National de la Santé et de la Recherche Médicale (INSERM), the Agence Nationale pour la Recherche (ANR), GlaxoSmithKline and an Équipe FRM grant (P.P.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Table S1-S2 and Supplementary Figures S1-S9 with Legends. (PDF 9020 kb)
Rights and permissions
About this article
Cite this article
Gielen, M., Retchless, B., Mony, L. et al. Mechanism of differential control of NMDA receptor activity by NR2 subunits. Nature 459, 703–707 (2009). https://doi.org/10.1038/nature07993
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07993
This article is cited by
-
Features of the response to subchronic low-dose exposure to copper oxide nanoparticles in rats
Scientific Reports (2023)
-
GSK-126 Attenuates Cell Apoptosis in Ischemic Brain Injury by Modulating the EZH2-H3K27me3-Bcl2l1 Axis
Molecular Neurobiology (2023)
-
GluN2A and GluN2B NMDA receptors use distinct allosteric routes
Nature Communications (2021)
-
Splicing and editing of ionotropic glutamate receptors: a comprehensive analysis based on human RNA-Seq data
Cellular and Molecular Life Sciences (2021)
-
Structural basis of subtype-selective competitive antagonism for GluN2C/2D-containing NMDA receptors
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.