Abstract
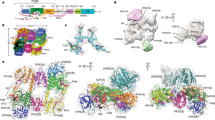
Phytochromes are a collection of bilin-containing photoreceptors that regulate numerous photoresponses in plants and microorganisms through their ability to photointerconvert between a red-light-absorbing, ground state (Pr) and a far-red-light-absorbing, photoactivated state (Pfr)1,2. Although the structures of several phytochromes as Pr have been determined3,4,5,6,7, little is known about the structure of Pfr and how it initiates signalling. Here we describe the three-dimensional solution structure of the bilin-binding domain as Pfr, using the cyanobacterial phytochrome from Synechococcus OSB′. Contrary to predictions, light-induced rotation of the A pyrrole ring but not the D ring is the primary motion of the chromophore during photoconversion. Subsequent rearrangements within the protein then affect intradomain and interdomain contact sites within the phytochrome dimer. On the basis of our models, we propose that phytochromes act by propagating reversible light-driven conformational changes in the bilin to altered contacts between the adjacent output domains, which in most phytochromes direct differential phosphotransfer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rockwell, N. C., Su, Y. S. & Lagarias, J. C. Phytochrome structure and signaling mechanisms. Annu. Rev. Plant Biol. 57, 837–858 (2006)
Vierstra, R. D. & Karniol, B. in Handbook of Photosensory Receptors (eds Briggs, W. R. & Spudich, J. L.) 171–196 (Wiley, 2005)
Essen, L. O., Mailliet, J. & Hughes, J. The structure of a complete phytochrome sensory module in the Pr ground state. Proc. Natl Acad. Sci. USA 105, 14709–14714 (2008)
Cornilescu, G., Ulijasz, A. T., Cornilescu, C. C., Markley, J. L. & Vierstra, R. D. Solution structure of a cyanobacterial phytochrome GAF domain in the red-light-absorbing ground state. J. Mol. Biol. 383, 403–413 (2008)
Wagner, J. R., Brunzelle, J. S., Forest, K. T. & Vierstra, R. D. A light-sensing knot revealed by the structure of the chromophore-binding domain of phytochrome. Nature 438, 325–331 (2005)
Wagner, J. R., Zhang, J., Brunzelle, J. S., Vierstra, R. D. & Forest, K. T. High resolution structure of Deinococcus bacteriophytochrome yields new insights into phytochrome architecture and evolution. J. Biol. Chem. 282, 12298–12309 (2007)
Yang, X., Kuk, J. & Moffat, K. Crystal structure of Pseudomonas aeruginosa bacteriophytochrome: photoconversion and signal transduction. Proc. Natl Acad. Sci. USA 105, 14715–14720 (2008)
Quail, P. H. Phytochrome photosensory signalling networks. Nature Rev. Mol. Cell Biol. 3, 85–93 (2002)
Rudiger, W., Thummler, F., Cmiel, E. & Schneider, S. Chromophore structure of the physiologically active form (Pfr) of phytochrome. Proc. Natl Acad. Sci. USA 80, 6244–6248 (1983)
Kneip, C. et al. Protonation state and structural changes of the tetrapyrrole chromophore during the Pr → Pfr phototransformation of phytochrome: a resonance Raman spectroscopic study. Biochemistry 38, 15185–15192 (1999)
Inomata, K. et al. Sterically locked synthetic bilin derivatives and phytochrome Agp1 from Agrobacterium tumefaciens form photosensitive Pr- and Pfr-like adducts. J. Biol. Chem. 280, 24491–24497 (2005)
Inomata, K. et al. Assembly of synthetic locked chromophores with Agrobacterium phytochromes Agp1 and Agp2. J. Biol. Chem. 281, 28162–28173 (2006)
Fodor, S. P., Lagarias, J. C. & Mathies, R. A. Resonance Raman analysis of the Pr and Pfr forms of phytochrome. Biochemistry 29, 11141–11146 (1990)
Yang, X., Kuk, J. & Moffat, K. Conformational differences between the Pfr and Pr states of Pseudomonas aeruginosa bacteriophytochrome. Proc. Natl Acad. Sci. USA 106, 15639–15644 (2009)
Ulijasz, A. T. et al. Characterization of two thermostable cyanobacterial phytochromes reveals global movements in the chromophore-binding domain during photoconversion. J. Biol. Chem. 283, 21251–21266 (2008)
Wagner, J. R. et al. Mutational analysis of Deinococcus radiodurans bacteriophytochrome reveals key amino acids necessary for the photochromicity and proton exchange cycle of phytochromes. J. Biol. Chem. 283, 12212–12226 (2008)
von Stetten, D. et al. Highly conserved residues Asp-197 and His-250 in Agp1 phytochrome control the proton affinity of the chromophore and Pfr formation. J. Biol. Chem. 282, 2116–2123 (2007)
van Thor, J. J. et al. Light-induced proton release and proton uptake reactions in the cyanobacterial phytochrome Cph1. Biochemistry 40, 11460–11471 (2001)
Borucki, B. et al. Light-induced proton release of phytochrome is coupled to the transient deprotonation of the tetrapyrrole chromophore. J. Biol. Chem. 280, 34358–34364 (2005)
Fischer, A. J. et al. Multiple roles of a conserved GAF domain tyrosine residue in cyanobacterial and plant phytochromes. Biochemistry 44, 15203–15215 (2005)
Oka, Y., Matsushita, T., Mochizuki, N., Quail, P. H. & Nagatani, A. Mutant screen distinguishes between residues necessary for light-signal perception and signal transfer by phytochrome B. PLoS Genet. 4, e1000158 (2008)
Karniol, B., Wagner, J. R., Walker, J. M. & Vierstra, R. D. Phylogenetic analysis of the phytochrome superfamily reveals distinct microbial subfamilies of photoreceptors. Biochem. J. 392, 103–116 (2005)
Rohmer, T. et al. Light-induced chromophore activity and signal transduction in phytochromes observed by 13C and 15N magic-angle spinning NMR. Proc. Natl Acad. Sci. USA 105, 15229–15234 (2008)
Seibeck, S. et al. Locked 5Zs-biliverdin blocks the Meta-RA to Meta-RC transition in the functional cycle of bacteriophytochrome Agp1. FEBS Lett. 581, 5425–5429 (2007)
Inomata, K. et al. Assembly of Agrobacterium phytochromes Agp1 and Agp2 with doubly locked bilin chromophores. Biochemistry 48, 2817–2827 (2009)
van Thor, J. J., Mackeen, M., Kuprov, I., Dwek, R. A. & Wormald, M. R. Chromophore structure in the photocycle of the cyanobacterial phytochrome Cph1. Biophys. J. 91, 1811–1822 (2006)
Strauss, H. M., Hughes, J. & Schmieder, P. Heteronuclear solution-state NMR studies of the chromophore in cyanobacterial phytochrome Cph1. Biochemistry 44, 8244–8250 (2005)
Yang, X., Stojkovic, E. A., Kuk, J. & Moffat, K. Crystal structure of the chromophore binding domain of an unusual bacteriophytochrome, RpBphP3, reveals residues that modulate photoconversion. Proc. Natl Acad. Sci. USA 104, 12571–12576 (2007)
Harper, S. M., Neil, L. C. & Gardner, K. H. Structural basis of a phototropin light switch. Science 301, 1541–1544 (2003)
Vreede, J., Crielaard, W., Hellingwerf, K. J. & Bolhuis, P. G. Predicting the signaling state of photoactive yellow protein. Biophys. J. 88, 3525–3535 (2005)
Cornilescu, G., Ulijasz, A. T., Cornilescu, C. C., Markley, J. L. & Vierstra, R. D. Solution structure of a cyanobacterial phytochrome GAF domain in the red-light-absorbing ground state. J. Mol. Biol. 383, 403–413 (2008)
Ulijasz, A. T. et al. Characterization of two thermostable cyanobacterial phytochromes reveals global movements in the chromophore-binding domain during photoconversion. J. Biol. Chem. 283, 21251–21266 (2008)
Ulmer, T. S., Ramirez, B. E., Delaglio, F. & Bax, A. Evaluation of backbone proton positions and dynamics in a small protein by liquid crystal NMR spectroscopy. J. Am. Chem. Soc. 125, 9179–9191 (2003)
Cornilescu, G. & Bax, A. Measurement of proton, nitrogen, and carbonyl chemical shielding anisotropies in a protein dissolved in a dilute liquid crystalline phase. J. Am. Chem. Soc. 122, 10143–10154 (2000)
Ottiger, M., Delaglio, F., Marquardt, J. L., Tjandra, N. & Bax, A. Measurement of dipolar couplings for methylene and methyl sites in weakly oriented macromolecules and their use in structure determination. J. Magn. Reson. 134, 365–369 (1998)
Cornilescu, G., Delaglio, F. & Bax, A. Protein backbone angle restraints from searching a database for chemical shift and sequence homology. J. Biomol. NMR 13, 289–302 (1999)
Wüthrich, K. NMR of Proteins and Nucleic Acids (Wiley Interscience, 1986)
Bishop, J. E. et al. Phycobiliprotein-bilin linkage diversity. I. Structural studies on A- and D-ring-linked phycocyanobilins. J. Biol. Chem. 261, 6790–6796 (1986)
Lagarias, J. C., Glazer, A. N. & Rapoport, H. Chromopeptides from C-phycocyanin. Structure and linkage of a phycocyanobilin bound to the β subunit. J. Am. Chem. Soc. 101, 5030–5037 (1979)
Schwieters, C. D., Kuszewski, J. J., Tjandra, N. & Clore, G. M. The Xplor-NIH NMR molecular structure determination package. J. Magn. Reson. 160, 65–73 (2003)
Schuttelkopf, A. W. & van Aalten, D. M. PRODRG: a tool for high-throughput crystallography of protein–ligand complexes. Acta Crystallogr. D 60, 1355–1363 (2004)
Garrett, D. S., Powers, R., Gronenborn, A. M. & Clore, G. M. A common sense approach to peak picking two-, three- and four-dimensional spectra using automatic computer analysis of contour diagrams. J. Magn. Reson. 95, 214–220 (1991)
Ottiger, M. & Bax, A. Bicelle-based liquid crystals for NMR-measurement of dipolar couplings at acidic and basic pH values. J. Biomol. NMR 13, 187–191 (1999)
Tjandra, N. & Bax, A. Direct measurement of distances and angles in biomolecules by NMR in a dilute liquid crystalline medium. Science 278, 1111–1114 (1997)
Laskowski, R. A., MacArthur, M. W., Moss, D. S. & Thornton, J. M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Cryst. 26, 283–291 (1993)
Acknowledgements
We thank K. T. Forest and W. M. Westler for technical advice. This work was supported by a grant from the US National Science Foundation (R.D.V.) and a postdoctoral fellowship from the American Heart Association (A.T.U.). C.C.C. was supported by the U.S. National Institutes of Health. This study was a collaboration with the National Magnetic Resonance Facility at Madison, which is supported by the US National Institute of Health.
Author Contributions R.D.V. and A.T.U. initiated the collaboration. A.T.U. and J.Z. purified the chromoproteins and A.T.U. characterized the samples. M.R. provided the isotopically labelled ALA. G.C. collected the NMR spectra and solved the NMR structures with C.C.C. and J.L.M. All authors interpreted the three-dimensional structures. R.D.V., A.T.U., C.C.C. and G.C. prepared the manuscript, tables and figures.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Table 1, Supplementary Figures 1-7 with Legends and Supplementary References. The y-axis of Supplementary Fig. 4e was corrected on January 25 2010. (PDF 2162 kb)
Rights and permissions
About this article
Cite this article
Ulijasz, A., Cornilescu, G., Cornilescu, C. et al. Structural basis for the photoconversion of a phytochrome to the activated Pfr form. Nature 463, 250–254 (2010). https://doi.org/10.1038/nature08671
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08671
This article is cited by
-
Light- and pH-dependent structural changes in cyanobacteriochrome AnPixJg2
Photochemical & Photobiological Sciences (2022)
-
Far-red light acclimation in diverse oxygenic photosynthetic organisms
Photosynthesis Research (2019)
-
Sequences, Domain Architectures, and Biological Functions of the Serine/Threonine and Histidine Kinases in Synechocystis sp. PCC 6803
Applied Biochemistry and Biotechnology (2019)
-
Mapping light-driven conformational changes within the photosensory module of plant phytochrome B
Scientific Reports (2016)
-
Identification of a new electron-transfer relaxation pathway in photoexcited pyrrole dimers
Nature Communications (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.