Abstract
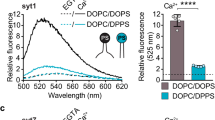
Neuronal exocytosis is catalysed by the SNAP receptor protein syntaxin-1A1, which is clustered in the plasma membrane at sites where synaptic vesicles undergo exocytosis2,3. However, how syntaxin-1A is sequestered is unknown. Here we show that syntaxin clustering is mediated by electrostatic interactions with the strongly anionic lipid phosphatidylinositol-4,5-bisphosphate (PIP2). Using super-resolution stimulated-emission depletion microscopy on the plasma membranes of PC12 cells, we found that PIP2 is the dominant inner-leaflet lipid in microdomains about 73 nanometres in size. This high accumulation of PIP2 was required for syntaxin-1A sequestering, as destruction of PIP2 by the phosphatase synaptojanin-1 reduced syntaxin-1A clustering. Furthermore, co-reconstitution of PIP2 and the carboxy-terminal part of syntaxin-1A in artificial giant unilamellar vesicles resulted in segregation of PIP2 and syntaxin-1A into distinct domains even when cholesterol was absent. Our results demonstrate that electrostatic protein–lipid interactions can result in the formation of microdomains independently of cholesterol or lipid phases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jahn, R. & Scheller, R. H. SNAREs – engines for membrane fusion. Nature Rev. Mol. Cell Biol. 7, 631–643 (2006)
Aoyagi, K. et al. The activation of exocytotic sites by the formation of phosphatidylinositol 4,5-bisphosphate microdomains at syntaxin clusters. J. Biol. Chem. 280, 17346–17352 (2005)
Lang, T. et al. SNAREs are concentrated in cholesterol-dependent clusters that define docking and fusion sites for exocytosis. EMBO J. 20, 2202–2213 (2001)
McLaughlin, S., Wang, J., Gambhir, A. & Murray, D. PIP(2) and proteins: interactions, organization, and information flow. Annu. Rev. Biophys. Biomol. Struct. 31, 151–175 (2002)
McLaughlin, S. & Murray, D. Plasma membrane phosphoinositide organization by protein electrostatics. Nature 438, 605–611 (2005)
Wen, P. J., Osborne, S. L. & Meunier, F. A. Dynamic control of neuroexocytosis by phosphoinositides in health and disease. Prog. Lipid Res. 50, 52–61 (2011)
Milosevic, I. et al. Plasmalemmal phosphatidylinositol-4,5-bisphosphate level regulates the releasable vesicle pool size in chromaffin cells. J. Neurosci. 25, 2557–2565 (2005)
Hay, J. C. & Martin, T. F. Phosphatidylinositol transfer protein required for ATP-dependent priming of Ca2+-activated secretion. Nature 366, 572–575 (1993)
James, D. J., Khodthong, C., Kowalchyk, J. A. & Martin, T. F. Phosphatidylinositol 4,5-bisphosphate regulates SNARE-dependent membrane fusion. J. Cell Biol. 182, 355–366 (2008)
Griesbeck, O., Baird, G. S., Campbell, R. E., Zacharias, D. A. & Tsien, R. Y. Reducing the environmental sensitivity of yellow fluorescent protein. Mechanism and applications. J. Biol. Chem. 276, 29188–29194 (2001)
Hell, S. W. & Wichmann, J. Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt. Lett. 19, 780–782 (1994)
Sieber, J. J. et al. Anatomy and dynamics of a supramolecular membrane protein cluster. Science 317, 1072–1076 (2007)
Williams, D., Vicôgne, J., Zaitseva, I., McLaughlin, S. & Pessin, J. E. Evidence that electrostatic interactions between vesicle-associated membrane protein 2 and acidic phospholipids may modulate the fusion of transport vesicles with the plasma membrane. Mol. Biol. Cell 20, 4910–4919 (2009)
Denisov, G., Wanaski, S., Luan, P., Glaser, M. & McLaughlin, S. Binding of basic peptides to membranes produces lateral domains enriched in the acidic lipids phosphatidylserine and phosphatidylinositol 4,5-bisphosphate: an electrostatic model and experimental results. Biophys. J. 74, 731–744 (1998)
Kweon, D. H., Kim, C. S. & Shin, Y. K. The membrane-dipped neuronal SNARE complex: a site-directed spin labeling electron paramagnetic resonance study. Biochemistry 41, 9264–9268 (2002)
Lam, A. D., Tryoen-Toth, P., Tsai, B., Vitale, N. & Stuenkel, E. L. SNARE-catalyzed fusion events are regulated by syntaxin1A-lipid interactions. Mol. Biol. Cell 19, 485–497 (2008)
Murray, D. H. & Tamm, L. K. Clustering of syntaxin-1A in model membranes is modulated by phosphatidylinositol 4,5-bisphosphate and cholesterol. Biochemistry 48, 4617–4625 (2009)
Bacia, K., Schuette, C. G., Kahya, N., Jahn, R. & Schwille, P. SNAREs prefer liquid-disordered over “raft” (liquid-ordered) domains when reconstituted into giant unilamellar vesicles. J. Biol. Chem. 279, 37951–37955 (2004)
Carvalho, K., Ramos, L., Roy, C. & Picart, C. Giant unilamellar vesicles containing phosphatidylinositol(4,5)bisphosphate: characterization and functionality. Biophys. J. 95, 4348–4360 (2008)
Levental, I. et al. Calcium-dependent lateral organization in phosphatidylinositol 4,5-bisphosphate (PIP2)- and cholesterol-containing monolayers. Biochemistry 48, 8241–8248 (2009)
Christian, D. A. et al. Spotted vesicles, striped micelles and Janus assemblies induced by ligand binding. Nature Mater. 8, 843–849 (2009)
Kaiser, H. J. et al. Order of lipid phases in model and plasma membranes. Proc. Natl Acad. Sci. USA 106, 16645–16650 (2009)
Laage, R., Rohde, J., Brosig, B. & Langosch, D. A conserved membrane-spanning amino acid motif drives homomeric and supports heteromeric assembly of presynaptic SNARE proteins. J. Biol. Chem. 275, 17481–17487 (2000)
Sieber, J. J., Willig, K. I., Heintzmann, R., Hell, S. W. & Lang, T. The SNARE motif is essential for the formation of syntaxin clusters in the plasma membrane. Biophys. J. 90, 2843–2851 (2006)
Marrink, S. J., Risselada, H. J., Yefimov, S., Tieleman, D. P. & de Vries, A. H. The MARTINI forcefield: coarse grained model for biomolecular simulations. J. Phys. Chem. B 111, 7812–7824 (2007)
Yesylevskyy, S., Schafer, L. V., Sengupta, D. & Marrink, S. J. Polarizable water model for the coarse-grained Martini force field. PLoS Comp. Biol. 6, e1000810 (2010)
Fujiwara, T., Ritchie, K., Murakoshi, H., Jacobson, K. & Kusumi, A. Phospholipids undergo hop diffusion in compartmentalized cell membrane. J. Cell Biol. 157, 1071–1082 (2002)
van den Bogaart, G. & Jahn, R. Counting the SNAREs needed for membrane fusion. J. Mol. Cell Biol. 3, 204–205 (2011)
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997)
Acknowledgements
We thank M. Holt, G. Bunt, F. S. Wouters and C. Eggeling for advice, and V. Haucke and S. Joo for the red-fluorescent-protein-tagged synaptojanin-1 construct. G.v.d.B. is financed by the Human Frontier Science Program. This work was supported by the US National Institutes of Health (P01 GM072694, to R.J.) and the Deutsche Forschungsgemeinschaft (SFB803, to K.M., H.J.R., U.D., H.G. and R.J.).
Author information
Authors and Affiliations
Contributions
G.v.d.B. and R.J. designed the experiments and wrote the paper. K.M., B.E.H. and U.D. synthesized the peptides. H.J.R. and H.G. performed the simulations. K.I.W. and S.W.H performed the STED microscopy. H.A. and M.D. contributed to the protein purification, immunofluorescence and microscopy. G.v.d.B. performed all other experiments. All authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Methods, Supplementary Figures 1-15 with legends, legend for Supplementary Movie 1 and additional references. (PDF 1455 kb)
Supplementary Movie 1
The movie shows a simulation of the dynamic and amorphous PIP2-syntaxin-1A microdomains - see Supplementary information file for full legend. (MPG 23880 kb)
Rights and permissions
About this article
Cite this article
van den Bogaart, G., Meyenberg, K., Risselada, H. et al. Membrane protein sequestering by ionic protein–lipid interactions. Nature 479, 552–555 (2011). https://doi.org/10.1038/nature10545
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10545
This article is cited by
-
Mechanisms of SNARE proteins in membrane fusion
Nature Reviews Molecular Cell Biology (2024)
-
Long-term depression in neurons involves temporal and ultra-structural dynamics of phosphatidylinositol-4,5-bisphosphate relying on PIP5K, PTEN and PLC
Communications Biology (2023)
-
Transmission-type high-resolution laser scanning microscope
Optical Review (2023)
-
Hydroxychloroquine blocks SARS-CoV-2 entry into the endocytic pathway in mammalian cell culture
Communications Biology (2022)
-
Spatiotemporal dynamics of membrane surface charge regulates cell polarity and migration
Nature Cell Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.