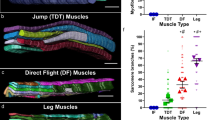
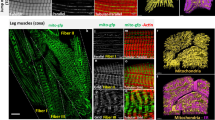
Abstract
Flying insects oscillate their wings at high frequencies of up to 1,000 Hz1,2 and produce large mechanical forces of 80 W per kilogram of muscle3. They utilize a pair of perpendicularly oriented indirect flight muscles that contain fibrillar, stretch-activated myofibres. In contrast, all other, more slowly contracting, insect body muscles have a tubular muscle morphology4. Here we identify the transcription factor Spalt major (Salm) as a master regulator of fibrillar flight muscle fate in Drosophila. salm is necessary and sufficient to induce fibrillar muscle fate. salm switches the entire transcriptional program from tubular to fibrillar fate by regulating the expression and splicing of key sarcomeric components specific to each muscle type. Spalt function is conserved in insects evolutionarily separated by 280 million years. We propose that Spalt proteins switch myofibres from tubular to fibrillar fate during development, a function potentially conserved in the vertebrate heart—a stretch-activated muscle sharing features with insect flight muscle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dudley, R. in The Biomechanics of Insect Flight (ed. Dudley, R. ) 75–158 (Princeton Univ. Press, 2000)
Dickinson, M. Insect flight. Curr. Biol. 16, R309–R314 (2006)
Lehmann, F. O. & Dickinson, M. H. The changes in power requirements and muscle efficiency during elevated force production in the fruit fly Drosophila melanogaster . J. Exp. Biol. 200, 1133–1143 (1997)
Bernstein, S. I., O’Donnell, P. T. & Cripps, R. M. Molecular genetic analysis of muscle development, structure, and function in Drosophila . Int. Rev. Cytol. 143, 63–152 (1993)
Dudley, R. in The Biomechanics of Insect Flight (ed. Dudley, R. ) 36–74 (Princeton Univ. Press, 2000)
Schnorrer, F. et al. Systematic genetic analysis of muscle morphogenesis and function in Drosophila . Nature 464, 287–291 (2010)
de Celis, J. F. & Barrio, R. Regulation and function of Spalt proteins during animal development. Int. J. Dev. Biol. 53, 1385–1398 (2009)
Dutta, D., Anant, S., Ruiz-Gomez, M., Bate, M. & VijayRaghavan, K. Founder myoblasts and fibre number during adult myogenesis in Drosophila . Development 131, 3761–3772 (2004)
Anant, S., Roy, S. & VijayRaghavan, K. Twist and Notch negatively regulate adult muscle differentiation in Drosophila . Development 125, 1361–1369 (1998)
Franch-Marro, X. & Casanova, J. spalt-induced specification of distinct dorsal and ventral domains is required for Drosophila tracheal patterning. Dev. Biol. 250, 374–382 (2002)
Mollereau, B. et al. Two-step process for photoreceptor formation in Drosophila . Nature 412, 911–913 (2001)
Reedy, M. C., Bullard, B. & Vigoreaux, J. O. Flightin is essential for thick filament assembly and sarcomere stability in Drosophila flight muscles. J. Cell Biol. 151, 1483–1500 (2000)
Qiu, F. et al. Myofilin, a protein in the thick filaments of insect muscle. J. Cell Sci. 118, 1527–1536 (2005)
Stronach, B. E., Siegrist, S. E. & Beckerle, M. C. Two muscle-specific LIM proteins in Drosophila . J. Cell Biol. 134, 1179–1195 (1996)
Bernard, F. et al. Control of apterous by vestigial drives indirect flight muscle development in Drosophila . Dev. Biol. 260, 391–403 (2003)
Halder, G. et al. The Vestigial and Scalloped proteins act together to directly regulate wing-specific gene expression in Drosophila . Genes Dev. 12, 3900–3909 (1998)
Maqbool, T. et al. Shaping leg muscles in Drosophila: role of ladybird, a conserved regulator of appendicular myogenesis. PLoS ONE 1, e122 (2006)
Agianian, B. et al. A troponin switch that regulates muscle contraction by stretch instead of calcium. EMBO J. 23, 772–779 (2004)
Arredondo, J. J. et al. Control of Drosophila paramyosin/miniparamyosin gene expression. Differential regulatory mechanisms for muscle-specific transcription. J. Biol. Chem. 276, 8278–8287 (2001)
Patel, S. R. & Saide, J. D. Stretchin-klp, a novel Drosophila indirect flight muscle protein, has both myosin dependent and independent isoforms. J. Muscle Res. Cell Motil. 26, 213–224 (2005)
Ayme-Southgate, A., Lasko, P., French, C. & Pardue, M. L. Characterization of the gene for mp20: a Drosophila muscle protein that is not found in asynchronous oscillatory flight muscle. J. Cell Biol. 108, 521–531 (1989)
Nongthomba, U., Pasalodos-Sanchez, S., Clark, S., Clayton, J. D. & Sparrow, J. C. Expression and function of the Drosophila ACT88F actin isoform is not restricted to the indirect flight muscles. J. Muscle Res. Cell Motil. 22, 111–119 (2001)
Savard, J. et al. Phylogenomic analysis reveals bees and wasps (Hymenoptera) at the base of the radiation of holometabolous insects. Genome Res. 16, 1334–1338 (2006)
Tomoyasu, Y. & Denell, R. E. Larval RNAi in Tribolium (Coleoptera) for analyzing adult development. Dev. Genes Evol. 214, 575–578 (2004)
Parrish, M. et al. Loss of the Sall3 gene leads to palate deficiency, abnormalities in cranial nerves, and perinatal lethality. Mol. Cell. Biol. 24, 7102–7112 (2004)
Nishinakamura, R. et al. Murine homolog of SALL1 is essential for ureteric bud invasion in kidney development. Development 128, 3105–3115 (2001)
Manisastry, S. M., Zaal, K. J. & Horowits, R. Myofibril assembly visualized by imaging N-RAP, α-actinin, and actin in living cardiomyocytes. Exp. Cell Res. 315, 2126–2139 (2009)
Shiels, H. A. & White, E. The Frank–Starling mechanism in vertebrate cardiac myocytes. J. Exp. Biol. 211, 2005–2013 (2008)
Surka, W. S., Kohlhase, J., Neunert, C. E., Schneider, D. S. & Proud, V. K. Unique family with Townes–Brocks syndrome, SALL1 mutation, and cardiac defects. Am. J. Med. Genet. 102, 250–257 (2001)
Ranganayakulu, G., Schulz, R. A. & Olson, E. N. Wingless signaling induces nautilus expression in the ventral mesoderm of the Drosophila embryo. Dev. Biol. 176, 143–148 (1996)
Grieder, N. C., Morata, G., Affolter, M. & Gehring, W. J. Spalt major controls the development of the notum and of wing hinge primordia of the Drosophila melanogaster wing imaginal disc. Dev. Biol. 329, 315–326 (2009)
Rebeiz, M., Reeves, N. L. & Posakony, J. W. SCORE: a computational approach to the identification of cis-regulatory modules and target genes in whole-genome sequence data. Site clustering over random expectation. Proc. Natl Acad. Sci. USA 99, 9888–9893 (2002)
Chen, E. H. & Olson, E. N. Antisocial, an intracellular adaptor protein, is required for myoblast fusion in Drosophila . Dev. Cell 1, 705–715 (2001)
Newsome, T. P., Asling, B. & Dickson, B. J. Analysis of Drosophila photoreceptor axon guidance in eye-specific mosaics. Development 127, 851–860 (2000)
Kuhnlein, R. P. et al. spalt encodes an evolutionarily conserved zinc finger protein of novel structure which provides homeotic gene function in the head and tail region of the Drosophila embryo. EMBO J. 13, 168–179 (1994)
Fernandes, J. J., Celniker, S. E. & VijayRaghavan, K. Development of the indirect flight muscle attachment sites in Drosophila: role of the PS integrins and the stripe gene. Dev. Biol. 176, 166–184 (1996)
Klein, T. in Drosophila: Methods and Protocols (ed. Dahmann, C.) 253–264 (Humana Press, 2008).
Acknowledgements
We thank M. Affolter, D. Bäumer, M. Beckerle, B. Bullard, E. Chen, K. Clark, C. Desplan, K. Jagla, A. Lalouette, J. Posakony, D. Reiff, J. Saide, S. Sprecher, R. Schuh, G. Tanentzapf, J. Vigoreaux, K. VijayRaghavan, the Bloomington and the VDRC stock centres for fly stocks, antibodies and insect species. We are grateful to B. Dickson, I. Hein, M. Klingler and M. Sixt for discussions, and to R. Fässler for support and discussions. We thank A. Kaya-Copur, H. Knaut, M. Spletter and N. Vogt for critical comments on the manuscript. This work was supported by the Max-Planck-Society, a Career Development Award by the Human Frontier Science Programme to F.S., a Doc-fForte predoctoral fellowship from the Austrian Academy of Sciences to C.S., and DFG grants to M.F.
Author information
Authors and Affiliations
Contributions
C.S. performed most of the experiments, analysed the data and created most of the figures. F.S. acquired the time-lapse movies and performed western blots. J.D. and M.F conducted the Tribolium RNAi experiments, M.R. performed the microarray analysis, and N.J. and H.-U.D. were involved in the initial characterisation of the salm mutant phenotype. F.S. conceived and supervised the project and wrote the manuscript with input from C.S. and M.F.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-8 with legends, Supplementary Table 1, legends for Supplementary Table 2 and Supplementary Movies 1-3 and Supplementary Data for the Tc’sal fragment for RNAi injections 3320bp. (PDF 9022 kb)
Supplementary Table 2
The table shows top 500 salm targets and IFM specific genes of microarray analysis (see Supplementary Information for full legend). (XLS 493 kb)
Supplementary Movie 1
The movie shows early IFM development in a wild-type pupa (see Supplementary Information for full legend). (MOV 4343 kb)
Supplementary Movie 2
The movie shows early IFM development in a UAS-salm-IR pupa (see Supplementary Information for full legend). (MOV 4592 kb)
Supplementary Movie 3
The movie shows salm expression in developing IFMs (see Supplementary Information for full legend). (MOV 7703 kb)
Rights and permissions
About this article
Cite this article
Schönbauer, C., Distler, J., Jährling, N. et al. Spalt mediates an evolutionarily conserved switch to fibrillar muscle fate in insects. Nature 479, 406–409 (2011). https://doi.org/10.1038/nature10559
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10559
This article is cited by
-
M1BP is an essential transcriptional activator of oxidative metabolism during Drosophila development
Nature Communications (2023)
-
The red flour beetle T. castaneum: elaborate genetic toolkit and unbiased large scale RNAi screening to study insect biology and evolution
EvoDevo (2022)
-
Regulation of the evolutionarily conserved muscle myofibrillar matrix by cell type dependent and independent mechanisms
Nature Communications (2022)
-
Identification of evolutionarily conserved regulators of muscle mitochondrial network organization
Nature Communications (2022)
-
Mitochondrial network configuration influences sarcomere and myosin filament structure in striated muscles
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.