Abstract
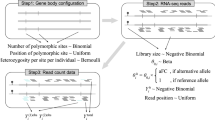
The mapping of expression quantitative trait loci (eQTLs) has emerged as an important tool for linking genetic variation to changes in gene regulation1,2,3,4,5. However, it remains difficult to identify the causal variants underlying eQTLs, and little is known about the regulatory mechanisms by which they act. Here we show that genetic variants that modify chromatin accessibility and transcription factor binding are a major mechanism through which genetic variation leads to gene expression differences among humans. We used DNase I sequencing to measure chromatin accessibility in 70 Yoruba lymphoblastoid cell lines, for which genome-wide genotypes and estimates of gene expression levels are also available6,7,8. We obtained a total of 2.7 billion uniquely mapped DNase I-sequencing (DNase-seq) reads, which allowed us to produce genome-wide maps of chromatin accessibility for each individual. We identified 8,902 locations at which the DNase-seq read depth correlated significantly with genotype at a nearby single nucleotide polymorphism or insertion/deletion (false discovery rate = 10%). We call such variants ‘DNase I sensitivity quantitative trait loci’ (dsQTLs). We found that dsQTLs are strongly enriched within inferred transcription factor binding sites and are frequently associated with allele-specific changes in transcription factor binding. A substantial fraction (16%) of dsQTLs are also associated with variation in the expression levels of nearby genes (that is, these loci are also classified as eQTLs). Conversely, we estimate that as many as 55% of eQTL single nucleotide polymorphisms are also dsQTLs. Our observations indicate that dsQTLs are highly abundant in the human genome and are likely to be important contributors to phenotypic variation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Data deposits
All raw data and tables of all dsQTLs are deposited in GEO under accession number GSE31388 and at http://eqtl.uchicago.edu.
References
Brem, R. B., Yvert, G., Clinton, R. & Kruglyak, L. Genetic dissection of transcriptional regulation in budding yeast. Science 296, 752–755 (2002)
Cheung, V. G. et al. Mapping determinants of human gene expression by regional and genome-wide association. Nature 437, 1365–1369 (2005)
Nicolae, D. L. et al. Trait-associated SNPs are more likely to be eQTLs: annotation to enhance discovery from GWAS. PLoS Genet. 6, e1000888 (2010)
Nica, A. C. et al. Candidate causal regulatory effects by integration of expression QTLs with complex trait genetic associations. PLoS Genet. 6, e1000895 (2010)
Lango Allen, H. et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 467, 832–838 (2010)
Frazer, K. A. et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 449, 851–861 (2007)
Durbin, R. M. et al. A map of human genome variation from population-scale sequencing. Nature 467, 1061–1073 (2010)
Pickrell, J. K. et al. Understanding mechanisms underlying human gene expression variation with RNA sequencing. Nature 464, 768–772 (2010)
Gaulton, K. J. et al. A map of open chromatin in human pancreatic islets. Nature Genet. 42, 255–259 (2010)
Kasowski, M. et al. Variation in transcription factor binding among humans. Science 328, 232–235 (2010)
McDaniell, R. et al. Heritable individual-specific and allele-specific chromatin signatures in humans. Science 328, 235–239 (2010)
Zheng, W., Zhao, H., Mancera, E., Steinmetz, L. M. & Snyder, M. Genetic analysis of variation in transcription factor binding in yeast. Nature 464, 1187–1191 (2010)
Galas, D. & Schmitz, A. DNAse footprinting: a simple method for the detection of protein-DNA binding specificity. Nucleic Acids Res. 5, 3157–3170 (1978)
Boyle, A. P. et al. High-resolution mapping and characterization of open chromatin across the genome. Cell 132, 311–322 (2008)
Hesselberth, J. R. et al. Global mapping of protein-DNA interactions in vivo by digital genomic footprinting. Nature Methods 6, 283–289 (2009)
Boyle, A. P. et al. High-resolution genome-wide in vivo footprinting of diverse transcription factors in human cells. Genome Res. 21, 456–464 (2011)
Pique-Regi, R. et al. Accurate inference of transcription factor binding from DNA sequence and chromatin accessibility data. Genome Res. 21, 447–455 (2011)
Ernst, J. et al. Mapping and analysis of chromatin state dynamics in nine human cell types. Nature 473, 43–49 (2011)
Veyrieras, J. B. et al. High-resolution mapping of expression-QTLs yields insight into human gene regulation. PLoS Genet. 4, e1000214 (2008)
Mirny, L. A. Nucleosome-mediated cooperativity between transcription factors. Proc. Natl Acad. Sci. USA 107, 22534–22539 (2010)
Wasson, T. & Hartemink, A. J. An ensemble model of competitive multi-factor binding of the genome. Genome Res. 19, 2101–2112 (2009)
Raveh-Sadka, T., Levo, M. & Segal, E. Incorporating nucleosomes into thermodynamic models of transcription regulation. Genome Res. 19, 1480–1496 (2009)
Myers, R. M. et al. A user’s guide to the encyclopedia of DNA elements (ENCODE). PLoS Biol. 9, e1001046 (2011)
Storey, J. D., Taylor, J. E. & Siegmund, D. Strong control, conservative point estimation, and simultaneous conservative consistency of false discovery rates: a unified approach. J. R. Stat. Soc., B 66, 187–205 (2004)
Dixon, A. L. et al. A genome-wide association study of global gene expression. Nature Genet. 39, 1202–1207 (2007)
Stranger, B. E. et al. Population genomics of human gene expression. Nature Genet. 39, 1217–1224 (2007)
Bell, J. T. et al. DNA methylation patterns associate with genetic and gene expression variation in HapMap cell lines. Genome Biol. 12, R10 (2011)
Visel, A. et al. ChIP-seq accurately predicts tissue-specific activity of enhancers. Nature 457, 854–858 (2009)
Song, L. & Crawford, G. E. DNase-seq: a high-resolution technique for mapping active gene regulatory elements across the genome from mammalian cells. Cold Spring Harb. Protocols 10.1101/pdb.prot5384 (2010)
Guan, Y. & Stephens, M. Practical issues in imputation-based association mapping. PLoS Genet. 4, e1000279 (2008)
Acknowledgements
We thank members of the Pritchard, Przeworski, Stephens and Gilad laboratories for many helpful comments or discussions, and the ENCODE Project for publicly available ChIP-seq data. This work was supported by grants from the National Institutes of Health to Y.G. (HG006123) and J.K.P. (MH084703 and MH090951), by the Howard Hughes Medical Institute, by the Chicago Fellows Program (to R.P.R.), by the American Heart Association (to A.A.P.), and by the NIH Genetics and Regulation Training grant (A.A.P. and J.F.D.).
Author information
Authors and Affiliations
Contributions
A.A.P. led the data collection with assistance from S.D.L., K.M. and N.L. The data analysis was performed jointly by J.F.D. and R.P.R., with contributions from A.A.P., J.B.V., D.J.G. and J.K.Pi. G.E.C. and M.S. provided technical assistance and discussion of methods and results. The manuscript was written by J.F.D., A.A.P., R.P.R., Y.G. and J.K.Pr. The project was jointly supervised by Y.G. and J.K.Pr.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, Supplementary Figures 1-28 with legends, Supplementary Tables 1-9 and additional references. (PDF 3101 kb)
Rights and permissions
About this article
Cite this article
Degner, J., Pai, A., Pique-Regi, R. et al. DNase I sensitivity QTLs are a major determinant of human expression variation. Nature 482, 390–394 (2012). https://doi.org/10.1038/nature10808
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10808
This article is cited by
-
Inferring cell-type-specific causal gene regulatory networks during human neurogenesis
Genome Biology (2023)
-
Molecular quantitative trait loci
Nature Reviews Methods Primers (2023)
-
An autoimmune pleiotropic SNP modulates IRF5 alternative promoter usage through ZBTB3-mediated chromatin looping
Nature Communications (2023)
-
Human forebrain organoid-based multi-omics analyses of PCCB as a schizophrenia associated gene linked to GABAergic pathways
Nature Communications (2023)
-
Immune-response 3′UTR alternative polyadenylation quantitative trait loci contribute to variation in human complex traits and diseases
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.