Abstract
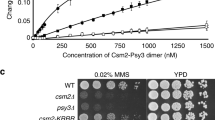
DNA double-strand break repair is critical for cell viability and involves highly coordinated pathways to restore DNA integrity at the lesion. An early event during homology-dependent repair is resection of the break to generate progressively longer 3′ single-strand tails that are used to identify suitable templates for repair1. Sister chromatids provide near-perfect sequence homology and are therefore the preferred templates during homologous recombination2,3. To provide a bias for the use of sisters as donors, cohesin—the complex that tethers sister chromatids together4—is recruited to the break5,6 to enforce physical proximity. Here we show that DNA breaks promote dissociation of cohesin loaded during the previous S phase in budding yeast, and that damage-induced dissociation of cohesin requires separase, the protease that dissolves cohesion in anaphase7. Moreover, a separase-resistant allele of the gene coding for the α-kleisin subunit of cohesin, Mcd1 (also known as Scc1), reduces double-strand break resection and compromises the efficiency of repair even when loaded during DNA damage. We conclude that post-replicative DNA repair involves cohesin dissociation by separase to promote accessibility to repair factors during the coordinated cellular response to restore DNA integrity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pâques, F. & Haber, J. E. Multiple pathways of recombination induced by double-strand breaks in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 63, 349–404 (1999)
Kadyk, L. C. & Hartwell, L. H. Sister chromatids are preferred over homologs as substrates for recombinational repair in Saccharomyces cerevisiae. Genetics 132, 387–402 (1992)
Johnson, R. D. & Jasin, M. Sister chromatid gene conversion is a prominent double-strand break repair pathway in mammalian cells. EMBO J. 19, 3398–3407 (2000)
Nasmyth, K. Cohesin: a catenase with separate entry and exit gates? Nature Cell Biol. 13, 1170–1177 (2011)
Unal, E. et al. DNA damage response pathway uses histone modification to assemble a double-strand break-specific cohesin domain. Mol. Cell 16, 991–1002 (2004)
Strom, L., Lindroos, H. B., Shirahige, K. & Sjogren, C. Postreplicative recruitment of cohesin to double-strand breaks is required for DNA repair. Mol. Cell 16, 1003–1015 (2004)
Uhlmann, F., Lottspeich, F. & Nasmyth, K. Sister-chromatid separation at anaphase onset is promoted by cleavage of the cohesin subunit Scc1. Nature 400, 37–42 (1999)
Strom, L. et al. Postreplicative formation of cohesion is required for repair and induced by a single DNA break. Science 317, 242–245 (2007)
Unal, E., Heidinger-Pauli, J. M. & Koshland, D. DNA double-strand breaks trigger genome-wide sister-chromatid cohesion through Eco1 (Ctf7). Science 317, 245–248 (2007)
Heidinger-Pauli, J. M., Mert, O., Davenport, C., Guacci, V. & Koshland, D. Systematic reduction of cohesin differentially affects chromosome segregation, condensation, and DNA repair. Curr. Biol. 20, 957–963 (2010)
Heidinger-Pauli, J. M., Unal, E. & Koshland, D. Distinct targets of the Eco1 acetyltransferase modulate cohesion in S phase and in response to DNA damage. Mol. Cell 34, 311–321 (2009)
Nagao, K., Adachi, Y. & Yanagida, M. Separase-mediated cleavage of cohesin at interphase is required for DNA repair. Nature 430, 1044–1048 (2004)
Baskerville, C., Segal, M. & Reed, S. I. The protease activity of yeast separase (esp1) is required for anaphase spindle elongation independently of its role in cleavage of cohesin. Genetics 178, 2361–2372 (2008)
Heidinger-Pauli, J. M., Unal, E., Guacci, V. & Koshland, D. The kleisin subunit of cohesin dictates damage-induced cohesion. Mol. Cell 31, 47–56 (2008)
Zhu, Z., Chung, W. H., Shim, E. Y., Lee, S. E. & Ira, G. Sgs1 helicase and two nucleases Dna2 and Exo1 resect DNA double-strand break ends. Cell 134, 981–994 (2008)
Mimitou, E. P. & Symington, L. S. Sae2, Exo1 and Sgs1 collaborate in DNA double-strand break processing. Nature 455, 770–774 (2008)
Shim, E. Y. et al. Saccharomyces cerevisiae Mre11/Rad50/Xrs2 and Ku proteins regulate association of Exo1 and Dna2 with DNA breaks. EMBO J. 29, 3370–3380 (2010)
Vaze, M. B. et al. Recovery from checkpoint-mediated arrest after repair of a double-strand break requires Srs2 helicase. Mol. Cell 10, 373–385 (2002)
Jain, S. et al. A recombination execution checkpoint regulates the choice of homologous recombination pathway during DNA double-strand break repair. Genes Dev. 23, 291–303 (2009)
Acknowledgements
We would like to thank D. Koshland, E. Unal, J. Haber, G. Ira and K. Nasmyth for providing valuable strains. We thank members of the Aragon laboratory, P. Ullal, S. Farmer, K. Pegram, J. Torres-Rosell and M. Merkenschlager for discussions and reading of the manuscript. This work was funded by the Intramural Research Program of the Medical Research Council UK.
Author information
Authors and Affiliations
Contributions
A.M. made the original observation that transcriptional repression of MCD1 in the presence of DNA damage causes defects in sister chromatid cohesion. A.M. and A.C.-B. performed all ChIP analysis. A.M., A.C.-B., N.S. and V.C.-P. performed microscopic analysis of cohesion. A.C.-B. and M.E. performed physical assays to measure resection and repair. A.M. and A.C.-B. performed biochemical assays to measure Mcd1 stability. A.J. provided technical support. L.A. wrote the paper, and all authors analysed the data, discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1–2 and Supplementary Figures 1-10. (PDF 7171 kb)
Rights and permissions
About this article
Cite this article
McAleenan, A., Clemente-Blanco, A., Cordon-Preciado, V. et al. Post-replicative repair involves separase-dependent removal of the kleisin subunit of cohesin. Nature 493, 250–254 (2013). https://doi.org/10.1038/nature11630
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11630
This article is cited by
-
Separase activity distribution can be a marker of major molecular response and proliferation of CD34+ cells in TKI-treated chronic myeloid leukemia patients
Annals of Hematology (2020)
-
AIMP3 Deletion Induces Acute Radiation Syndrome-like Phenotype in Mice
Scientific Reports (2018)
-
Cohesin dynamic association to chromatin and interfacing with replication forks in genome integrity maintenance
Current Genetics (2018)
-
Overexpression and constitutive nuclear localization of cohesin protease Separase protein correlates with high incidence of relapse and reduced overall survival in glioblastoma multiforme
Journal of Neuro-Oncology (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.