Abstract
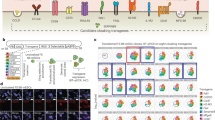
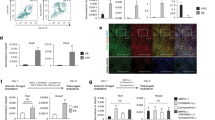
The advantages of using induced pluripotent stem cells (iPSCs) instead of embryonic stem (ES) cells in regenerative medicine centre around circumventing concerns about the ethics of using ES cells and the likelihood of immune rejection of ES-cell-derived tissues1,2. However, partial reprogramming and genetic instabilities in iPSCs3,4,5,6 could elicit immune responses in transplant recipients even when iPSC-derived differentiated cells are transplanted. iPSCs are first differentiated into specific types of cells in vitro for subsequent transplantation. Although model transplantation experiments have been conducted using various iPSC-derived differentiated tissues7,8,9,10 and immune rejections have not been observed, careful investigation of the immunogenicity of iPSC-derived tissue is becoming increasingly critical, especially as this has not been the focus of most studies done so far. A recent study reported immunogenicity of iPSC- but not ES-cell-derived teratomas11 and implicated several causative genes. Nevertheless, some controversy has arisen regarding these findings12. Here we examine the immunogenicity of differentiated skin and bone marrow tissues derived from mouse iPSCs. To ensure optimal comparison of iPSCs and ES cells, we established ten integration-free iPSC and seven ES-cell lines using an inbred mouse strain, C57BL/6. We observed no differences in the rate of success of transplantation when skin and bone marrow cells derived from iPSCs were compared with ES-cell-derived tissues. Moreover, we observed limited or no immune responses, including T-cell infiltration, for tissues derived from either iPSCs or ES cells, and no increase in the expression of the immunogenicity-causing Zg16 and Hormad1 genes in regressing skin and teratoma tissues. Our findings suggest limited immunogenicity of transplanted cells differentiated from iPSCs and ES cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006)
Kiskinis, E. & Eggan, K. Progress toward the clinical application of patient-specific pluripotent stem cells. J. Clin. Invest. 120, 51–59 (2010)
Polo, J. M. et al. Cell type of origin influences the molecular and functional properties of mouse induced pluripotent stem cells. Nature Biotechnol. 28, 848–855 (2010)
Gore, A. et al. Somatic coding mutations in human induced pluripotent stem cells. Nature 471, 63–67 (2011)
Puri, M. C. & Nagy, A. Concise review: embryonic stem cells versus induced pluripotent stem cells: the game is on. Stem Cells 30, 10–14 (2012)
Martins-Taylor, K. & Xu, R. H. Concise review: genomic stability of human induced pluripotent stem cells. Stem Cells 30, 22–27 (2012)
Hanna, J. et al. Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science 318, 1920–1923 (2007)
Wernig, M. et al. Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proc. Natl Acad. Sci. USA 105, 5856–5861 (2008)
Tsuji, O. et al. Therapeutic potential of appropriately evaluated safe-induced pluripotent stem cells for spinal cord injury. Proc. Natl Acad. Sci. USA 107, 12704–12709 (2010)
Tucker, B. A. et al. Transplantation of adult mouse iPS cell-derived photoreceptor precursors restores retinal structure and function in degenerative mice. PLoS ONE 6, e18992 (2011)
Zhao, T., Zhang, Z. N., Rong, Z. & Xu, Y. Immunogenicity of induced pluripotent stem cells. Nature 474, 212–215 (2011)
Okita, K., Nagata, N. & Yamanaka, S. Immunogenicity of induced pluripotent stem cells. Circ. Res. 109, 720–721 (2011)
Chen, Y. T. et al. Identification of CT46/HORMAD1, an immunogenic cancer/testis antigen encoding a putative meiosis-related protein. Cancer Immun. 5, 9 (2005)
Okita, K., Nakagawa, M., Hyenjong, H., Ichisaka, T. & Yamanaka, S. Generation of mouse induced pluripotent stem cells without viral vectors. Science 322, 949–953 (2008)
Jincho, Y. et al. Generation of genome integration-free induced pluripotent stem cells from fibroblasts of C57BL/6 mice without c-Myc transduction. J. Biol. Chem. 285, 26384–26389 (2010)
Bosma, G. C., Custer, R. P. & Bosma, M. J. A severe combined immunodeficiency mutation in the mouse. Nature 301, 527–530 (1983)
Dressel, R. et al. The tumorigenicity of mouse embryonic stem cells and in vitro differentiated neuronal cells is controlled by the recipients’ immune response. PLoS ONE 3, e2622 (2008)
Okabe, M., Ikawa, M., Kominami, K., Nakanishi, T. & Nishimune, Y. ‘Green mice’ as a source of ubiquitous green cells. FEBS Lett. 407, 313–319 (1997)
Araki, R. et al. Crucial role of c-Myc in the generation of induced pluripotent stem cells. Stem Cells 29, 1362–1370 (2011)
Billingham, R. E. & Medawar, P. B. The technique of free skin grafting in mammals. J. Exp. Biol. 28, 385–391 (1951)
Bubnic, S. J., Nagy, A. & Keating, A. Donor hematopoietic cells from transgenic mice that express GFP are immunogenic in immunocompetent recipients. Hematology 10, 289–295 (2005)
Boland, M. J. et al. Adult mice generated from induced pluripotent stem cells. Nature 461, 91–94 (2009)
Kang, L., Wang, J., Zhang, Y., Kou, Z. & Gao, S. iPS cells can support full-term development of tetraploid blastocyst-complemented embryos. Cell Stem Cell 5, 135–138 (2009)
Zhao, X. Y. et al. iPS cells produce viable mice through tetraploid complementation. Nature 461, 86–90 (2009)
Miura, K. et al. Variation in the safety of induced pluripotent stem cell lines. Nature Biotechnol. 27, 743–745 (2009)
Marks, H. et al. The transcriptional and epigenomic foundations of ground state pluripotency. Cell 149, 590–604 (2012)
Ying, Q. L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008)
Carey, B. W. et al. Reprogramming factor stoichiometry influences the epigenetic state and biological properties of induced pluripotent stem cells. Cell Stem Cell 9, 588–598 (2011)
Stadtfeld, M. et al. Ascorbic acid prevents loss of Dlk1-Dio3 imprinting and facilitates generation of all–iPS cell mice from terminally differentiated B cells. Nature Genet. 44, 398–405 (2012)
Nagy, A., Rossant, J., Nagy, R., Abramow-Newerly, W. & Roder, J. C. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc. Natl Acad. Sci. USA 90, 8424–8428 (1993)
Patten, P. E. et al. CD38 expression in chronic lymphocytic leukemia is regulated by the tumor microenvironment. Blood 111, 5173–5181 (2008)
Le Moine, A. et al. Critical roles for IL-4, IL-5, and eosinophils in chronic skin allograft rejection. J. Clin. Invest. 103, 1659–1667 (1999)
Surquin, M. et al. Skin graft rejection elicited by β2-microglobulin as a minor transplantation antigen involves multiple effector pathways: role of Fas-Fas ligand interactions and Th2-dependent graft eosinophil infiltrates. J. Immunol. 169, 500–506 (2002)
Baruah, P. et al. Mice lacking C1q or C3 show accelerated rejection of minor H disparate skin grafts and resistance to induction of tolerance. Eur. J. Immunol. 40, 1758–1767 (2010)
Huang, T. T. et al. Genetic modifiers of the phenotype of mice deficient in mitochondrial superoxide dismutase. Hum. Mol. Genet. 15, 1187–1194 (2006)
Morizane, A., Doi, D., Kikuchi, T., Nishimura, K. & Takahashi, J. Small-molecule inhibitors of bone morphogenic protein and activin/nodal signals promote highly efficient neural induction from human pluripotent stem cells. J. Neurosci. Res. 89, 117–126 (2011)
Takahashi, T. et al. Ascorbic acid enhances differentiation of embryonic stem cells into cardiac myocytes. Circulation 107, 1912–1916 (2003)
Guo, X. M. et al. Creation of engineered cardiac tissue in vitro from mouse embryonic stem cells. Circulation 113, 2229–2237 (2006)
Acknowledgements
We thank S. Yamanaka for providing the 2A plasmid vectors; K. Ito, N. Nakajima and K. Kawabata for technical advice on the transplantation experiments; H. Yoshida, K. Nishikawa, A. Ishibashi, R. Watanabe, M. Nakamura and T. Maeda for technical assistance. This work was partially funded by a research grant from Precursory Research for Embryonic Science and Technology (PRESTO), Japan Science and Technology Agency.
Author information
Authors and Affiliations
Contributions
R.A. and M.A. designed the experiments, analysed the data and wrote the manuscript. R.A. and Y.H. established the iPSC and ES-cell lines. M.U., S.A., M.N. and Y.H. performed teratoma formation assays and transplantation assays. M. Sunayama and M. Sugiura performed molecular biological analyses. M.U., H.I., A.S. and A.N. performed immunohistochemical analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Table 1 and Supplementary Figures 1-13. (PDF 1770 kb)
Rights and permissions
About this article
Cite this article
Araki, R., Uda, M., Hoki, Y. et al. Negligible immunogenicity of terminally differentiated cells derived from induced pluripotent or embryonic stem cells. Nature 494, 100–104 (2013). https://doi.org/10.1038/nature11807
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11807
This article is cited by
-
iPSC-derived cells lack immune tolerance to autologous NK-cells due to imbalance in ligands for activating and inhibitory NK-cell receptors
Stem Cell Research & Therapy (2023)
-
Applications of synthetic biology in medical and pharmaceutical fields
Signal Transduction and Targeted Therapy (2023)
-
Macrophages derived from pluripotent stem cells: prospective applications and research gaps
Cell & Bioscience (2022)
-
Human-induced pluripotent stem cells-derived retinal pigmented epithelium, a new horizon for cells-based therapies for age-related macular degeneration
Stem Cell Research & Therapy (2022)
-
Dental pulp stem cells as a therapy for congenital entero-neuropathy
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.