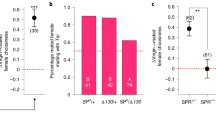
Abstract
Animals display a repertoire of different social behaviours. Appropriate behavioural responses depend on sensory input received during social interactions. In mice, social behaviour is driven by pheromones, chemical signals that encode information related to age, sex and physiological state1. However, although mice show different social behaviours towards adults, juveniles and neonates, sensory cues that enable specific recognition of juvenile mice are unknown. Here we describe a juvenile pheromone produced by young mice before puberty, termed exocrine-gland secreting peptide 22 (ESP22). ESP22 is secreted from the lacrimal gland and released into tears of 2- to 3-week-old mice. Upon detection, ESP22 activates high-affinity sensory neurons in the vomeronasal organ, and downstream limbic neurons in the medial amygdala. Recombinant ESP22, painted on mice, exerts a powerful inhibitory effect on adult male mating behaviour, which is abolished in knockout mice lacking TRPC2, a key signalling component of the vomeronasal organ2,3. Furthermore, knockout of TRPC2 or loss of ESP22 production results in increased sexual behaviour of adult males towards juveniles, and sexual responses towards ESP22-deficient juveniles are suppressed by ESP22 painting. Thus, we describe a pheromone of sexually immature mice that controls an innate social behaviour, a response pathway through the accessory olfactory system and a new role for vomeronasal organ signalling in inhibiting sexual behaviour towards young. These findings provide a molecular framework for understanding how a sensory system can regulate behaviour.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tirindelli, R., Dibattista, M., Pifferi, S. & Menini, A. From pheromones to behavior. Physiol. Rev. 89, 921–956 (2009)
Leypold, B. G. et al. Altered sexual and social behaviors in trp2 mutant mice. Proc. Natl Acad. Sci. USA 99, 6376–6381 (2002)
Stowers, L., Holy, T. E., Meister, M., Dulac, C. & Koentges, G. Loss of sex discrimination and male-male aggression in mice deficient for TRP2. Science 295, 1493–1500 (2002)
Ferrero, D. M. & Liberles, S. D. The secret codes of mammalian scents. Wiley Interdiscip. Rev. Syst. Biol. Med. 2, 23–33 (2010)
Nodari, F. et al. Sulfated steroids as natural ligands of mouse pheromone-sensing neurons. J. Neurosci. 28, 6407–6418 (2008)
Novotny, M. V. Pheromones, binding proteins and receptor responses in rodents. Biochem. Soc. Trans. 31, 117–122 (2003)
Touhara, K. Sexual communication via peptide and protein pheromones. Curr. Opin. Pharmacol. 8, 759–764 (2008)
Karn, R. C. & Laukaitis, C. M. The roles of gene duplication, gene conversion and positive selection in rodent esp and mup pheromone gene families with comparison to the abp family. PLoS ONE 7, e47697 (2012)
Kimoto, H., Haga, S., Sato, K. & Touhara, K. Sex-specific peptides from exocrine glands stimulate mouse vomeronasal sensory neurons. Nature 437, 898–901 (2005)
Logan, D. W., Marton, T. F. & Stowers, L. Species specificity in major urinary proteins by parallel evolution. PLoS ONE 3, e3280 (2008)
Chamero, P. et al. G protein Gαo is essential for vomeronasal function and aggressive behavior in mice. Proc. Natl Acad. Sci. USA 108, 12898–12903 (2011)
Papes, F., Logan, D. W. & Stowers, L. The vomeronasal organ mediates interspecies defensive behaviors through detection of protein pheromone homologs. Cell 141, 692–703 (2010)
Haga, S. et al. The male mouse pheromone ESP1 enhances female sexual receptive behaviour through a specific vomeronasal receptor. Nature 466, 118–122 (2010)
Kimoto, H. et al. Sex- and strain-specific expression and vomeronasal activity of mouse ESP family peptides. Curr. Biol. 17, 1879–1884 (2007)
Li, Q. et al. Synchronous evolution of an odor biosynthesis pathway and behavioral response. Curr. Biol. 23, 11–20 (2013)
Mandiyan, V. S., Coats, J. K. & Shah, N. M. Deficits in sexual and aggressive behaviors in Cnga2 mutant mice. Nature Neurosci. 8, 1660–1662 (2005)
Wang, Z. et al. Pheromone detection in male mice depends on signaling through the type 3 adenylyl cyclase in the main olfactory epithelium. J. Neurosci. 26, 7375–7379 (2006)
He, J. et al. Distinct signals conveyed by pheromone concentrations to the mouse vomeronasal organ. J. Neurosci. 30, 7473–7483 (2010)
Dulac, C. & Wagner, S. Genetic analysis of brain circuits underlying pheromone signaling. Annu. Rev. Genet. 40, 449–467 (2006)
Choi, G. B. et al. Lhx6 delineates a pathway mediating innate reproductive behaviors from the amygdala to the hypothalamus. Neuron 46, 647–660 (2005)
Yang, C. F. et al. Sexually dimorphic neurons in the ventromedial hypothalamus govern mating in both sexes and aggression in males. Cell 153, 896–909 (2013)
Chamero, P. et al. Identification of protein pheromones that promote aggressive behaviour. Nature 450, 899–902 (2007)
Novotny, M., Harvey, S., Jemiolo, B. & Alberts, J. Synthetic pheromones that promote inter-male aggression in mice. Proc. Natl Acad. Sci. USA 82, 2059–2061 (1985)
Liberles, S. D. & Buck, L. B. A second class of chemosensory receptors in the olfactory epithelium. Nature 442, 645–650 (2006)
Montmayeur, J. P., Liberles, S. D., Matsunami, H. & Buck, L. B. A candidate taste receptor gene near a sweet taste locus. Nature Neurosci. 4, 492–498 (2001)
Liberles, S. D. et al. Formyl peptide receptors are candidate chemosensory receptors in the vomeronasal organ. Proc. Natl Acad. Sci. USA 106, 9842–9847 (2009)
Shevchenko, A., Wilm, M., Vorm, O. & Mann, M. Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal. Chem. 68, 850–858 (1996)
Peng, J. & Gygi, S. P. Proteomics: the move to mixtures. J. Mass Spectrom. 36, 1083–1091 (2001)
Eng, J. K., Mccormack, A. L. & Yates, J. R. An approach to correlate tandem mass-spectral data of peptides with amino-acid-sequences in a protein database. J. Am. Soc. Mass Spectrom. 5, 976–989 (1994)
Hagendorf, S., Fluegge, D., Engelhardt, C. & Spehr, M. Homeostatic control of sensory output in basal vomeronasal neurons: activity-dependent expression of ether-a-go-go-related gene potassium channels. J. Neurosci. 29, 206–221 (2009)
Spehr, J. et al. Ca2+-calmodulin feedback mediates sensory adaptation and inhibits pheromone-sensitive ion channels in the vomeronasal organ. J. Neurosci. 29, 2125–2135 (2009)
Franklin, K. & Paxinos, G. The Mouse Brain in Stereotaxic Coordinates Fig. 44 (Academic, 2008)
Acknowledgements
We thank M. Albers and S. R. Datta for reading the manuscript, J. Yang, C. Mark Fletcher and Y. Tachie-Baffour for experimental assistance, and the Taplin Mass Spectrometry Facility for mass spectrometry analysis. This work was supported by a grant from the National Institutes of Health (to S.D.L., award number R01 DC010155) and in part by a Grant-in-Aid for Young Scientists (S) from the Japan Society for the Promotion of Science, and by ERATO Touhara Chemosensory Signal Project from the Japan Science and Technology Agency (to K.T.). N.H. is supported by a Grant-in-Aid for JSPS Fellows, M.S. is a Lichtenberg-Professor of the Volkswagen Foundation and D.M.F. is supported by a Boehringer Ingelheim Fonds PhD Fellowship.
Author information
Authors and Affiliations
Contributions
D.M.F., S.D.L., M.S. and K.T. conceived the project, designed the experiments and wrote the manuscript. D.M.F. performed molecular biology, biochemistry and behaviour experiments. D.S.R. and Q.L. performed in situ hybridization analysis. L.M.M., A.C., T.O. and N.H. performed electrophysiological analysis. T.O. performed cFos analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 RNA in situ hybridization to characterize expression of Esp genes in the lacrimal gland.
a–c, Colorimetric analysis in tissue from animals indicated using cRNA riboprobes for (a) Esp15 and (b) Esp22, and two-colour fluorescence analysis (c) in juvenile lacrimal gland with cRNA riboprobes for Esp22 (red) and a marker for acinal secretory cells, Rab3D (green). cRNA riboprobes for Esp15 are expected to cross-hybridize with Esp16 mRNA. Some images used in b are identical to panels in Fig. 1d, and are included for reference. Dashed boxes (c) indicate regions magnified below. Arrows, acinar cells; arrowheads, ductal cells; scale bars, 100 μm (a, b, c top), 20 μm (c bottom).
Extended Data Figure 2 qPCR analysis of gene expression.
a, Esp22 qPCR primers specifically detect a plasmid containing cloned Esp22, but not plasmids containing other Esps with greater than 60% identity to Esp22. b–f, cDNA was derived from lacrimal gland (b–e), submaxillary gland (e) or other tissues (f) of animals indicated. In f, abundance is calculated by normalization to amounts of Gapdh. C57BL/6 mice were used (b–d) unless otherwise indicated (b). Experiments where sex is not indicated involved equal numbers of males and females; olfactory epithelium (OE), olfactory bulb (OB), harderian gland (HG), submaxillary gland (SMG), parotid gland (PG), sublingual gland (SLG) (n = 6–12, averages ± s.e.m., **P < 0.01, two-way ANOVA followed by Tukey’s HSD post hoc tests).
Extended Data Figure 3 Quantification of protein concentrations in tear fluid by western blot analysis using an anti-ESP22 antibody.
a, b, A standard curve based on signal intensity was generated using different concentrations of recombinant ESP22 (a, left panel; b). The arrow indicates the intensity level of the band in the juvenile tear sample (a, right panel). c, Entire western blot analysis of tear fluid using anti-ESP22 antibody.
Extended Data Figure 4 ESP22-derived tryptic peptides identified by mass spectrometry.
a, The amino-acid sequence of immature ESP22 is depicted, along with a predicted signal peptide and the epitope used for antibody generation. Four tryptic peptides were identified by mass spectrometry (highlighted in red), including one peptide containing the first amino acid after the predicted signal sequence and another containing the encoded carboxy (C)-terminal residue. Trypsin does not efficiently cleave amino (N)-terminal lysines or arginines, consistent with R23 being the first amino acid in mature ESP22. b, Mass spectrum of an high-performance liquid chromatography fraction of juvenile tear fluid showing the ESP22-derived tryptic peptide GIVFNTIK, with sequence identity confirmed by tandem mass spectrometry analysis.
Extended Data Figure 5 Electrophysiological responses to ESP22 in VNO sensory neurons.
a, Single-unit extracellular loose-seal recording from a single VNO sensory neuron repeatedly exposed to different stimuli indicates reproducibility of responses. b, The percentage of basal VNO sensory neurons responsive to 20 pM (n = 383) and 2 nM (n = 749) ESP22.
Extended Data Figure 6 cFos responses to ESP22 in the amygdala.
a, ESP22 and juvenile tear fluid, but not MBP, induce cFos expression in the postero-ventral MeA. Dashed lines and arrows indicate boundaries of MeA regions. b, Similar responses were not observed in other amygdala nuclei that receive olfactory input, including the postero-medial cortical amygdala (PMCo), anterior cortical amygdala (CoA) and postero-lateral cortical amygdala (PLCo) (mean ± s.e.m., n = 3).
Extended Data Figure 7 Trpc2−/− males show increased sexual behaviour towards wild-type juveniles.
a, b, Histograms of mounts by minute of social interaction and intermount intervals shown towards juveniles byTrpc2+/+ and Trpc2−/− males (sum, n = 12). Inset depicts average intermount intervals (mean ± s.e.m., *P < 0.05, **P < 0.01, Mann–Whitney U-test). c, Analysis of adult male sexual behaviour during simultaneous interaction with juvenile and adult oestrous females. Trpc2+/+ and Trpc2−/− males show similar amounts of sexual behaviour towards adult oestrous females, but Trpc2−/− males show increased sexual behaviour towards juveniles (n = 10, averages ± s.e.m., *P < 0.05, **P < 0.01, one-way multivariate ANOVA).
Extended Data Figure 8 Trpc2−/− males show sexual behaviour towards juvenile males.
a, Raster plots depicting individual mounting displays of adult Trpc2+/+ and Trpc2−/− males towards juvenile males (C57BL/6, postnatal day 17) during social interaction (30 min). Each tick indicates onset of one mount. b, Quantitative analysis of parameters associated with sexual behaviour towards juvenile males shown by Trpc2+/+ and Trpc2−/− males (n = 11 or 12, averages ± s.e.m., *P < 0.05, **P < 0.01, Mann–Whitney U-test).
Extended Data Figure 9 ESP22 did not decrease social investigation time.
Wild-type C57BL/6 males were introduced to C3H juvenile females painted with stimuli indicated. Social investigation time of the male was recorded as time spent with the nose in direct contact with the female. These data were extracted from the same experiments reported in Fig. 4c, d, with additional experiments involving TMT (100 μl, 155 mM, n = 11 or 12, averages ± s.e.m., **P < 0.01, one-way ANOVA followed by Tukey’s HSD post hoc tests).
Extended Data Figure 10 ESP22 (10 μg) inhibits sexual behaviour of C3H males.
a, Raster plots of sexual behaviour shown by C3H males towards C3H juvenile females (postnatal day 17) painted with indicated stimuli (30 min social interaction). Each tick indicates onset of one mount. b, Quantitative analysis of parameters associated with sexual behaviour towards juvenile females shown by C3H males (n = 11, averages ± s.e.m., *P < 0.05, **P < 0.01, one-way ANOVA followed by Tukey’s HSD post hoc tests).
Supplementary information
A Trpc2+/+ adult male interacts with a juvenile female
Video showing a social interaction between a wild type adult male mouse and a juvenile female mouse (C57BL/6, p17). Animals in the video display active social investigation but not sexual behavior. (MOV 4084 kb)
A Trpc2-/- adult male displays sexual behavior towards a juvenile female
Video showing a social interaction between a Trpc2-/- adult male mouse and a juvenile female mouse (C57BL/6, p17). Frequent mounting displays characteristic of mouse sexual behavior are observed. (MOV 3320 kb)
Rights and permissions
About this article
Cite this article
Ferrero, D., Moeller, L., Osakada, T. et al. A juvenile mouse pheromone inhibits sexual behaviour through the vomeronasal system. Nature 502, 368–371 (2013). https://doi.org/10.1038/nature12579
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12579
This article is cited by
-
Reproductives signature revealed by protein profiling and behavioral bioassays in termite
Scientific Reports (2023)
-
Natural and Pathological Aging Distinctively Impacts the Pheromone Detection System and Social Behavior
Molecular Neurobiology (2023)
-
Hemoglobin in the blood acts as a chemosensory signal via the mouse vomeronasal system
Nature Communications (2022)
-
From immune to olfactory expression: neofunctionalization of formyl peptide receptors
Cell and Tissue Research (2021)
-
Coding of pheromones by vomeronasal receptors
Cell and Tissue Research (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.