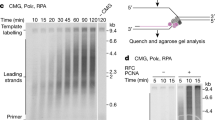
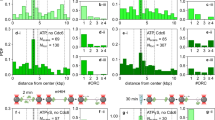
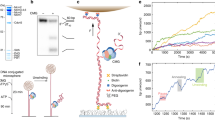
Abstract
Eukaryotic cells initiate DNA replication from multiple origins, which must be tightly regulated to promote precise genome duplication in every cell cycle. To accomplish this, initiation is partitioned into two temporally discrete steps: a double hexameric minichromosome maintenance (MCM) complex is first loaded at replication origins during G1 phase, and then converted to the active CMG (Cdc45–MCM–GINS) helicase during S phase. Here we describe the reconstitution of budding yeast DNA replication initiation with 16 purified replication factors, made from 42 polypeptides. Origin-dependent initiation recapitulates regulation seen in vivo. Cyclin-dependent kinase (CDK) inhibits MCM loading by phosphorylating the origin recognition complex (ORC) and promotes CMG formation by phosphorylating Sld2 and Sld3. Dbf4-dependent kinase (DDK) promotes replication by phosphorylating MCM, and can act either before or after CDK. These experiments define the minimum complement of proteins, protein kinase substrates and co-factors required for regulated eukaryotic DNA replication.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tanaka, S. & Araki, H. Helicase activation and establishment of replication forks at chromosomal origins of replication. Cold Spring Harb. Perspect. Biol. 5, a010371 (2013)
Siddiqui, K., On, K. F. & Diffley, J. F. X. Regulating DNA replication in Eukarya. Cold Spring Harb. Perspect. Biol. 5, a012930 (2013)
Remus, D. et al. Concerted loading of Mcm2–7 double hexamers around DNA during DNA replication origin licensing. Cell 139, 719–730 (2009)
Evrin, C. et al. A double-hexameric MCM2-7 complex is loaded onto origin DNA during licensing of eukaryotic DNA replication. Proc. Natl Acad. Sci. USA 106, 20240–20245 (2009)
Gros, J., Devbhandari, S. & Remus, D. Origin plasticity during budding yeast DNA replication in vitro. EMBO J. 33, 621–636 (2014)
On, K. F. et al. Prereplicative complexes assembled in vitro support origin-dependent and independent DNA replication. EMBO J. 33, 605–620 (2014)
Heller, R. C. et al. Eukaryotic origin-dependent DNA replication in vitro reveals sequential action of DDK and S-CDK kinases. Cell 146, 80–91 (2011)
Labib, K. How do Cdc7 and cyclin-dependent kinases trigger the initiation of chromosome replication in eukaryotic cells? Genes Dev. 24, 1208–1219 (2010)
Nguyen, V. Q., Co, C. & Li, J. J. Cyclin-dependent kinases prevent DNA re-replication through multiple mechanisms. Nature 411, 1068–1073 (2001)
Zegerman, P. & Diffley, J. F. X. Phosphorylation of Sld2 and Sld3 by cyclin-dependent kinases promotes DNA replication in budding yeast. Nature 445, 281–285 (2007)
Tanaka, S. et al. CDK-dependent phosphorylation of Sld2 and Sld3 initiates DNA replication in budding yeast. Nature 445, 328–332 (2007)
Masumoto, H., Muramatsu, S., Kamimura, Y. & Araki, H. S-Cdk-dependent phosphorylation of Sld2 essential for chromosomal DNA replication in budding yeast. Nature 415, 651–655 (2002)
Randell, J. C. et al. Mec1 is one of multiple kinases that prime the Mcm2–7 helicase for phosphorylation by Cdc7. Mol. Cell 40, 353–363 (2010)
Sheu, Y. J. & Stillman, B. Cdc7-Dbf4 phosphorylates MCM proteins via a docking site-mediated mechanism to promote S phase progression. Mol. Cell 24, 101–113 (2006)
Sheu, Y. J. & Stillman, B. The Dbf4–Cdc7 kinase promotes S phase by alleviating an inhibitory activity in Mcm4. Nature 463, 113–117 (2010)
Moyer, S. E., Lewis, P. W. & Botchan, M. R. Isolation of the Cdc45/Mcm2–7/GINS (CMG) complex, a candidate for the eukaryotic DNA replication fork helicase. Proc. Natl Acad. Sci. USA 103, 10236–10241 (2006)
Gambus, A. et al. GINS maintains association of Cdc45 with MCM in replisome progression complexes at eukaryotic DNA replication forks. Nature Cell Biol. 8, 358–366 (2006)
Bell, S. P. & Dutta, A. DNA replication in eukaryotic cells. Annu. Rev. Biochem. 71, 333–374 (2002)
Gambus, A. et al. A key role for Ctf4 in coupling the MCM2–7 helicase to DNA polymerase α within the eukaryotic replisome. EMBO J. 28, 2992–3004 (2009)
Rowles, A., Tada, S. & Blow, J. J. Changes in association of the Xenopus origin recognition complex with chromatin on licensing of replication origins. J. Cell Sci. 112, 2011–2018 (1999)
Watase, G., Takisawa, H. & Kanemaki, M. T. Mcm10 plays a role in functioning of the eukaryotic replicative DNA helicase, Cdc45-Mcm-GINS. Curr. Biol. 22, 343–349 (2012)
van Deursen, F., Sengupta, S., De Piccoli, G., Sanchez-Diaz, A. & Labib, K. Mcm10 associates with the loaded DNA helicase at replication origins and defines a novel step in its activation. EMBO J. 31, 2195–2206 (2012)
Georgescu, R. E. et al. Mechanism of asymmetric polymerase assembly at the eukaryotic replication fork. Nature Struct. Mol. Biol. 21, 664–670 (2014)
Clausen, A. R. et al. Tracking replication enzymology in vivo by genome-wide mapping of ribonucleotide incorporation. Nature Struct. Mol. Biol. http://dx.doi.org/10.1038/nsmb.2957 (2015)
Yu, C. et al. Strand-specific analysis shows protein binding at replication forks and PCNA unloading from lagging strands when forks stall. Mol. Cell 56, 551–563 (2014)
Chen, S. & Bell, S. P. CDK prevents Mcm2–7 helicase loading by inhibiting Cdt1 interaction with Orc6. Genes Dev. 25, 363–372 (2011)
Frigola, J., Remus, D., Mehanna, A. & Diffley, J. F. X. ATPase-dependent quality control of DNA replication origin licensing. Nature 495, 339–343 (2013)
Janke, C. et al. A versatile toolbox for PCR-based tagging of yeast genes: new fluorescent proteins, more markers and promoter substitution cassettes. Yeast 21, 947–962 (2004)
Boos, D. et al. Regulation of DNA replication through Sld3-Dpb11 interaction is conserved from yeast to humans. Curr. Biol. 21, 1152–1157 (2011)
Simon, A. C. et al. A Ctf4 trimer couples the CMG helicase to DNA polymerase alpha in the eukaryotic replisome. Nature 510, 293–297 (2014)
Coster, G., Frigola, J., Beuron, F., Morris, E. P. & Diffley, J. F. X. Origin licensing requires ATP binding and hydrolysis by the MCM replicative helicase. Mol. Cell 55, 666–677 (2014)
Brown, N. R. et al. The crystal structure of cyclin A. Structure 3, 1235–1247 (1995)
Itou, H., Muramatsu, S., Shirakihara, Y. & Araki, H. Crystal structure of the homology domain of the eukaryotic DNA replication proteins Sld3/Treslin. Structure 22, 1341–1347 (2014)
Sharp, P. M. & Li, W. H. The codon adaptation index-a measure of directional synonymous codon usage bias, and its potential applications. Nucleic Acids Res. 15, 1281–1295 (1987)
Acknowledgements
We are grateful to B. Pfander and M. Douglas for the Mcm10 expression plasmid and advice on purification, C. Kurat for construction of plasmids used to generate ARS1 linear templates, K. On and D. Boos for Sic1 and A-Cdk2 and K. Labib for Psf1 antibodies and the E. coli GINS expression strain. We thank A. Alidoust and N. Patel for growing yeast cultures. This work was supported by Cancer Research UK, a FEBS Return-to-Europe fellowship to J.T.P.Y., a Boehringer Ingelheim Fonds PhD fellowship to A.J. and an ERC grant (249883 – EUKDNAREP) to J.F.X.D.
Author information
Authors and Affiliations
Contributions
J.F.X.D. and J.T.P.Y. designed the experiments and wrote the manuscript. J.T.P.Y. performed the experiments. T.D.D., A.J. and A.E. generated protein expression constructs and strains, and established protein purification protocols.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 2 Analysis of firing factor recruitment.
a, Immunoblots of protein recruitment conducted as in Fig. 1b but with 0.3 M KCl washes. b, Stability of recruited firing factors following washes of varying strength (lanes 1 and 5, 0.3 M K-glu; lanes 2 and 6, 0.3 M KCl; lanes 3 and 7, 0.45 M KCl; lanes 4 and 8, 0.6 M KCl). c, Psf2–Flag was depleted from a yJY18 S phase extract by two rounds of incubation with anti-Flag M2 magnetic beads. Levels of Psf2–Flag were determined by immunoblotting with the Flag-M2 antibody. Soluble and bead bound protein fractions are illustrated. d, Extract-based replication reaction schemes. In the left pathway (i), loaded MCM is treated with DDK and added to a KO3 extract (Sld3, Sld7, Cdc45, Dpb11, Sld2 overexpression). In the right pathway (ii), firing factors are recruited to MCM as illustrated in Fig. 1b and the complex is added to a yJY18 extract (no firing factor overexpression) in which Psf2 (GINS complex) has been depleted. e, Replication reactions as described in d using A-Cdk2 for firing factor recruitment. Where indicated, Sic1 was added to the extract 20 min before replication.
Extended Data Figure 3 Characterization of the in vitro DNA replication reaction.
a, b, Replication reactions conducted as in Fig. 3a with A-Cdk2 on ARS1 circular DNA templates for 1 h. c, Time course of a standard replication reaction using A-Cdk2 on the ARS1 linear DNA template. d, Quantitation of a time course conducted as in c. DNA synthesis was normalized to the total DNA synthesis at 90 min. Small and large replication products were not quantified separately at 5 min as they are not well resolved at this time point. e, Pulse chase experiment conducted with A-Cdk2 on the ARS1 linear DNA template. For the pulse the dCTP concentration was reduced to 4 μM. Following a 10 min incubation unlabelled dCTP was added to 100 μM.
Extended Data Figure 4 Characterization of RPA recruitment.
Unless stated reactions were conducted on ARS1 circular templates. a, Vaccinia virus topoisomerase I supports DNA replication with purified proteins. Replication reactions with either Topo II (25 nM) or vaccinia virus topoisomerase I (0.125 units per μl). Two different Topo II fractions (Fr1 and Fr2) were used for comparison. b, Nucleotide dependence of RPA recruitment in a complete replication reaction with Topo II. c, RPA recruitment reactions were conducted on ARS1 circular template in the presence of vaccinia virus topoisomerase I (0.125 units per μl), or on a linear template in the absence of a topoisomerase. dNTPs, C/G/UTP, pol α and Ctf4 were omitted from the final step of the reaction.
Extended Data Figure 5 Regulation of MCM loading and origin firing by S-CDK is ATP dependent.
a, Replication reaction where ORC was pre-incubated with S-CDK before MCM loading. When Sic1 was added before ORC the mix was incubated for 5 min and ORC was then added for 10 min. b, Pre-incubation of ORC with S-CDK in the presence or absence of ATP. After incubation with Sic1, ATP was added to the reaction lacking ATP. c, Sld3/7 and Sld2 were pre-incubated with S-CDK as illustrated in Fig. 5d in the presence or absence of ATP. Following incubation with Sic1, samples that did not contain ATP for the pre-incubation step were supplemented with ATP.
Extended Data Figure 6 Cartoon illustrating protein kinase regulated eukaryotic DNA replication origin firing with purified proteins.
Firing factors are recruited to loaded MCM in a DDK- and CDK-dependent manner. DNA synthesis is initiated once the DNA template has been unwound. CDK also functions to inhibit MCM loading by phosphorylating ORC.
Extended Data Figure 7 Internally Flag-tagged Cdc45 supports normal DNA replication in S phase extracts.
a, The previously reported interaction between Sld3 and Cdc45 (ref. 33) was exploited to co-immunoprecipitate Cdc45 from yJY16 extracts (Dpb11 and Sld2 overexpression) by incubation with Flag–Sld3/7 that was pre-coupled to anti-Flag M2 magnetic beads. b, In vitro extract-based replication reaction on soluble ARS1 circular template using yJY16 extracts where endogenous Cdc45 was depleted as indicated. The extract was supplemented with purified Sld3/7 as the complex is not overexpressed in yJY16. The experiment was conducted for 30 min as described previously6 and products were separated through a 1% native agarose gel. Internally Flag-tagged Cdc45 (52 nM) was added back as indicated. The locations of the different replication products are illustrated.
Supplementary information
Supplementary Information
This file contains the DNA sequences used for expression in S.cerevisiae. (PDF 292 kb)
Rights and permissions
About this article
Cite this article
Yeeles, J., Deegan, T., Janska, A. et al. Regulated eukaryotic DNA replication origin firing with purified proteins. Nature 519, 431–435 (2015). https://doi.org/10.1038/nature14285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14285
This article is cited by
-
CST–polymerase α-primase solves a second telomere end-replication problem
Nature (2024)
-
Control of DNA replication in vitro using a reversible replication barrier
Nature Protocols (2024)
-
Unwinding Helicase MCM Functionality for Diagnosis and Therapeutics of Replication Abnormalities Associated with Cancer: A Review
Molecular Diagnosis & Therapy (2024)
-
Identification of genetic profile and biomarkers involved in acute respiratory distress syndrome
Intensive Care Medicine (2024)
-
A chromatinized origin reduces the mobility of ORC and MCM through interactions and spatial constraint
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.