Abstract
The flipping of membrane-embedded lipids containing large, polar head groups is slow and energetically unfavourable, and is therefore catalysed by flippases, the mechanisms of which are unknown. A prominent example of a flipping reaction is the translocation of lipid-linked oligosaccharides that serve as donors in N-linked protein glycosylation. In Campylobacter jejuni, this process is catalysed by the ABC transporter PglK. Here we present a mechanism of PglK-catalysed lipid-linked oligosaccharide flipping based on crystal structures in distinct states, a newly devised in vitro flipping assay, and in vivo studies. PglK can adopt inward- and outward-facing conformations in vitro, but only outward-facing states are required for flipping. While the pyrophosphate-oligosaccharide head group of lipid-linked oligosaccharides enters the translocation cavity and interacts with positively charged side chains, the lipidic polyprenyl tail binds and activates the transporter but remains exposed to the lipid bilayer during the reaction. The proposed mechanism is distinct from the classical alternating-access model applied to other transporters.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Burda, P. & Aebi, M. The dolichol pathway of N-linked glycosylation. Biochim. Biophys. Acta 1426, 239–257 (1999)
Helenius, J. et al. Translocation of lipid-linked oligosaccharides across the ER membrane requires Rft1 protein. Nature 415, 447–450 (2002)
Sprong, H., van der Sluijs, P. & van Meer, G. How proteins move lipids and lipids move proteins. Nature Rev. Mol. Cell Biol. 2, 504–513 (2001)
Sebastian, T. T., Baldridge, R. D., Xu, P. & Graham, T. R. Phospholipid flippases: building asymmetric membranes and transport vesicles. Biochim. Biophys. Acta 1821, 1068–1077 (2012)
Hankins, H. M., Baldridge, R. D., Xu, P. & Graham, T. R. Role of flippases, scramblases and transfer proteins in phosphatidylserine subcellular distribution. Traffic 16, 35–47 (2015)
Krahling, S., Callahan, M. K., Williamson, P. & Schlegel, R. A. Exposure of phosphatidylserine is a general feature in the phagocytosis of apoptotic lymphocytes by macrophages. Cell Death Differ. 6, 183–189 (1999)
Balasubramanian, K. & Schroit, A. J. Aminophospholipid asymmetry: A matter of life and death. Annu. Rev. Physiol. 65, 701–734 (2003)
Cuthbertson, L., Kos, V. & Whitfield, C. ABC transporters involved in export of cell surface glycoconjugates. Microbiol. Mol. Biol. Rev. 74, 341–362 (2010)
Cuthbertson, L., Kimber, M. S. & Whitfield, C. Substrate binding by a bacterial ABC transporter involved in polysaccharide export. Proc. Natl Acad. Sci. USA 104, 19529–19534 (2007)
Clarke, B. R., Cuthbertson, L. & Whitfield, C. Nonreducing terminal modifications determine the chain length of polymannose O antigens of Escherichia coli and couple chain termination to polymer export via an ATP-binding cassette transporter. J. Biol. Chem. 279, 35709–35718 (2004)
Sharom, F. J. Flipping and flopping–lipids on the move. IUBMB Life 63, 736–746 (2011)
Kodigepalli, K. M., Bowers, K., Sharp, A. & Nanjundan, M. Roles and regulation of phospholipid scramblases. FEBS Lett. 589, 3–14 (2015)
Brunner, J. D., Lim, N. K., Schenck, S., Duerst, A. & Dutzler, R. X-ray structure of a calcium-activated TMEM16 lipid scramblase. Nature 516, 207–212 (2014)
Hvorup, R. N. et al. The multidrug/oligosaccharidyl-lipid/polysaccharide (MOP) exporter superfamily. Eur. J. Biochem. 270, 799–813 (2003)
Lopez-Marques, R. L., Theorin, L., Palmgren, M. G. & Pomorski, T. G. P4-ATPases: lipid flippases in cell membranes. Pflugers Arch. 466, 1227–1240 (2014)
Eckford, P. D. & Sharom, F. J. The reconstituted Escherichia coli MsbA protein displays lipid flippase activity. Biochem. J. 429, 195–203 (2010)
Ward, A., Reyes, C. L., Yu, J., Roth, C. B. & Chang, G. Flexibility in the ABC transporter MsbA: Alternating access with a twist. Proc. Natl Acad. Sci. USA 104, 19005–19010 (2007)
Wacker, M. et al. N-linked glycosylation in Campylobacter jejuni and its functional transfer into E. coli . Science 298, 1790–1793 (2002)
Young, N. M. et al. Structure of the N-linked glycan present on multiple glycoproteins in the Gram-negative bacterium, Campylobacter jejuni . J. Biol. Chem. 277, 42530–42539 (2002)
Alaimo, C. et al. Two distinct but interchangeable mechanisms for flipping of lipid-linked oligosaccharides. EMBO J. 25, 967–976 (2006)
Lizak, C., Gerber, S., Numao, S., Aebi, M. & Locher, K. P. X-ray structure of a bacterial oligosaccharyltransferase. Nature 474, 350–355 (2011)
Tatar, L. D., Marolda, C. L., Polischuk, A. N., van Leeuwen, D. & Valvano, M. A. An Escherichia coli undecaprenyl-pyrophosphate phosphatase implicated in undecaprenyl phosphate recycling. Microbiology 153, 2518–2529 (2007)
Sanyal, S. & Menon, A. K. Specific transbilayer translocation of dolichol-linked oligosaccharides by an endoplasmic reticulum flippase. Proc. Natl Acad. Sci. USA 106, 767–772 (2009)
Sanyal, S. & Menon, A. K. Stereoselective transbilayer translocation of mannosyl phosphoryl dolichol by an endoplasmic reticulum flippase. Proc. Natl Acad. Sci. USA 107, 11289–11294 (2010)
Linton, D. et al. Functional analysis of the Campylobacter jejuni N-linked protein glycosylation pathway. Mol. Microbiol. 55, 1695–1703 (2005)
Lizak, C. et al. A catalytically essential motif in external loop 5 of the bacterial oligosaccharyltransferase PglB. J. Biol. Chem. 289, 735–746 (2014)
Troutman, J. M. & Imperiali, B. Campylobacter jejuni PglH is a single active site processive polymerase that utilizes product inhibition to limit sequential glycosyl transfer reactions. Biochemistry 48, 2807–2816 (2009)
Abeijon, C. & Hirschberg, C. B. Topography of initiation of N-glycosylation reactions. J. Biol. Chem. 265, 14691–14695 (1990)
Hanover, J. A. & Lennarz, W. J. The topological orientation of N,N′-diacetylchitobiosylpyrophosphoryldolichol in artificial and natural membranes. J. Biol. Chem. 254, 9237–9246 (1979)
Gerber, S. et al. Mechanism of bacterial oligosaccharyltransferase: in vitro quantification of sequon binding and catalysis. J. Biol. Chem. 288, 8849–8861 (2013)
Lizak, C. et al. Unexpected reactivity and mechanism of carboxamide activation in bacterial N-linked protein glycosylation. Nature Commun. 4, 2627 (2013)
Siarheyeva, A. & Sharom, F. J. The ABC transporter MsbA interacts with lipid A and amphipathic drugs at different sites. Biochem. J. 419, 317–328 (2009)
Jin, M. S., Oldham, M. L., Zhang, Q. & Chen, J. Crystal structure of the multidrug transporter P-glycoprotein from Caenorhabditis elegans . Nature 490, 566–569 (2012)
Raetz, C. R., Reynolds, C. M., Trent, M. S. & Bishop, R. E. Lipid A modification systems in gram-negative bacteria. Annu. Rev. Biochem. 76, 295–329 (2007)
Lee, J. Y., Yang, J. G., Zhitnitsky, D., Lewinson, O. & Rees, D. C. Structural basis for heavy metal detoxification by an Atm1-type ABC exporter. Science 343, 1133–1136 (2014)
Dawson, R. J. & Locher, K. P. Structure of a bacterial multidrug ABC transporter. Nature 443, 180–185 (2006)
Zaitseva, J. et al. A structural analysis of asymmetry required for catalytic activity of an ABC-ATPase domain dimer. EMBO J. 25, 3432–3443 (2006)
Smith, P. C. et al. ATP binding to the motor domain from an ABC transporter drives formation of a nucleotide sandwich dimer. Mol. Cell 10, 139–149 (2002)
Choudhury, H. G. et al. Structure of an antibacterial peptide ATP-binding cassette transporter in a novel outward occluded state. Proc. Natl Acad. Sci. USA 111, 9145–9150 (2014)
Shintre, C. A. et al. Structures of ABCB10, a human ATP-binding cassette transporter in apo- and nucleotide-bound states. Proc. Natl Acad. Sci. USA 110, 9710–9715 (2013)
Voss, N. R. & Gerstein, M. 3V: cavity, channel and cleft volume calculator and extractor. Nucleic Acids Res. 38, W555–W562 (2010)
Whitfield, C. & Trent, M. S. Biosynthesis and export of bacterial lipopolysaccharides. Annu. Rev. Biochem. 83, 99–128 (2014)
Korkhov, V. M., Mireku, S. A. & Locher, K. P. Structure of AMP-PNP-bound vitamin B12 transporter BtuCD-F. Nature 490, 367–372 (2012)
Vestergaard, A. L. et al. Critical roles of isoleucine-364 and adjacent residues in a hydrophobic gate control of phospholipid transport by the mammalian P4-ATPase ATP8A2. Proc. Natl Acad. Sci. USA 111, E1334–E1343 (2014)
Stone, A. & Williamson, P. Outside of the box: recent news about phospholipid translocation by P4 ATPases. J. Chem. Biol. 5, 131–136 (2012)
Pomorski, T. & Menon, A. K. Lipid flippases and their biological functions. Cell. Mol. Life Sci. 63, 2908–2921 (2006)
Hug, I. & Feldman, M. F. Analogies and homologies in lipopolysaccharide and glycoprotein biosynthesis in bacteria. Glycobiology 21, 138–151 (2011)
Jones, C. Vaccines based on the cell surface carbohydrates of pathogenic bacteria. An. Acad. Bras. Cienc. 77, 293–324 (2005)
Kabsch, W. Xds. Acta Crystallogr. D 66, 125–132 (2010)
Strong, M. et al. Toward the structural genomics of complexes: crystal structure of a PE/PPE protein complex from Mycobacterium tuberculosis. Proc. Natl Acad. Sci. USA 103, 8060–8065 (2006)
Karplus, P. A. & Diederichs, K. Linking crystallographic model and data quality. Science 336, 1030–1033 (2012)
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
Sheldrick, G. M. A short history of SHELX. Acta Crystallogr. A 64, 112–122 (2008)
Bricogne, G., Vonrhein, C., Flensburg, C., Schiltz, M. & Paciorek, W. Generation, representation and flow of phase information in structure determination: recent developments in and around SHARP 2.0. Acta Crystallogr. D. 59, 2023–2030 (2003)
Abrahams, J. P. & Leslie, A. G. Methods used in the structure determination of bovine mitochondrial F1 ATPase. Acta Crystallogr. D 52, 30–42 (1996)
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Lizak, C., Fan, Y. Y., Weber, T. C. & Aebi, M. N-Linked glycosylation of antibody fragments in Escherichia coli. Bioconjug. Chem. 22, 488–496 (2011)
Lefebre, M. D. & Valvano, M. A. Construction and evaluation of plasmid vectors optimized for constitutive and regulated gene expression in Burkholderia cepacia complex isolates. Appl. Environ. Microbiol. 68, 5956–5964 (2002)
Liu, F. et al. Rationally designed short polyisoprenol-linked PglB substrates for engineered polypeptide and protein N-glycosylation. J. Am. Chem. Soc. 136, 566–569 (2014)
Acknowledgements
We thank the staff scientists at the PX beamline of the Swiss Light Source for help with data collection, and M. Napiorkowska and A.Ramirez for assistance with PglB assays. This work was supported by the Swiss National Science Foundation (SNF 31003A–146191 to K.P.L. and Transglyco Sinergia program to M.A., J.-L.R. and K.P.L.). C.P. acknowledges support from the ETH postdoctoral fellowship program.
Author information
Authors and Affiliations
Contributions
C.P. determined the structures of PglK, established the in vitro flipping assay, and performed in vivo flipping studies. S.G. crystallized PglK in the apo-inward-2 state, M.B. assisted in expression and purification of PglK. J.B., T.D. and J.-L.R. synthesized LLO analogues. K.P.L., S.G. and C.P. conceived the project. K.P.L., M.A., and C.P. analysed the data. K.P.L. and C.P. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 2 LLO synthesis and N-glycosylation in C. jejuni.
Red hexagon denotes di-N-acetylbacillosamine; yellow square denotes N-acetylgalactosamine; and blue circle denotes glucose. The LLO polyprenyl tail is shown in purple. A periplasmic polypeptide chain is shown in blue. PglH (red) is the glycosyltransferace used in the in vitro tLLO flipping assays. Und-PP-ase, undecaprenyl pyrophosphatase.
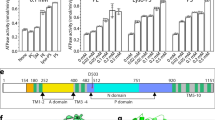
Extended Data Figure 3 Biochemical characterization of wild-type and mutant PglK.
a, In vitro tLLO flipping rates of PglK wild-type and E510Q mutant. Bars indicate initial velocities. b, In vitro tLLO flipping of PglK mutants E510Q, EHm1 and EHm2. +ADP, assay in the presence of 4 mM ADP; −ATP, assay in the absence of ATP. Liposomes, empty liposomes containing only tLLO. ADP alone does not cause tLLO to disappear from the external liposomes leaflet. c, In vivo LLO flipping of wild-type PglK and E510Q mutant expressed in E. coli SCM6 cells containing the C. jejuni pgl operon30. E.V., empty vector, N.I.C., non-induced cells. Anti-glycan refers to HR6 antibody recognizing the N-glycan of a substrate protein (see Methods), whereas anti-PglK is used to monitor PglK expression level. d, Determination of Km values of ATP hydrolysis and ATP-driven in vitro tLLO flipping. The black curve represents the ATPase rate of PglK at distinct ATP concentrations. The blue curve represents the initial LLO flipping rate in proteoliposomes at distinct ATP concentrations. Cpm, counts per million. e, ATPase activity in the presence (+) or absence (−) of native LLO of wild-type PglK and E510Q mutant in detergent (LMNG) or proteoliposomes. f, ATPase activity of PglK in detergent in the presence of LLO (20 μM), tLLO (20 μM), diverse drugs (50 μM) and lipid A (20 μM). g, Determination of Km values of ATP hydrolysis in the presence (+) or absence (−) of LLO (20 μM). Stimulation results in higher Vmax, while the Km for ATP remains almost unaltered in the presence (Km(+LLO) = 0.54 ± 0.03 mM) or absence (Km(−LLO) = 0.36 ± 0.04 mM) of native LLO. h, Determination of Km value of ATPase stimulation by native LLO in detergent (rates are normalized against the basal activity in absence of LLO). Error bars denote s.d. (n = 3).
Extended Data Figure 4 Electron density maps.
a, b, Stereo views (cross-eyed) of the 2Fo − Fc electron density maps for the complete PglK dimer of the structures at 2.9 and 3.9 Å, respectively. c, Stereo view of the non-crystallographic symmetry (NCS)-averaged electron density map for the complete PglK dimer of the structure at 5.9 Å and close-up of the NBDs showing the Fo − Fc electron density map for the bound ADP molecules. 2Fo − Fc and NCS maps are shown at 1.0σ level. Fo − Fc maps are shown at 3.0σ level.
Extended Data Figure 5 Validation of side-chain register of outward-occluded PglK model.
a–c, Anomalous electron density maps define selenomethionine (a), cysteine-bound mercury (b) and PtCl4 sites (c). Contour levels are between 4.0 to 5.0σ. In a, anomalous density was observed for 9 out of 10 selenomethionines of PglK.
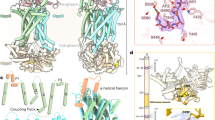
Extended Data Figure 6 Structural features of PglK.
a, Ribbon diagram of one PglK subunit depicting the secondary structure arrangement, based on the Sav1866 nomenclature36. b, Conformational changes of TM4 and TM5 visualized after superposition of subunits of apo-inward-1 (light orange) and apo-inward-2 (dark orange) structures. c, Structures of the antibacterial peptide ABC exporter McjD in occluded state (PDB code 4PLO), PglK in outward-occluded state, and the ABC exporter Sav1866 (PDB code 2HYD) in outward-open state. Transmembrane helices TM1 and TM3 (purple) define the extent of the external opening. Subunits in each dimer are shown in orange and grey. d, Side and cytoplasmic view cut-off of PglK apo-inward-1 structure and vacuum electrostatic surface representation showing the internal cavity.
Extended Data Figure 7 PglK crystal packing.
a–c, The main crystal contacts of apo-inward-1 (2.9 Å resolution) (a), apo-inward-2 (3.9 Å resolution) (b), and outward-occluded (5.9 Å resolution) (c) states.
Extended Data Figure 8 Putative interactions of native and synthetic LLO analogues with PglK in apo-inward and outward-occluded states.
a, b, Native LLO (a) and synthetic LLO analogues (b). Red hexagon denotes di-N-acetylbacillosamine; yellow square denotes N-acetylgalactosamine; blue circle denotes glucose; and blue square denotes N-acetylglucosamine. The LLO polyprenyl tail is shown in purple.
Extended Data Figure 9 Product analysis and control experiments of PglK-catalysed in vitro flipping assay.
a, Product analysis of tLLO glycosylation catalysed by PglH. The reaction products were analysed by in vitro glycosylation of a fluorescently labelled substrate peptide (DQNAT sequon) catalysed by PglB as reported earlier30,31. Depending on the presence and size of the N-glycan, peptides show a different mobility after Tricine–SDS–PAGE. Bands were visualized using a fluorescence gel scan (488 nm excitation and 526 nm emission). Lane 1, product of the deglycosylation reaction of hexasaccharide LLO catalysed by α-N-acetylgalactosaminidase, which removes terminal GalNAc molecules. This demonstrates the purity of the LLO used with respect to GalNAc. Lane 2, tLLO used in biochemical assays. Lane 3, products of tLLO glycosylation when the tLLO:GalNAc molar ratio is 2:1. Lane 4, products of tLLO glycosylation when the tLLO:GalNAc molar ratio is 1:1. Lane 5, products of tLLO glycosylation when the tLLO:GalNAc molar ratio is 1:10. b, PglK proteoliposomes incubated in the presence of 4 mM ADP. The level of tLLO in the external leaflet after t = 40 min remains unchanged (104.2 ± 10.8%) relative to the amount at t = 0. ADP alone does not cause a decrease in the concentration of tLLO in the external liposomes leaflet, ruling out a potential sequestration of tLLO in the outward-occludded state of PglK. c, Determination of tLLO orientation in proteoliposomes. Disrupted liposomes (0.3% Triton X-100) and non-disrupted proteoliposomes were incubated with PglH in the presence of excess GalNAc. The amount of glycopeptide was determined from band intensities of fluorescence gel scans. 48.2 ± 7.5% of tLLO is located in the outer leaflet of the bilayer. d, Determination of PglK orientation in proteoliposomes. The fully functional mutant PglK(C269L/C352S/C386S/C549L/S544C) was reconstituted in proteoliposomes and labelled with negatively charged Alexa Fluor 488 C5 maleimide. The fluorescence of non-disrupted and disrupted proteoliposomes was compared. 51.8 ± 2.8% of the PglK molecules are oriented with NBDs facing outwards. Red hexagon denotes di-N-acetylbacillosamine; yellow square denotes N-acetylgalactosamine.
Rights and permissions
About this article
Cite this article
Perez, C., Gerber, S., Boilevin, J. et al. Structure and mechanism of an active lipid-linked oligosaccharide flippase. Nature 524, 433–438 (2015). https://doi.org/10.1038/nature14953
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14953
This article is cited by
-
Structural insights into substrate recognition and translocation of human peroxisomal ABC transporter ALDP
Signal Transduction and Targeted Therapy (2023)
-
Substrate binding-induced conformational transitions in the omega-3 fatty acid transporter MFSD2A
Nature Communications (2023)
-
Energetics of lipid transport by the ABC transporter MsbA is lipid dependent
Communications Biology (2021)
-
Structural basis of omega-3 fatty acid transport across the blood–brain barrier
Nature (2021)
-
Structure of a proton-dependent lipid transporter involved in lipoteichoic acids biosynthesis
Nature Structural & Molecular Biology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.