Abstract
The eye is a complex organ with highly specialized constituent tissues derived from different primordial cell lineages. The retina, for example, develops from neuroectoderm via the optic vesicle, the corneal epithelium is descended from surface ectoderm, while the iris and collagen-rich stroma of the cornea have a neural crest origin. Recent work with pluripotent stem cells in culture has revealed a previously under-appreciated level of intrinsic cellular self-organization, with a focus on the retina and retinal cells1,2,3,4,5. Moreover, we and others have demonstrated the in vitro induction of a corneal epithelial cell phenotype from pluripotent stem cells6,7,8,9. These studies, however, have a single, tissue-specific focus and fail to reflect the complexity of whole eye development. Here we demonstrate the generation from human induced pluripotent stem cells of a self-formed ectodermal autonomous multi-zone (SEAM) of ocular cells. In some respects the concentric SEAM mimics whole-eye development because cell location within different zones is indicative of lineage, spanning the ocular surface ectoderm, lens, neuro-retina, and retinal pigment epithelium. It thus represents a promising resource for new and ongoing studies of ocular morphogenesis. The approach also has translational potential and to illustrate this we show that cells isolated from the ocular surface ectodermal zone of the SEAM can be sorted and expanded ex vivo to form a corneal epithelium that recovers function in an experimentally induced animal model of corneal blindness.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eiraku, M. et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472, 51–56 (2011)
Nakano, T. et al. Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell 10, 771–785 (2012)
Zhong, X. et al. Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nature Commun. 5, 4047 (2014)
Reichman, S. et al. From confluent human iPS cells to self-forming neural retina and retinal pigmented epithelium. Proc. Natl Acad. Sci. USA 111, 8518–8523 (2014)
Mellough, C. B. et al. IGF-1 signaling plays an important role in the formation of three-dimensional laminated neural retina and other ocular structures from human embryonic stem cells. Stem Cells 33, 2416–2430 (2015)
Hayashi, R. et al. Generation of corneal epithelial cells from induced pluripotent stem cells derived from human dermal fibroblast and corneal limbal epithelium. PLoS ONE 7, e45435 (2012)
Shalom-Feuerstein, R. et al. Pluripotent stem cell model reveals essential roles for miR-450b-5p and miR-184 in embryonic corneal lineage specification. Stem Cells 30, 898–909 (2012)
Ahmad, S. et al. Differentiation of human embryonic stem cells into corneal epithelial-like cells by in vitro replication of the corneal epithelial stem cell niche. Stem Cells 25, 1145–1155 (2007)
Brzeszczynska, J. et al. Differentiation and molecular profiling of human embryonic stem cell-derived corneal epithelial cells. Int. J. Mol. Med. 33, 1597–1606 (2014)
Lavker, R. M., Tseng, S. C. & Sun, T.-T. Corneal epithelial stem cells at the limbus: looking at some old problems from a new angle. Exp. Eye Res. 78, 433–446 (2004)
Liem, K. F. Jr, Tremml, G., Roelink, H. & Jessell, T. M. Dorsal differentiation of neural plate cells induced by BMP-mediated signals from epidermal ectoderm. Cell 82, 969–979 (1995)
McMahon, J. A. et al. Noggin-mediated antagonism of BMP signaling is required for growth and patterning of the neural tube and somite. Genes Dev. 12, 1438–1452 (1998)
Truong, T. T., Huynh, K., Nakatsu, M. N. & Deng, S. X. SSEA4 is a potential negative marker for the enrichment of human corneal epithelial stem/progenitor cells. Invest. Ophthalmol. Vis. Sci. 52, 6315–6320 (2011)
Estey, T., Piatigorsky, J., Lassen, N. & Vasiliou, V. ALDH3A1: a corneal crystallin with diverse functions. Exp. Eye Res. 84, 3–12 (2007)
Pearson, J. C., Lemons, D. & McGinnis, W. Modulating Hox gene functions during animal body patterning. Nature Rev. Genet. 6, 893–904 (2005)
Mallo, M. & Alonso, C. R. The regulation of Hox gene expression during animal development. Development 140, 3951–3963 (2013)
Krenzer, K. L. & Freddo, T. F. Cytokeratin expression in normal human bulbar conjunctiva obtained by impression cytology. Invest. Ophthalmol. Vis. Sci. 38, 142–152 (1997)
Nishida, K. et al. Functional bioengineered corneal epithelial sheet grafts from corneal stem cells expanded ex vivo on a temperature-responsive cell culture surface. Transplantation 77, 379–385 (2004)
Pellegrini, G. et al. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 349, 990–993 (1997)
Nakamura, T. et al. Transplantation of cultivated autologous oral mucosal epithelial cells in patients with severe ocular surface disorders. Br. J. Ophthalmol. 88, 1280–1284 (2004)
Nakagawa, M. et al. Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nature Biotechnol. 26, 101–106 (2008)
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007)
Nakagawa, M. et al. A novel efficient feeder-free culture system for the derivation of human induced pluripotent stem cells. Sci. Rep. 4, 3594 (2014)
Miyazaki, T. et al. Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells. Nature Commun. 3, 1236 (2012)
Kawasaki, H. et al. Generation of dopaminergic neurons and pigmented epithelia from primate ES cells by stromal cell-derived inducing activity. Proc. Natl Acad. Sci. USA 99, 1580–1585 (2002)
Miyashita, H. et al. Long-term maintenance of limbal epithelial progenitor cells using Rho kinase inhibitor and keratinocyte growth factor. Stem Cells Transl. Med. 2, 758–765 (2013)
Barrandon, Y. & Green, H. Three clonal types of keratinocyte with different capacities for multiplication. Proc. Natl Acad. Sci. USA 84, 2302–2306 (1987)
Hayashi, R. et al. N-Cadherin is expressed by putative stem/progenitor cells and melanocytes in the human limbal epithelial stem cell niche. Stem Cells 25, 289–296 (2007)
Acknowledgements
We thank K. Baba, Y. Oie, H. Takayanagi, S. Hara, Y. Yasukawa, J. Toga and M. Yagi of Osaka University and M. Nakagawa of Kyoto University for technical assistance and scientific discussions. This work was supported in part by the project for the realization of regenerative medicine of The Japan Agency for Medical Research and Development (AMED), The Japan Science and Technology Agency (JST) and The Ministry of Health, Labour, and Welfare of Japan and the Grants-in-Aid for Scientific Research from The Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Contributions
R.H., M.T. and K.N. designed the research; R.H, Y.I., R.K. and S.A. performed the in vitro experiments and acquired the data; Y.S., N.N., T.I. and T.S. performed animal experiments and acquired the data; K.S. provided reagents (LN511E8); R.H., Y.I. and R.K. analysed the data and wrote the respective methods and results; S.K., K.S. and A.J.Q. supervised the project; and R.H., M.T., A.J.Q. and K.N. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
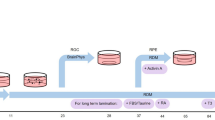
Extended Data Figure 1 Differentiation of multiple ocular cells in the SEAM.
a, A differentiated human iPS cell colony after 40 days of culture (macro photograph, representative of six independent experiments). Scale bar, 5 mm. b, Magnified views of each SEAM zone margin after six weeks in culture (phase-contrast and bright-field views, each representative of three independent experiments). Arrow heads indicate borders between each zone. Scale bars, 50 μm. c, Schematic for the development of the anterior eye. CE, corneal epithelium; CEnd, corneal endothelium; IS, iris stroma; NR, neuroretina; RPE, retinal pigment epithelium; LE, lens. d, Immunostaining for p75 (green) and SOX10 (red) in SEAM zones 1 and 2, two weeks (w) after the start of the differentiation culture (representative of three independent experiments). Asterisks indicate SOX10+/p75+ neural crest cells. Nuclei, blue. Scale bar, 100 μm. e, Immunostaining of lens differentiation marker α-crystallin (green) and epithelial marker p63 (red) in zones 2 and 3 of the SEAM after six weeks of culture (representative of three independent experiments). Nuclei, blue. Scale bar, 100 μm.
Extended Data Figure 2 Enrichment and differentiation of a SEAM-derived ocular surface epithelium.
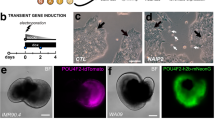
a, The enrichment of zone 3 ocular surface epithelium by manual pipetting. Cells in the SEAM before (upper panel) and after (lower panel) pipetting are shown (n = 1). Scale bar, 200 μm. b, The expression of ocular-cell-related genes in removed and attached cells after pipetting (n = 1). c, The time course of PAX6, DN-p63, K14, K12 and LIN28A expression during human iPS cell differentiation culture (0, 2, 4, 6, 8 and 12 weeks, each n = 5 independent experiments). Error bars are s.d. d, Immunostaining for vimentin (VIM, green) and p63 (red) before and after FACS purification conducted at 11 weeks of culture (representative of three independent experiments). VIM+ stroma-like cells of zone 3 were removed by FACS. Nuclei, blue. Scale bar, 50 μm. e, Immunostaining for anterior eye development-related markers Pax6 (green), p63 (red), K14 (green) and K12 (red) during mouse eye development (E9.5–18.5, each representative of three experiments). Nuclei, blue. Asterisks indicate p63-expressing cells. PCE, presumptive corneal epithelium; OSEpi, ocular surface epithelium; CE, corneal epithelium; CS, corneal stroma; LV, lens vesicle; LE, lens; AC, anterior chamber; EL, eyelid; OV, optic vesicle. Scale bar, 50 μm.
Extended Data Figure 3 The effect of BMP/TGFβ inhibitors on the development of ocular surface epithelium in the SEAM.
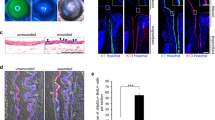
a, Microscopic observation of the SEAM pre-treated with Noggin, LDN-193189 (LDN) or SB-431542 (SB) for four days at the start of differentiation culture (control (−), Noggin and LDN data are representative of six independent experiments, while SB data are representative of four independent experiments). Both BMP and TGFβ inhibitors resulted in the abolishment of zone 3. Scale bar, 200 μm. b, The effects of BMP or TGFβ inhibitors on the expression of ocular ectoderm-related genes at five to six weeks of differentiation culture (*P < 0.05, control; n = 13: Noggin, LDN and SB; n = 8 independent experiments, respectively, Mann–Whitney test). Error bars are s.d.
Extended Data Figure 4 Isolation of corneal epithelial cells from the SEAM.
a, Schematic for the induction of ocular surface epithelium from human iPS cells. The ocular surface ectoderm expressing PAX6 and p63 in zone 3 of the SEAM further differentiated into functional ocular surface epithelial stem cells that expressed K14. Among the ocular surface epithelial lineage cells, corneal epithelial progenitor cells were isolated by FACS as SSEA-4+/ITGB4+ cells. Conjunctival epithelial cells (PAX6+/K13+/K12−) were obtained as SSEA-4− cells. NE, neuroectoderm; NR, neuroretina; NC, neural crest; OSE, ocular surface ectoderm; SE, surface ectoderm. b, Immunostaining for SSEA-4 (green) and K12 (red) in stratified epithelial tissues, including human ocular surface epithelium (cornea, limbus and conjunctiva), epidermis and oral mucosa. A magnified view of the limbus (highlighted box) indicated that SSEA-4 was expressed in all layers of the limbal epithelium, including the basal layer, which had no K12 (asterisks). No other stratified epithelium expressed SSEA-4 and K12. Nuclei, blue. Scale bars, 50 μm. (Data for corneal, limbal and conjunctival tissue; n = 3; epidermis and oral mucosa; n = 1). c, Immunostaining for PAX6 (green) in colonies derived from P3 and P2 cells (representative of five independent cell-sorting experiments). Nuclei, blue. Scale bar, 100 μm. d, The expression of corneal epithelial-specific genes and non-corneal epithelial genes in the sorted P3 cells (that is, human iPS cell-derived corneal epithelial cells, iCECs) and P2 cells. *P < 0.05 (n = 7 independent cell sorting experiments, Mann–Whitney test). Error bars are s.d.
Extended Data Figure 5 Characterization of SEAM-derived ocular surface epithelial cells.
a, Triple colour immunostaining for PAX6 (green), K12 (orange) and K13 (magenta) in the epithelial colonies from sorted P3 cells (that is, SEAM-derived human iCECs) and P2 cells (representative of five independent cell-sorting experiments). Nuclei, blue. Scale bars, 100 μm. b, HOX gene expression in the sorted P3 cells, P2 cells, human epidermal keratinocytes (EKs) and human corneal limbal epithelial cells (CECs). (P3 and P2 cells; n = 7: human EKs and human CECs; n = 5 independent experiments). Error bars are s.d. c, The PAX6 (green) and HOXB4 (red) expression in the colonies of P2 cells (n = 1). Nuclei, blue. Scale bar, 100 μm. d, Goblet-cell-like differentiation in the SEAM-derived epithelium after long-term culture (more than 12 weeks of differentiation) without FACS in CEM (n = 1). Goblet-cell-like morphology was observed in presumptive P2 cell regions (left panel). These cells were PAS-positive and expressed the goblet cell markers MUC5AC (green) and K7 (red) in the superficial region and PAX6 in the basal region. Nuclei, blue. Scale bars, 50 μm.
Extended Data Figure 6 Characterization of the SEAM-derived corneal epithelium.
a, Immunostaining for K19 and CX43 (green) in the stratified SEAM-derived human iCECs (representative of n = 3 independent experiments). Magnified view of the dotted area is shown in the lower panel. Nuclei, red. Scale bars, 50 μm. b, Scanning electron microscopy of the apical surface of the stratified human iCECs (representative of two human iCEC sheets) and human CECs (n = 1). Scale bars, 10 μm (upper panels) and 1 μm (lower panels). c, Results of a hierarchical cluster analysis based on the global gene expression as examined by microarrays. Data are shown for human iPS cells (n = 3 technical replicates), human iPS cell-derived ocular surface ectoderm (OSE; that is, human iPS cell-derived cells after six weeks of differentiation, n = 3 technical replicates), human iCECs (n = 4 independent experiments), human oral keratinocytes (OKs, n = 3 technical replicates), human dermal fibroblasts (DFs, n = 3 technical replicates) and human corneal epithelial cells obtained from the limbus (CECs, n = 3 independent experiments). A total of 25,262 significantly changed genes (fold change >2.0, false discovery rate <0.05) were analysed. d, SEAM-derived human iCECs at passage (P) 1, 3, 9 and 15 during serial passages (representative of three independent experiments). Scale bar, 200 μm. e, The G-band karyotype of human iCECs at P = 1, 6 and 12 (n = 1, respectively).
Extended Data Figure 7 Induction of corneal epithelial cells from different human iPS cell clones.
a, SEAM formation patterns of five different human iPS cell clones (201B7 and 1383D2; representative of three independent experiments: 253G1, 454E2 and 1231A3; n = 1). PBMC, peripheral blood mononuclear cell; TF, transcription factor. Scale bar, 200 μm. b, Stratified human iCECs from 454E2, 253G1, 1231A3 and 1383D2 clones in vitro (454E2, 253G1 and 1231A3, n = 1; 1383D2, representative of three independent experiments). Phase-contrast microscopy (upper panel), haematoxylin and eosin (H&E) staining (middle panel) and immunostaining for K12 (lower panel, green) are shown. Nuclei, red. Scale bars, 200 μm (phase-contrast), 50 μm (H&E staining and immunostaining). c, Efficiency of corneal epithelial differentiation of the various human iPS cell clones (201B7, n = 23; 253G1, n = 5; 454E2, n = 11; 1231A3, n = 9 and 1383D2, n = 6 independent experiments). Error bars are s.d.
Extended Data Figure 8 Transplantation of the human iCEC sheet to repair the ocular surface.
a, Phase-contrast microscopy of SEAM-derived human iCECs cultivated on a temperature-responsive dish at 1, 5 and 14 days (representative of three independent experiments). A harvested human iCEC sheet is shown in the lower right panel. Scale bar, 200 μm (phase-contrast), 5 mm (macro photo). b, Immunostaining for PAX6 (green), K12 (orange) and K14 (magenta) in the human iCEC sheets after 21 days in culture (left panels, representative of three human iCEC sheets). The right panel shows an image used for a colony-forming assay (CFA) for the human iCEC sheets (15,000 cells per well, representative of eight independent experiments). The colony-forming efficiency (CFE) was 1.04 ± 0.43% (s.d., n = 8 independent experiments). Nuclei, blue. Scale bars, 100 μm (immunostaining) and 5 mm (CFA). c, Treatment of rabbit ocular surface with a surgical swab soaked in 99% ethanol for 30–60 s. d, e, Elimination of corneal epithelial stem cells by the surgical removal of corneal and limbal epithelial tissue (that is, lamellar keratectomy). f, The ocular surface was invaded by conjunctival tissue and vessels at postoperative day 28. g, Removal of the conjunctival tissue that covered the ocular surface. h, Widespread fluorescein staining of the ocular surface after surgical removal of epithelial tissue. i, The harvest of a human iCEC sheet from a temperature-responsive dish after lowering the temperature. j, Transplantation of the human iCEC sheet onto the rabbit cornea with a surgically induced corneal epithelial stem-cell deficiency treated as described in c–h. k, H&E staining for human iCEC-sheet-transplanted and control corneas on postoperative day 14 (n = 6 animal transplantation experiments). Scale bar, 100 μm.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-3. (PDF 212 kb)
Differentiating hiPSCs forming a SEAM
A hiPSC colony shown throughout the first 25 days of culture in differentiation medium spontaneously generating a concentric SEAM and its multiple cellular zones. (MP4 11491 kb)
Rights and permissions
About this article
Cite this article
Hayashi, R., Ishikawa, Y., Sasamoto, Y. et al. Co-ordinated ocular development from human iPS cells and recovery of corneal function. Nature 531, 376–380 (2016). https://doi.org/10.1038/nature17000
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17000
This article is cited by
-
Ocular instillation of conditioned medium from mesenchymal stem cells is effective for dry eye syndrome by improving corneal barrier function
Scientific Reports (2023)
-
Induced pluripotent stem cell–derived extracellular vesicles overexpressing SFPQ protect retinal Müller cells against hypoxia-induced injury
Cell Biology and Toxicology (2023)
-
Application of Human Stem Cell Derived Retinal Organoids in the Exploration of the Mechanisms of Early Retinal Development
Stem Cell Reviews and Reports (2023)
-
Advances in corneal regenerative medicine with iPS cells
Japanese Journal of Ophthalmology (2023)
-
The Potential of Stem Cells as Treatment for Ocular Surface Diseases
Current Ophthalmology Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.