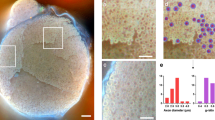
Abstract
Nerve preservation is an important goal during surgery because accidental transection or injury leads to significant morbidity, including numbness, pain, weakness or paralysis. Nerves are usually identified by their appearance and relationship to nearby structures or detected by local electrical stimulation (electromyography), but thin or buried nerves are sometimes overlooked. Here, we use phage display to select a peptide that binds preferentially to nerves. After systemic injection of a fluorescently labeled version of the peptide in mice, all peripheral nerves are clearly delineated within 2 h. Contrast between nerve and adjacent tissue is up to tenfold, and useful contrast lasts up to 8 h. No changes in behavior or activity are observed after treatment, indicating a lack of obvious toxicity. The fluorescent probe also labels nerves in human tissue samples. Fluorescence highlighting is independent of axonal integrity, suggesting that the probe could facilitate surgical repair of injured nerves and help prevent accidental transection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gantz, B.J. Intraoperative facial nerve monitoring. Am. J. Otol. Nov., (Suppl.), 58–61 (1985).
Davis, W.E., Rea, J.L. & Templer, J. Recurrent laryngeal nerve localization using a microlaryngeal electrode. Otolaryngol. Head Neck Surg. 87, 330–333 (1979).
Miller, M.C. & Spiegel, J.R. Identification and monitoring of the recurrent laryngeal nerve during thyroidectomy. Surg. Oncol. Clin. N. Am. 17, 121–144 (2008).
Walz, J., Graefen, M. & Huland, H. Basic principles of anatomy for optimal surgical treatment of prostate cancer. World J. Urol. 25, 31–38 (2007).
Walz, J. et al. A critical analysis of the current knowledge of surgical anatomy related to optimization of cancer control and preservation of continence and erection in candidates for radical prostatectomy. Eur. Urol. 57, 179–192 (2010).
Kübler, H.R. et al. Impact of nerve sparing technique on patient self-assessed outcomes after radical perineal prostatectomy. J. Urol. 178, 488–492 (2007).
Zhivov, A., Blum, M., Guthoff, R. & Stachs, O. Real-time mapping of the subepithelial nerve plexus by in vivo confocal laser scanning microscopy. Br. J. Ophthalmol. 94, 1133–1135 (2010).
Zysk, A.M., Nguyen, F.T., Oldenburg, A.L., Marks, D.L. & Boppart, S.A. Optical coherence tomography: a review of clinical development from bench to bedside. J. Biomed. Opt. 12, 051403 (2007).
Kobbert, C. et al. Currents concepts in neuroanatomical tracing. Prog. Neurobiol. 62, 327–351 (2000).
Richmond, F.J.R. et al. Efficacy of seven retrograde tracers, compared in multiple-labelling studies of feline motoneurones. J. Neurosci. Methods 53, 35–46 (1994).
Marangos, N., Illing, R., Kruger, J. & Laszig, R. In vivo visualization of the cochlear nerve and nuclei with fluorescent axonal tracers. Hear. Res. 162, 48–52 (2001).
O'Malley, M. et al. Fluorescent retrograde axonal tracing of the facial nerve. Laryngoscope 116, 1792–1797 (2006).
Pasqualini, R. & Ruoslahti, E. Organ targeting in vivo using phage display peptide libraries. Nature 380, 364–366 (1996).
Shtatland, T., Buettler, D., Kossodo, M., Pivovarov, M. & Weissleder, R. PepBank—a database of peptides based on sequence text mining and public peptide data sources. BMC Bioinformatics 8, 280 (2007).
Berger, S., Bannantine, J.P. & Griffin, J.F.T. Autoreactive antibodies are present in sheep with Johne's disease and cross-react with mycobacterium avium subsp. Paratuberculosis antigens. Microbes Infect. 9, 963–970 (2007).
Kim, G.S. et al. Suppression of receptor-mediated apoptosis by death effecter domain recruiting domain binding peptide aptamer. Biochem. Biophys. Res. Commun. 343, 1165–1170 (2006).
Jiang, Y. et al. Targeting of hepatoma cell and suppression of tumor growth by a novel 12mer peptide fused to superantigen TSST-1. Mol. Med. 12, 81–87 (2006).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Olson, E.S. et al. Activatable cell penetrating peptides attached to nanoparticles: dual probes for fluorescence and magnetic resonance imaging of proteases in vivo. Proc. Natl. Acad. Sci. USA 107, 4311–4316 (2010).
Nguyen, Q.T. et al. Surgery with molecular fluorescence imaging using activatable cell penetrating peptides decreases residual cancer and improves survival. Proc. Natl. Acad. Sci. USA 107, 4317–4322 (2010).
Osuchowski, M.F., Teener, J. & Remick, D. Noninvasive model of sciatic nerve conduction in healthy and septic mice: reliability and normative data. Muscle Nerve 40, 610–616 (2009).
Acknowledgements
We are indebted to members of our laboratory for discussions and comments on the manuscript. Results described here are being used in support of a patent filing by the University of California, San Diego. This work was supported by the Howard Hughes Medical Institute, grants from the Burrough-Wellcome Fund (Career Award for Medical Scientists) and National Institutes of Health (NIH, 5K08EB008122) to Q.T.N. and NIH grant NS27177 to R.Y.T.
Author information
Authors and Affiliations
Contributions
M.A.W. designed and performed experiments, interpreted data and wrote manuscript. J.L.C. designed and performed experiments and interpreted data. L.T.N. designed and performed experiments and interpreted data. B.F. designed and performed experiments and interpreted data. L.A.G. designed and performed experiments and interpreted data. P.S. provided computer support for experiments, R.Y.T. designed experiments, interpreted data and wrote manuscript. Q.T.N. designed and performed experiments, interpreted data and wrote manuscript.
Corresponding author
Ethics declarations
Competing interests
M.A.W., R.Y.T. and Q.T.N. are scientific advisors for Avelas Biosciences, Inc., who has licensed the technology described above from the University of California San Diego.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1, Supplementary Figs. 1–8 and Supplementary Video 1 (PDF 12353 kb)
Rights and permissions
About this article
Cite this article
Whitney, M., Crisp, J., Nguyen, L. et al. Fluorescent peptides highlight peripheral nerves during surgery in mice. Nat Biotechnol 29, 352–356 (2011). https://doi.org/10.1038/nbt.1764
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1764
This article is cited by
-
Label-free intraoperative nerve detection and visualization using ratiometric diffuse reflectance spectroscopy
Scientific Reports (2023)
-
Nerve autofluorescence in near-ultraviolet light markedly enhances nerve visualization in vivo
Surgical Endoscopy (2022)
-
Near-infrared intraoperative imaging of pelvic autonomic nerves: a pilot study
Surgical Endoscopy (2022)
-
Nerve autofluorescence under near-ultraviolet light: cutting-edge technology for intra-operative neural tissue visualization in 17 patients
Surgical Endoscopy (2022)
-
Molecular imaging and disease theranostics with renal-clearable optical agents
Nature Reviews Materials (2021)