Abstract
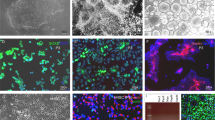
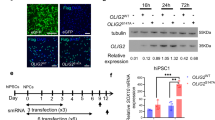
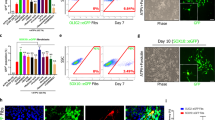
Transplantation of oligodendrocyte precursor cells (OPCs) is a promising potential therapeutic strategy for diseases affecting myelin. However, the derivation of engraftable OPCs from human pluripotent stem cells has proven difficult and primary OPCs are not readily available. Here we report the generation of induced OPCs (iOPCs) by direct lineage conversion. Forced expression of the three transcription factors Sox10, Olig2 and Zfp536 was sufficient to reprogram mouse and rat fibroblasts into iOPCs with morphologies and gene expression signatures resembling primary OPCs. More importantly, iOPCs gave rise to mature oligodendrocytes that could ensheath multiple host axons when co-cultured with primary dorsal root ganglion cells and formed myelin after transplantation into shiverer mice. We propose direct lineage reprogramming as a viable alternative approach for the generation of OPCs for use in disease modeling and regenerative medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Potter, G.B., Rowitch, D.H. & Petryniak, M.A. Myelin restoration: progress and prospects for human cell replacement therapies. Arch. Immunol. Ther. Exp. (Warsz.) 59, 179–193 (2011).
Franklin, R.J. & Ffrench-Constant, C. Remyelination in the CNS: from biology to therapy. Nat. Rev. Neurosci. 9, 839–855 (2008).
Goldman, S.A. Progenitor cell-based treatment of the pediatric myelin disorders. Arch. Neurol. 68, 848–856 (2011).
Cummings, B.J. et al. Human neural stem cells differentiate and promote locomotor recovery in spinal cord-injured mice. Proc. Natl. Acad. Sci. USA 102, 14069–14074 (2005).
Franklin, R.J. & Ffrench-Constant, C. Stem cell treatments and multiple sclerosis. Br. Med. J. 340, c1387 (2010).
Brustle, O. et al. Embryonic stem cell-derived glial precursors: a source of myelinating transplants. Science 285, 754–756 (1999).
Learish, R.D., Brustle, O., Zhang, S.C. & Duncan, I.D. Intraventricular transplantation of oligodendrocyte progenitors into a fetal myelin mutant results in widespread formation of myelin. Ann. Neurol. 46, 716–722 (1999).
Sim, F.J. et al. CD140a identifies a population of highly myelinogenic, migration-competent and efficiently engrafting human oligodendrocyte progenitor cells. Nat. Biotechnol. 29, 934–941 (2011).
Windrem, M.S. et al. Fetal and adult human oligodendrocyte progenitor cell isolates myelinate the congenitally dysmyelinated brain. Nat. Med. 10, 93–97 (2004).
Windrem, M.S. et al. Neonatal chimerization with human glial progenitor cells can both remyelinate and rescue the otherwise lethally hypomyelinated shiverer mouse. Cell Stem Cell 2, 553–565 (2008).
Wang, S. et al. Human iPSC-derived oligodendrocyte progenitor cells can myelinate and rescue a mouse model of congenital hypomyelination. Cell Stem Cell 12, 252–264 (2013).
Keirstead, H.S. et al. Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants remyelinate and restore locomotion after spinal cord injury. J. Neurosci. 25, 4694–4705 (2005).
Najm, F.J. et al. Rapid and robust generation of functional oligodendrocyte progenitor cells from epiblast stem cells. Nat. Methods 8, 957–962 (2011).
Vierbuchen, T. et al. Direct conversion of fibroblasts to functional neurons by defined factors. Nature 463, 1035–1041 (2010).
Pang, Z.P. et al. Induction of human neuronal cells by defined transcription factors. Nature 476, 220–223 (2011).
Caiazzo, M. et al. Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature 476, 224–227 (2011).
Pfisterer, U. et al. Direct conversion of human fibroblasts to dopaminergic neurons. Proc. Natl. Acad. Sci. USA 108, 10343–10348 (2011).
Son, E.Y. et al. Conversion of mouse and human fibroblasts into functional spinal motor neurons. Cell Stem Cell 9, 205–218 (2011).
Kim, J. et al. Direct reprogramming of mouse fibroblasts to neural progenitors. Proc. Natl. Acad. Sci. USA 108, 7838–7843 (2011).
Lujan, E., Chanda, S., Ahlenius, H., Sudhof, T.C. & Wernig, M. Direct conversion of mouse fibroblasts to self-renewing, tripotent neural precursor cells. Proc. Natl. Acad. Sci. USA 109, 2527–2532 (2012).
Thier, M. et al. Direct conversion of fibroblasts into stably expandable neural stem cells. Cell Stem Cell 10, 473–479 (2012).
Han, D.W. et al. Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell 10, 465–472 (2012).
Matsui, T. et al. Neural stem cells directly differentiated from partially reprogrammed fibroblasts rapidly acquire gliogenic competency. Stem Cells 30, 1109–1119 (2012).
Ring, K.L. et al. Direct reprogramming of mouse and human fibroblasts into multipotent neural stem cells with a single factor. Cell Stem Cell 11, 100–109 (2012).
Cahoy, J.D. et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J. Neurosci. 28, 264–278 (2008).
Dugas, J.C., Tai, Y.C., Speed, T.P., Ngai, J. & Barres, B.A. Functional genomic analysis of oligodendrocyte differentiation. J. Neurosci. 26, 10967–10983 (2006).
Mallon, B.S., Shick, H.E., Kidd, G.J. & Macklin, W.B. Proteolipid promoter activity distinguishes two populations of NG2-positive cells throughout neonatal cortical development. J. Neurosci. 22, 876–885 (2002).
Barres, B.A. et al. Cell death and control of cell survival in the oligodendrocyte lineage. Cell 70, 31–46 (1992).
Chan, J.R. et al. NGF controls axonal receptivity to myelination by Schwann cells or oligodendrocytes. Neuron 43, 183–191 (2004).
Roach, A., Boylan, K., Horvath, S., Prusiner, S.B. & Hood, L.E. Characterization of cloned cDNA representing rat myelin basic protein: absence of expression in brain of shiverer mutant mice. Cell 34, 799–806 (1983).
Lazzarini, R.A. Myelin Biology and Disorders Vol. 2 (Academic Press, 2004).
Meissner, A., Wernig, M. & Jaenisch, R. Direct reprogramming of genetically unmodified fibroblasts into pluripotent stem cells. Nat. Biotechnol. 25, 1177–1181 (2007).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Wernig, M. et al. A drug-inducible transgenic system for direct reprogramming of multiple somatic cell types. Nat. Biotechnol. 26, 916–924 (2008).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Yoo, A.S. et al. MicroRNA-mediated conversion of human fibroblasts to neurons. Nature 476, 228–231 (2011).
Eisen, M.B., Spellman, P.T., Brown, P.O. & Botstein, D. Cluster analysis and display of genome-wide expression patterns. Proc. Natl. Acad. Sci. USA 95, 14863–14868 (1998).
Chen, Y. et al. NS21: re-defined and modified supplement B27 for neuronal cultures. J. Neurosci. Methods 171, 239–247 (2008).
Acknowledgements
We thank W. Macklin for sharing the Plp-EGFP mouse line and B. Stallcup for providing the rabbit NG2 antibody. We are extremely grateful to N. Uchida for crucial advice and would also like to thank A. Olson for help with the confocal analysis and the Neuroscience Institute Imaging facility and the Stanford genomics core for microarray hybridization. This work was supported by the Ellison Medical Foundation (M.W.), the Stinehart-Reed Foundation (M.W.), the National Institutes of Health grants R01MH092931 (M.W.), the New York Stem Cell Foundation (M.W.) and R01EY10257 (B.A.B). N.Y. is the recipient of a Walter V. and Idun Berry Fellowship. J.B.Z. is a Howard Hughes Medical Institute Fellow of the Life Sciences Research Foundation. H.A. is supported by a postdoctoral fellowship from the Swedish Research Council and the Swedish Society for Medical Research. M.W. is a New York Stem Cell Foundation–Robertson Investigator.
Author information
Authors and Affiliations
Contributions
N.Y., J.B.Z., H.A. and M.W. designed research; N.Y., J.B.Z., H.A., S.M., Y.H.N., T.V., J.S.H. and R.G. performed research; N.Y., J.B.Z., H.A., S.M., B.A.B. and M.W. analyzed data; and N.Y., J.B.Z., H.A. and M.W. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–3 (PDF 9473 kb)
Rights and permissions
About this article
Cite this article
Yang, N., Zuchero, J., Ahlenius, H. et al. Generation of oligodendroglial cells by direct lineage conversion. Nat Biotechnol 31, 434–439 (2013). https://doi.org/10.1038/nbt.2564
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2564
This article is cited by
-
Cell replacement therapy with stem cells in multiple sclerosis, a systematic review
Human Cell (2023)
-
EGF signaling promotes the lineage conversion of astrocytes into oligodendrocytes
Molecular Medicine (2022)
-
OCT4-induced oligodendrocyte progenitor cells promote remyelination and ameliorate disease
npj Regenerative Medicine (2022)
-
scRNA-seq generates a molecular map of emerging cell subtypes after sciatic nerve injury in rats
Communications Biology (2022)
-
OLIG2 regulates lncRNAs and its own expression during oligodendrocyte lineage formation
BMC Biology (2021)