Abstract
Technical variation in metagenomic analysis must be minimized to confidently assess the contributions of microbiota to human health. Here we tested 21 representative DNA extraction protocols on the same fecal samples and quantified differences in observed microbial community composition. We compared them with differences due to library preparation and sample storage, which we contrasted with observed biological variation within the same specimen or within an individual over time. We found that DNA extraction had the largest effect on the outcome of metagenomic analysis. To rank DNA extraction protocols, we considered resulting DNA quantity and quality, and we ascertained biases in estimates of community diversity and the ratio between Gram-positive and Gram-negative bacteria. We recommend a standardized DNA extraction method for human fecal samples, for which transferability across labs was established and which was further benchmarked using a mock community of known composition. Its adoption will improve comparability of human gut microbiome studies and facilitate meta-analyses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Accession codes
References
Meyer, F. et al. The metagenomics RAST server - a public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinformatics 9, 386 (2008).
Larsen, N. et al. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS One 5, e9085 (2010).
Qin, J. et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 (2012).
Forslund, K. et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 528, 262–266 (2015).
Manichanh, C. et al. Reduced diversity of faecal microbiota in Crohn's disease revealed by a metagenomic approach. Gut 55, 205–211 (2006).
Carroll, I.M. et al. Molecular analysis of the luminal- and mucosal-associated intestinal microbiota in diarrhea-predominant irritable bowel syndrome. Am. J. Physiol. Gastrointest. Liver Physiol. 301, G799–G807 (2011).
Zeller, G. et al. Potential of fecal microbiota for early-stage detection of colorectal cancer. Mol. Syst. Biol. 10, 766 (2014).
Dethlefsen, L., McFall-Ngai, M. & Relman, D.A. An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 449, 811–818 (2007).
Dominguez-Bello, M.G. et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 107, 11971–11975 (2010).
Yatsunenko, T. et al. Human gut microbiome viewed across age and geography. Nature 486, 222–227 (2012).
Le Chatelier, E. et al. Richness of human gut microbiome correlates with metabolic markers. Nature 500, 541–546 (2013).
Wesolowska-Andersen, A. et al. Choice of bacterial DNA extraction method from fecal material influences community structure as evaluated by metagenomic analysis. Microbiome 2, 19 (2014).
McOrist, A.L., Jackson, M. & Bird, A.R. A comparison of five methods for extraction of bacterial DNA from human faecal samples. J. Microbiol. Methods 50, 131–139 (2002).
Smith, B., Li, N., Andersen, A.S., Slotved, H.C. & Krogfelt, K.A. Optimising bacterial DNA extraction from faecal samples: comparison of three methods. Open Microbiol. J. 5, 14–17 (2011).
Maukonen, J., Simões, C. & Saarela, M. The currently used commercial DNA-extraction methods give different results of clostridial and actinobacterial populations derived from human fecal samples. FEMS Microbiol. Ecol. 79, 697–708 (2012).
Kennedy, N.A. et al. The impact of different DNA extraction kits and laboratories upon the assessment of human gut microbiota composition by 16S rRNA gene sequencing. PLoS One 9, e88982 (2014).
Salonen, A. et al. Comparative analysis of fecal DNA extraction methods with phylogenetic microarray: effective recovery of bacterial and archaeal DNA using mechanical cell lysis. J. Microbiol. Methods 81, 127–134 (2010).
Ariefdjohan, M.W., Savaiano, D.A. & Nakatsu, C.H. Comparison of DNA extraction kits for PCR-DGGE analysis of human intestinal microbial communities from fecal specimens. Nutr. J. 9, 23 (2010).
Sunagawa, S. et al. Metagenomic species profiling using universal phylogenetic marker genes. Nat. Methods 10, 1196–1199 (2013).
Manichanh, C., Borruel, N., Casellas, F. & Guarner, F. The gut microbiota in IBD. Nat. Rev. Gastroenterol. Hepatol. 9, 599–608 (2012).
Lozupone, C.A. et al. Meta-analyses of studies of the human microbiota. Genome Res. 23, 1704–1714 (2013).
Raes, J. & Bork, P. Molecular eco-systems biology: towards an understanding of community function. Nat. Rev. Microbiol. 6, 693–699 (2008).
Voigt, A.Y. et al. Temporal and technical variability of human gut metagenomes. Genome Biol. 16, 73 (2015).
Franzosa, E.A. et al. Relating the metatranscriptome and metagenome of the human gut. Proc. Natl. Acad. Sci. USA 111, E2329–E2338 (2014).
Song, S.J. et al. Preservation methods differ in fecal microbiome stability, affecting suitability for field studies. mSystems https://dx.doi.org/10.1128/mSystems.00021-16 (2016).
Gohl, D.M. et al. Systematic improvement of amplicon marker gene methods for increased accuracy in microbiome studies. Nat. Biotechnol. 34, 942–949 (2016).
Claassen, S. et al. A comparison of the efficiency of five different commercial DNA extraction kits for extraction of DNA from faecal samples. J. Microbiol. Methods 94, 103–110 (2013).
Yuan, S., Cohen, D.B., Ravel, J., Abdo, Z. & Forney, L.J. Evaluation of methods for the extraction and purification of DNA from the human microbiome. PLoS One 7, e33865 (2012).
Kultima, J.R. et al. MOCAT: a metagenomics assembly and gene prediction toolkit. PLoS One 7, e47656 (2012).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Huttenhower, C. et al. Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Franzosa, E.A. et al. Identifying personal microbiomes using metagenomic codes. Proc. Natl. Acad. Sci. USA 112, E2930–E2938 (2015).
Powell, S. et al. eggNOG v3.0: orthologous groups covering 1133 organisms at 41 different taxonomic ranges. Nucleic Acids Res. 40, D284–D289 (2012).
Lozupone, C.A., Stombaugh, J.I., Gordon, J.I., Jansson, J.K. & Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 489, 220–230 (2012).
Santiago, A. et al. Processing faecal samples: a step forward for standards in microbial community analysis. BMC Microbiol. 14, 112 (2014).
InhibitEx Tablets - QIAGEN Online Shop. Available at: https://www.qiagen.com/fr/shop/lab-basics/buffers-and-reagents/inhibitex-tablets/.
Henderson, G. et al. Effect of DNA extraction methods and sampling techniques on the apparent structure of cow and sheep rumen microbial communities. PLoS One 8, e74787 (2013).
Jones, M.B. et al. Library preparation methodology can influence genomic and functional predictions in human microbiome research. Proc. Natl. Acad. Sci. USA 112, 14024–14029 (2015).
Salter, S.J. et al. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 12, 87 (2014).
Acknowledgements
We thank S. Burz and K. Weizer for editing and web-posting the SOPs. We thank D. Ordonez and N.P. Gabrielli Lopez for advice on flow cytometry, which was provided by the Flow Cytometry Core Facility, EMBL. This study was funded by the European Community's Seventh Framework Programme via International Human Microbiome Standards (HEALTH-F4-2010-261376) grant. We also received support from Scottish Government Rural and Environmental Science and Analytical Services as well as from EMBL.
Author information
Authors and Affiliations
Contributions
P.I.C., S.S. and G.Z. analyzed data and drafted and finalized the manuscript. E.P. and A.A. analyzed data, sequenced samples and wrote the manuscript. F.L., J.R.K., M.R.H., L.P.C. and E.A.-V. analyzed data and wrote the manuscript. M.T., M. Driessen, R.H., F.-E.J. and K.R.P. created and quantified the mock community. M.B., J.R.M.B., L.B., T.C., S.C.-P., M. Derrien, A.D., M. Daigneault, R.A.L., W.M.d.V., B.B.F., H.J.F., F.G., M.H., H.H., J.v.H.V., J.J., I.K., P.L., E.L.C., V.M., C. Manichanh, J.C.M., C. Mery, H.M., C.O., P.W.O., J.P., S.P., N.P., M.P., A.S., D.S., K.P.S., B.S., K.S., P.V., J.V., L.Z. and E.G.Z. extracted samples and wrote the manuscript. S.D.E., J.D. and P.B. designed the study and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Inter-individual distance dependence on study.
Similar to Figure 3, we show the estimated effect sizes of different parameters in the context of inter-individual distance assessed within the different studies used. It is clear that while small, there are clear differences in the median distance within studies, with HMP samples appearing to be more homogenous that MetaHIT ones.
Supplementary Figure 2 Extraction bias across the two samples.
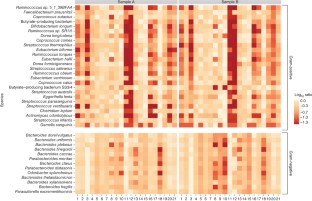
Extraction bias is consistent across the two samples, independent of the distance measure that was used. (a) shows a PCoA projection of the species abundances for each sample, independently, using a Spearman ranked correlation as well as a Euclidean distance. Most of the variation is captured by the first two principal coordinates and the clustering of extraction methods is easily observable. (b) shows a PCoA projection of the functional distance, both Spearman ranked and Euclidean.
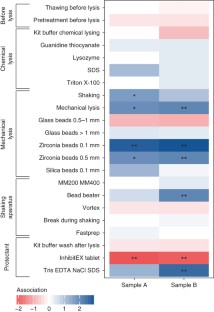
Supplementary Figure 3 Lysis of Gram-positive bacteria positively correlates with Shannon diversity.
Recovery of Gram-positive bacteria correlates with overall Shannon diversity. Considering only the top 20 most abundant species within each sample, ratios were computed between all Gram-positive and Gram-negative bacteria as well as Gram-negative to Gram-negative bacteria. The top panel shows the correlation of these ratios with the Shannon diversity index, while the lower panel exemplifies this correlation on the most abundant Gram-positive and Gram-negative bacteria that are common to both samples A and B, indicating the strong positive relation between recovery of Gram-positive bacteria and observed Shannon diversity.
Supplementary Figure 4 Shannon diversity of sample composition.
Observed Shannon diversity is consistently influenced by extraction method, as illustrated in both samples. Furthermore, there is a considerable difference in diversity between the two samples, which is not overwritten by extraction bias.
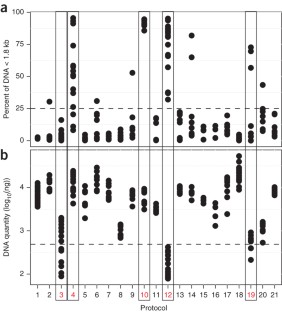
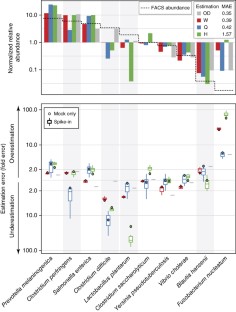
Supplementary Figure 5 Extraction bias of best performing protocols considered in Phase II.
Extraction variation is the same in Phase II replicates as that of Phase I (bars 1 and 2, respectively). Furthermore, the three protocols that have been merged into protocol Q for Phase II, namely 6, 9 and 15 produce similar results and present extraction bias below the biological replicate variation. The tree Phase II protocols (H, W and Q), when applied in different laboratories, with no previous experience in the particular protocol used, produce comparable abundance estimates, with errors below the level of biological variation within one specimen.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 693 kb)
Supplementary Methods
Supplementary Methods (PDF 1766 kb)
Supplementary Data 1
Protocol descriptors (XLSX 19 kb)
Supplementary Data 2
Members and composition of mock community (XLSX 13 kb)
Supplementary Data 3
Sample description (XLSX 15 kb)
Rights and permissions
About this article
Cite this article
Costea, P., Zeller, G., Sunagawa, S. et al. Towards standards for human fecal sample processing in metagenomic studies. Nat Biotechnol 35, 1069–1076 (2017). https://doi.org/10.1038/nbt.3960
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3960
This article is cited by
-
Gut microbial ecology and exposome of a healthy Pakistani cohort
Gut Pathogens (2024)
-
Microbial-derived imidazole propionate links the heart failure-associated microbiome alterations to disease severity
Genome Medicine (2024)
-
Choice of DNA extraction method affects stool microbiome recovery and subsequent phenotypic association analyses
Scientific Reports (2024)
-
A gut microbial signature for combination immune checkpoint blockade across cancer types
Nature Medicine (2024)
-
Comparison of DNA extraction methods on different sample matrices within the same terrestrial ecosystem
Scientific Reports (2024)



