Abstract
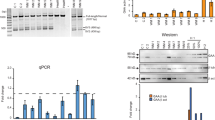
Antisense oligonucleotides are designed to specifically hybridize to a target messenger RNA (mRNA) and interfere with the synthesis of the encoded protein. Uniformly modified oligonucleotides containing N3′–P5′ phosphoramidate linkages exhibit (NP) extremely high-affinity binding to single-stranded RNA, do not induce RNase H activity, and are resistant to cellular nucleases. In the present work, we demonstrate that phosphoramidate oligonucleotides are effective at inhibiting gene expression at the mRNA level, by binding to their complementary target present in the 5′-untranslated region. Their mechanism of action was demonstrated by comparative analysis of three expression systems that differ only by the composition of the oligonucleotide target sequence (HIV-1 polypurine tract or PPT sequence) present just upstream from the AUG codon of the firefly luciferase reporter gene: the experiments have been done on isolated cells using oligonucleotide delivery mediated by cationic molecules or streptolysin O (SLO), and in vivo by oligonucleotide electrotransfer to skeletal muscle. In our experimental system phosphoramidate oligonucleotides act as potent and specific antisense agents by steric blocking of translation initiation; they may prove useful to modulate RNA metabolism while maintaining RNA integrity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lewis, J.G. et al. A serum-resistant cytofectin for cellular delivery of antisense oligodeoxynucleotides and plasmid DNA. Proc. Natl. Acad. Sci. USA 93, 3176–3181 ( 1996).
Flanagan, W.M. et al. A cytosine analog that confers enhanced potency to antisense oligonucleotides. Proc. Natl. Acad. Sci. USA 96, 3513–3518 (1999).
Baker, B.F. et al. 2′-O-(2-Methoxy)ethyl-modified anti-intercellular adhesion molecule 1 (ICAM-1) oligonucleotides selectively increase the ICAM-1 mRNA level and inhibit formation of the ICAM-1 translation initiation complex in human umbilical vein endothelial cells. J. Biol. Chem. 272, 11994–12000 (1997).
Iyer, R.P., Roland, A., Zhou, W. & Ghosh, K. Modified oligonucleotides-synthesis, properties and applications. Curr. Opin. Mol. Ther. 1, 344–358 (1999).
Gryaznov, S.M. & Chen, J.K. Oligodeoxyribonucleotide N3′-P5′ phosphoramidates—synthesis and hybridization properties . J. Am. Chem. Soc. 116, 3143– 3144 (1994).
Skorski, T., Perrotti, D., Nieborowska-Skorska, M., Gryaznov, S. & Calabretta, B. Antileukemia effect of c-myc N3′-P5′ phosphoramidate antisense oligonucleotides in vivo. Proc. Natl. Acad. Sci. USA 94, 3966–3971 (1997).
Gryaznov, S.M. et al. Oligonucleotide N3′–P5′ phosphoramidates as antisense agents. Nucleic Acids Res. 24, 1508–1514 (1996).
Heidenreich, O., Gryaznov, S. & Nerenberg, M. RNase H-independent antisense activity of oligonucleotide N3′–P5′ phosphoramidates. Nucleic Acids Res. 25, 776–780 (1997).
Charneau, P. et al. HIV-1 reverse transcription. A termination step at the center of the genome. J. Mol. Biol. 241, 651– 662 (1994).
Zennou, V. et al. HIV-1 genome nuclear import is mediated by a central DNA flap . Cell 101, 173–185 (2000).
Giovannangeli, C. & Hélène, C. Triplex-forming molecules for modulation of DNA information processing. Curr. Opin. Mol. Ther. 2, 288–297 (2000).
Faria, M. et al. Targeted inhibition of transcription elongation in cells mediated by triplex-forming oligonucleotides. Proc. Natl. Acad. Sci. USA 97, 3862–3867 ( 2000).
Escude, C. et al. Stable triple helices formed by oligonucleotide N3′-P5′ phosphoramidates inhibit transcription elongation. Proc. Natl. Acad. Sci. USA 93, 4365–4369 (1996).
Dias, N. et al. Antisense PNA tridecamers targeted to the coding region of Ha-ras mRNA arrest polypeptide chain elongation. J. Mol. Biol. 294, 403–416 (1999).
Dheur, S. et al. Polyethylenimine but not cationic lipid improves antisense activity of 3′-capped phosphodiester oligonucleotides. Antisense Nucleic Acid Drug Dev. 9, 515–525 (1999).
Giles, R.V., Ruddell, C.J., Spiller, D.G., Green, J.A. & Tidd, D.M. Single base discrimination for ribonuclease H-dependent antisense effects within intact human leukaemia cells. Nucleic Acids Res. 23, 954– 961 (1995).
Sierakowska, H., Sambade, M.J., Agrawal, S. & Kole, R. Repair of thalassemic human β-globin mRNA in mammalian cells by antisense oligonucleotides. Proc. Natl. Acad. Sci. USA 93, 12840–12844 (1996).
Mir, L.M., Bureau, M.F., Rangara, R., Schwartz, B. & Scherman, D. Long-term, high level in vivo gene expression after electric pulse-mediated gene transfer into skeletal muscle. C.R. Acad. Sci. III 321, 893– 899 (1998).
Mir, L. et al. High efficiency gene transfer into skeletal muscle mediated by electric pulses. Proc. Natl. Acad. Sci. USA 96, 4262–4267 (1999).
Stein, C.A. Does antisense exist? Nat. Med. 1, 1119– 1121 (1995).
Flanagan, W.M. Antisense comes of age. Cancer Metastasis Rev. 17, 169–176 (1998).
Condon, T.P. & Bennett, C.F. Altered mRNA splicing and inhibition of human E-selectin expression by an antisense oligonucleotide in human umbilical vein endothelial cells. J. Biol. Chem. 271, 30398–30403 (1996).
Giles, R.V., Spiller, D.G., Clark, R.E. & Tidd, M.D. Antisense morpholino oligonucleotide analog induces missplicing of C-myc mRNA. Antisense Nucleic Acid Drug Dev. 9, 213–220 (1999).
Hawley, P., Nelson, J.S., Fearon, K.L., Zon, G. & Gibson, I. Comparison of binding of N3′-P5' phosphoramidate and phosphorothioate oligonucleotides to cell surface proteins of cultured cells. Antisense Nucleic Acid Drug Dev. 9, 61–69 (1999).
Testa, S.M., Gryaznov, S.M. & Turner, D.H. In vitro suicide inhibition of self-splicing of a group I intron from Pneumocystis carinii by an N3′-P5′ phosphoramidate hexanucleotide. Proc. Natl. Acad. Sci. USA 96, 2734–2739 (1999).
Kang, S.H., Cho, M.J. & Kole, R. Up-regulation of luciferase gene expression with antisense oligognucleotides: implications and applications in functional assay development . Biochemistry 37, 6235– 6239 (1998).
Taylor, J.K., Zhang, Q.Q., Wyatt, J.R. & Dean, N.M. Induction of endogenous Bcl-xS through the control of Bcl-x pre-mRNA splicing by antisense oligonucleotides. Nat. Biotechnol. 17, 1097–1100 (1999).
Schmajuk, G., Sierakowska, H. & Kole, R. Antisense oligonucleotides with different backbones. Modification of splicing pathways and efficacy of uptake. J. Biol. Chem. 274, 21783–21789 ( 1999).
McCurdy, S.N., Nelson, J.S., Hirschbein, B.L. & Fearon, K.L. An improved method for the synthesis of N3′-P5′ phosphoramidate oligonucleotides. Tetrahedr. Lett. 38, 207–210 (1997).
Nelson, J.S. et al. N3′-P5′ oligodeoxynucleotide phosphoramidates: a new method of synthesis based on a phosphoramidite amine-exchange reaction . J. Org. Chem. 62, 7278– 7287 (1997).
Fearon, K.L. et al. An improved synthesis of oligodeoxynucleotide N3′-P5' phosphoramidates and their chimera using hindered phosphoramidite monomers and a novel handle for reverse phase purification. Nucleic Acids Res. 26, 3813–3824 ( 1998).
Petrie, C.R., Reed, M.W., Adams, A.D. & Meyer, R.B. An improved CPG support for the synthesis of 3′-amine-tailed oligonucleotides. Bioconjug. Chem. 3, 85–87 (1992).
Charneau, P., Alizon, M. & Clavel, F. A second origin of DNA plus-strand synthesis is required for optimal human immunodeficiency virus replication. J. Virol. 66, 2814–2820 ( 1992).
Giles, R.V., Grzybowski, J., Spiller, D.G. & Tidd, D.M. Enhanced antisense effects resulting from an improved streptolysin-O protocol for oligodeoxynucleotide delivery into human leukaemia cells. Nucleosides Nucleotides 16, 1155–1163 (1997).
Spiller, D.G. et al. The influence of target protein half-life on the effectiveness of antisense oligonucleotide analog-mediated biologic responses. Antisense Nucleic Acid Drug Dev. 8, 281– 293 (1998).
Acknowledgements
We wish to thank R.V. Giles and C.J. Ruddell for helpful technical assistance and discussions, A. Winter for generation of stable cell lines, G. Juslin for technical expertise in in vivo experiments. We thank S. Gryaznov, D. Lloyd, and J.K. Chen for providing the first samples of oligophosphoramidates employing the oxidative phosphorylation chemistry, and L. DeDionisio and A. Raible for many of the phosphoramidate syntheses by the phosphoramidite amine-exchange methods. This work was supported by Agence Nationale de Recherche sur le Sida, and Centre International des Etudiants et Stagiaires. M.F. is supported by Coordenaçao de Aperfeicoamento de Pessoal de nivel Superior.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Faria, M., Spiller, D., Dubertret, C. et al. Phosphoramidate oligonucleotides as potent antisense molecules in cells and in vivo. Nat Biotechnol 19, 40–44 (2001). https://doi.org/10.1038/83489
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/83489
This article is cited by
-
Nonviral delivery systems for antisense oligonucleotide therapeutics
Biomaterials Research (2022)
-
Electroporation Enhanced Effect of Dystrophin Splice Switching PNA Oligomers in Normal and Dystrophic Muscle
Molecular Therapy - Nucleic Acids (2015)
-
Inhibition of gene expression in mice muscle by in vivo electrically mediated siRNA delivery
Gene Therapy (2005)