Abstract
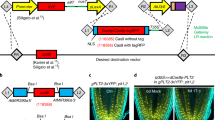
We have developed a chemical-inducible, site-specific DNA excision system in transgenic Arabidopsis plants mediated by the Cre/loxP DNA recombination system. Expression of the Cre recombinase was tightly controlled by an estrogen receptor-based fusion transactivator XVE. Upon induction by β-estradiol, sequences encoding the selectable marker, Cre, and XVE sandwiched by two loxP sites were excised from the Arabidopsis genome, leading to activation of the downstream GFP (green fluorescent protein) reporter gene. Genetic and molecular analyses indicated that the system is tightly controlled, showing high-efficiency inducible DNA excision in all 19 transgenic events tested with either single or multiple T-DNA insertions. The system provides a highly reliable method to generate marker-free transgenic plants after transformation through either organogenesis or somatic embryogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Yoder, J.I. & Goldsbrough, A.P. Transformation systems for generating marker free transgenic plants. Bio/Technology 12, 263–267 (1994).
Ow, D.W. & Medberry, S.L. Genome manipulations through site-specific recombination. Crit. Rev. Plant Sci. 14, 239–261 (1995).
Dale, E.C. & Ow, D.W. Gene transfer with subsequent removal of the selection gene from the host genome. Proc. Natl. Acad. Sci. USA 88, 10558–10562 (1991).
Russell, S.H., Hoopes, J.L. & Odell, J.T. Directed excision of a transgene from the plant genome. Mol. Gen. Genet. 234, 49–59 (1992).
Bayley, C.C., Morgan, M., Dale, E.C. & Ow, D.W. Exchange of gene activity in transgenic plants catalyzed by the Cre-lox site-specific recombination system. Plant Mol. Biol. 18, 353–361 (1992).
Qin, M., Bayley, C., Stockton, T. & Ow, D.W. Cre recombinase-mediated site-specific recombination between plant chromosomes. Proc. Natl. Acad. Sci. USA 91, 1706–1710 (1994).
Lloyd, A.M. & Davis, R.W. Functional expression of the yeast FLP/FRT site-specific recombination system in Nicotiana tabacum. Mol. Gen. Genet. 242, 653–657 (1994).
Komari, T., Hiei, Y., Saito, Y., Murai, N. & Kumashiro, T. Vectors carrying two separate T-DNAs for co-transformation of higher plants mediated by Agrobacterium tumefaciens and segregation of transformants free from selection markers. Plant J. 10, 165–174 (1996).
Gleave, A.P., Mitra, D.S., Mudge, S.R. & Morris, B.A. Selectable marker-free transgenic plants without sexual crossing: transient expression of cre recombinase and use of a conditional lethal dominant gene. Plant Mol. Biol. 40, 223–235 (1999).
Barry, G.F., Rogers, S.G., Fraley, R.T. & Brand, L. Identification of a cloned cytokinin biosynthetic gene. Proc. Natl. Acad. Sci. USA 81, 4776–4780 (1984).
Akiyoshi, D.E., Klee, H., Amasino, R.M., Nester, E.W. & Gordon, M.P. T-DNA of Agrobacterium tumefaciens encodes an enzyme of cytokinin biosynthesis. Proc. Natl. Acad. Sci. USA 81, 5994–5998 (1984).
Ebinuma, H., Sugita, K., Matsunaga, E. & Yamakado, M. Selection of marker-free transgenic plants using the isopentenyl transferase gene as a selectable marker. Proc. Natl. Acad. Sci. USA 94, 2117–2121 (1997).
Sugita, K., Kasahara, T., Matsunaga, E. & Ebinuma, H. A transformation vector for the production of marker-free transgenic plants containing a single copy transgene at high frequency. Plant J. 22, 461–469 (2000).
Kunkel, T., Niu, Q.-W., Chan, Y.S. & Chua, N.-H. Inducible isopentenyl transferase as a high-efficiency marker for plant transformation. Nat. Biotechnol. 17, 916–919 (1999).
Aoyama, T. & Chua, N.-H. A glucocorticoid-mediated transcriptional induction system in transgenic plants. Plant J. 11, 605–612 (1997).
Zubko, E., Scutt, C. & Meyer, P. Intrachromosomal recombination between two attP regions as a tool to remove selectable marker genes from tobacco transgenes. Nat. Biotechnol. 18, 442–445 (2000).
Satina, S., Blakeslee, A.F. & Avery, A.G. Demonstration of the three germ layers in the shoot apex of Datura by means of induced polyploidy in periclinal chimeras. Am. J. Bot. 27, 895–905 (1940).
Irish, V.F. & Sussex, I. M. A fate map of the Arabidopsis embryonic shoot apical meristem. Development 115, 745–753 (1992).
Furner, I.J. & Pumfrey, J.E. Cell fate in the shoot apical meristem of Arabidopsis thaliana. Development 115, 755–764 (1992).
Marcotrigiano, M. & Bernatzky, R. Arrangement of cell layers in the shoot apical meristems of periclinal chimeras influences cell fate. Plant J. 7, 193–202 (1995).
Zuo, J., Niu, Q.-W. & Chua, N.-H. An estrogen receptor-based transactivator XVE mediates highly inducible gene expression in transgenic plants. Plant J. 24, 265–273 (2000).
Austin, S., Ziese, M. & Sternberg, N. A novel role for site-specific recombination in maintenance of bacterial replicons. Cell 25, 729–736 (1981).
Odell, J., Caimi, P., Sauer, B. & Russell, S. Site-directed recombination in the genome of transgenic tobacco. Mol. Gen. Genet. 223, 369–378 (1990).
Zuo, J. et al. KORRIGAN, an Arabidopsis endo-1,4-β-glucanase, localizes to the cell plate by polarized targeting and is essential for cytokinesis. Plant Cell 12, 1137–1152 (2000).
Ishige, F., Takaichi, M., Foster, R., Chua, N.-H. & Oeda, K. A G-box motif (GCCACGTGCC) tetramer confers high-level constitutive expression in dicot and monocot plants. Plant J. 18, 443–448 (1999).
Koncz, C. et al. High-frequency T-DNA-mediated gene tagging in plants. Proc. Natl. Acad. Sci. USA 86, 8467–8471 (1989).
Murashige, T. & Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant 15, 473–497 (1962).
Sambrook, J., Fritsch, E.F. & Manistis, T. Molecular cloning: a laboratory manual. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY; 1989).
Acknowledgements
We thank Dr. D. Ow for providing pMM23. We are grateful to anonymous reviewers for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zuo, J., Niu, QW., Møller, S. et al. Chemical-regulated, site-specific DNA excision in transgenic plants. Nat Biotechnol 19, 157–161 (2001). https://doi.org/10.1038/84428
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/84428
This article is cited by
-
Agrobacterium rhizogenes-mediated marker-free transformation and gene editing system revealed that AeCBL3 mediates the formation of calcium oxalate crystal in kiwifruit
Molecular Horticulture (2024)
-
Cre-mediated autoexcision of selectable marker genes in soybean, cotton, canola and maize transgenic plants
Plant Cell Reports (2023)
-
Expression of Tobacco Lipid Transfer Protein NtLTP4 Enhances Tolerance to Abiotic and Biotic Stresses in Transgenic Potato Lines
Potato Research (2022)
-
A novel non-antibiotic selectable marker GASA6 for plant transformation
Plant Cell, Tissue and Organ Culture (PCTOC) (2022)
-
Gene activation via Cre/lox-mediated excision in cowpea (Vigna unguiculata)
Plant Cell Reports (2022)