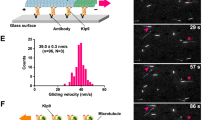
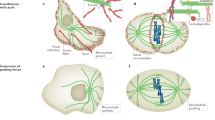
Abstract
The microtubule cytoskeleton and the mitotic spindle are highly dynamic structures1, yet their sizes are remarkably constant, thus indicating that the growth and shrinkage of their constituent microtubules are finely balanced2,3. This balance is achieved, in part, through kinesin-8 proteins (such as Kip3p in budding yeast and KLP67A in Drosophila) that destabilize microtubules3,4,5,6,7,8. Here, we directly demonstrate that Kip3p destabilizes microtubules by depolymerizing them — accounting for the effects of kinesin-8 perturbations on microtubule and spindle length observed in fungi and metazoan cells. Furthermore, using single-molecule microscopy assays9, we show that Kip3p has several properties that distinguish it from other depolymerizing kinesins, such as the kinesin-13 MCAK10,11. First, Kip3p disassembles microtubules exclusively at the plus end and second, remarkably, Kip3p depolymerizes longer microtubules faster than shorter ones. These properties are consequences of Kip3p being a highly processive, plus-end-directed motor12, both in vitro and in vivo. Length-dependent depolymerization provides a new mechanism for controlling the lengths of subcellular structures13.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Inoue, S. & Salmon, E. D. Force generation by microtubule assembly/disassembly in mitosis and related movements. Mol. Biol. Cell 6, 1619–1640 (1995).
Hildebrandt, E. R. & Hoyt, M. A. Mitotic motors in Saccharomyces cerevisiae. Biochim. Biophys. Acta 1496, 99–116 (2000).
Goshima, G., Wollman, R., Stuurman, N., Scholey, J. M. & Vale, R. D. Length control of the metaphase spindle. Curr. Biol. 15, 1979–1988 (2005).
Cottingham, F. R. & Hoyt, M. A. Mitotic spindle positioning in Saccharomyces cerevisiae is accomplished by antagonistically acting microtubule motor proteins. J. Cell Biol. 138, 1041–1053 (1997).
Miller, R. K. et al. The kinesin-related proteins, Kip2p and Kip3p, function differently in nuclear migration in yeast. Mol. Biol. Cell 9, 2051–2068 (1998).
Straight, A. F., Sedat, J. W. & Murray, A. W. Time-lapse microscopy reveals unique roles for kinesins during anaphase in budding yeast. J. Cell Biol. 143, 687–694 (1998).
Rischitor, P. E., Konzack, S. & Fischer, R. The Kip3-like kinesin KipB moves along microtubules and determines spindle position during synchronized mitoses in Aspergillus nidulans hyphae. Eukaryot. Cell 3, 632–645 (2004).
West, R. R., Malmstrom, T., Troxell, C. L. & McIntosh, J. R. Two related kinesins, klp5+ and klp6+, foster microtubule disassembly and are required for meiosis in fission yeast. Mol. Biol. Cell 12, 3919–3932 (2001).
Helenius, J., Brouhard, G., Kalaidzidis, Y., Diez, S. & Howard, J. The depolymerizing kinesin MCAK uses lattice diffusion to rapidly target microtubule ends. Nature 441, 115–119 (2006).
Desai, A., Verma, S., Mitchison, T. J. & Walczak, C. E. Kin I kinesins are microtubule-destabilizing enzymes. Cell 96, 69–78 (1999).
Hunter, A. W. et al. The kinesin-related protein MCAK is a microtubule depolymerase that forms an ATP-hydrolyzing complex at microtubule ends. Mol. Cell 11, 445–457 (2003).
Pereira, A. J., Dalby, B., Stewart, R. J., Doxsey, S. J. & Goldstein, L. S. Mitochondrial association of a plus end-directed microtubule motor expressed during mitosis in Drosophila. J. Cell Biol. 136, 1081–1090 (1997).
Marshall, W. F. Cellular length control systems. Annu. Rev. Cell Dev. Biol. 20, 677–693 (2004).
Goshima, G. & Vale, R. D. The roles of microtubule-based motor proteins in mitosis: comprehensive RNAi analysis in the Drosophila S2 cell line. J. Cell Biol. 162, 1003–1016 (2003).
Savoian, M. S., Gatt, M. K., Riparbelli, M. G., Callaini, G. & Glover, D. M. Drosophila Klp67A is required for proper chromosome congression and segregation during meiosis I. J. Cell Sci. 117, 3669–3677 (2004).
Walczak, C. E., Mitchison, T. J. & Desai, A. XKCM1: a Xenopus kinesin-related protein that regulates microtubule dynamics during mitotic spindle assembly. Cell 84, 37–47 (1996).
Maney, T., Wagenbach, M. & Wordeman, L. Molecular dissection of the microtubule depolymerizing activity of mitotic centromere-associated kinesin. J. Biol. Chem. 276, 34753–34758 (2001).
Lawrence, C. J., Malmberg, R. L., Muszynski, M. G. & Dawe, R. K. Maximum likelihood methods reveal conservation of function among closely related kinesin families. J. Mol. Evol. 54, 42–53 (2002).
Hyman, A. A., Salser, S., Drechsel, D. N., Unwin, N. & Mitchison, T. J. Role of GTP hydrolysis in microtubule dynamics: information from a slowly hydrolyzable analogue, GMPCPP. Mol. Biol. Cell 3, 1155–1167 (1992).
Howard, J. Mechanics of Motor Proteins and the Cytoskeleton. 367 (Sinauer Associates, Sunderland, MA, 2001).
Vale, R. D. et al. Direct observation of single kinesin molecules moving along microtubules. Nature 380, 451–453 (1996).
Sakamoto, T., Amitani, I., Yokota, E. & Ando, T. Direct observation of processive movement by individual myosin V molecules. Biochem. Biophys. Res. Commun. 272, 586–590 (2000).
Tanaka, K. et al. Molecular mechanisms of kinetochore capture by spindle microtubules. Nature 434, 987–994 (2005).
Akhmanova, A. & Hoogenraad, C. C. Microtubule plus-end-tracking proteins: mechanisms and functions. Curr. Opin. Cell Biol. 17, 47–54 (2005).
Howard, J. & Hyman, A. A. Dynamics and mechanics of the microtubule plus end. Nature 422, 753–758 (2003).
Dogterom, M., Felix, M. A., Guet, C. C. & Leibler, S. Influence of M-phase chromatin on the anisotropy of microtubule asters. J. Cell Biol. 133, 125–140 (1996).
Bringmann, H. et al. A kinesin-like motor inhibits microtubule dynamic instability. Science 303, 1519–1522 (2004).
Sproul, L. R., Anderson, D. J., Mackey, A. T., Saunders, W. S. & Gilbert, S. P. Cik1 targets the minus-end kinesin depolymerase kar3 to microtubule plus ends. Curr. Biol. 15, 1420–1427 (2005).
Bressan, D. A., Vazquez, J. & Haber, J. E. Mating type-dependent constraints on the mobility of the left arm of yeast chromosome III. J. Cell Biol. 164, 361–371 (2004).
Maekawa, H., Usui, T., Knop, M. & Schiebel, E. Yeast Cdk1 translocates to the plus end of cytoplasmic microtubules to regulate bud cortex interactions. EMBO J. 22, 438–449 (2003).
Acknowledgements
We thank R. Ciosk, J.E. Harber, K. Nasmyth, E. Schiebel, R. Tsien, F. Uhlmann and M. van Breugel for reagents, G. Brouhard, S. Diez, D. Drechsel, R. Hartman and Y. Kitamura for technical help, Y. Kalaidzidis for the motion tracking program, and V. Bormuth, G. Brouhard, S. Endow, E. Schaeffer and J. Stear for comments on an earlier draft of this manuscript. V.V., J.H., A.A.H. and J.H. were supported by the Max Planck Society and the National Institutes of Health. K.T. and T.T. were supported by Cancer Research UK, The Wellcome Trust, Human Frontier Science Program and a Lister Research Prize.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3 and Protein Purification Details (PDF 609 kb)
Supplementary Information
Supplementary Movie 1 (MOV 1160 kb)
Supplementary Information
Supplementary Movie 2 (MOV 511 kb)
Supplementary Information
Supplementary Movie 3 (MOV 1160 kb)
Supplementary Information
Supplementary Movie 4 (MOV 63 kb)
Supplementary Information
Supplementary Movie 5 (MOV 633 kb)
Supplementary Information
Supplementary Movie 6 (MOV 903 kb)
Supplementary Information
Supplementary Movie 7 (MOV 202 kb)
Rights and permissions
About this article
Cite this article
Varga, V., Helenius, J., Tanaka, K. et al. Yeast kinesin-8 depolymerizes microtubules in a length-dependent manner. Nat Cell Biol 8, 957–962 (2006). https://doi.org/10.1038/ncb1462
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1462
This article is cited by
-
Regulation of ciliary homeostasis by intraflagellar transport-independent kinesins
Cell Death & Disease (2024)
-
Minimal Mechanisms of Microtubule Length Regulation in Living Cells
Bulletin of Mathematical Biology (2024)
-
Mechanisms underlying spindle assembly and robustness
Nature Reviews Molecular Cell Biology (2023)
-
Determinant factors for residence time of kinesin motors at microtubule ends
Journal of Biological Physics (2023)
-
Mechanochemical tuning of a kinesin motor essential for malaria parasite transmission
Nature Communications (2022)