Abstract
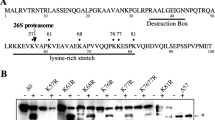
Xenopus RINGO/Speedy (XRINGO) is a potent inducer of oocyte meiotic maturation that can directly activate Cdk1 and Cdk2. Here, we show that endogenous XRINGO protein accumulates transiently during meiosis I entry and then is downregulated. This tight regulation of XRINGO expression is the consequence of two interconnected mechanisms: processing and degradation. XRINGO processing involves recognition of at least three distinct phosphorylated recognition motifs by the SCFβTrCP ubiquitin ligase, followed by proteasome-mediated limited degradation, resulting in an amino-terminal XRINGO fragment. XRINGO processing is directly stimulated by several kinases, including protein kinase A and glycogen synthase kinase-3β, and may contribute to the maintenance of G2 arrest. On the other hand, XRINGO degradation after meiosis I is mediated by the ubiquitin ligase Siah-2, which probably requires phosphorylation of XRINGO on Ser 243 and may be important for the omission of S phase at the meiosis-I–meiosis-II transition in Xenopus oocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Hershko, A. & Ciechanover, A. The ubiquitin system. Annu. Rev. Biochem. 67, 425–479 (1998).
Reed, S. I. Ratchets and clocks: the cell cycle, ubiquitylation and protein turnover. Nature Rev. Mol. Cell Biol. 4, 855–864 (2003).
Coux, O., Tanaka, K. & Goldberg, A. L. Structure and functions of the 20S and 26S proteasomes. Annu. Rev. Biochem. 65, 801–847 (1996).
Rape, M. & Jentsch, S. Taking a bite: proteasomal protein processing. Nature Cell Biol. 4, E113–E116 (2002).
Palombella, V. J., Rando, O. J., Goldberg, A. L. & Maniatis, T. The ubiquitin-proteasome pathway is required for processing the NF-κB1 precursor protein and the activation of NF-κB. Cell 78, 773–785 (1994).
Aza-Blanc, P., Ramirez-Weber, F. A., Laget, M. P., Schwartz, C. & Kornberg, T. B. Proteolysis that is inhibited by hedgehog targets Cubitus interruptus protein to the nucleus and converts it to a repressor. Cell 89, 1043–1053 (1997).
Hoppe, T. et al. Activation of a membrane-bound transcription factor by regulated ubiquitin/proteasome-dependent processing. Cell 102, 577–586 (2000).
Nebreda, A. R. & Ferby, I. Regulation of the meiotic cell cycle in oocytes. Curr. Opin. Cell Biol. 12, 666–675 (2000).
Castro, A. et al. Cyclin B/cdc2 induces c-Mos stability by direct phosphorylation in Xenopus oocytes. Mol. Biol. Cell 12, 2660–2671 (2001).
Hochegger, H. et al. New B-type cyclin synthesis is required between meiosis I and II during Xenopus oocyte maturation. Development 128, 3795–3807 (2001).
Mendez, R., Barnard, D. & Richter, J. D. Differential mRNA translation and meiotic progression require Cdc2-mediated CPEB destruction. EMBO J. 21, 1833–1844 (2002).
Ferby, I., Blazquez, M., Palmer, A., Eritja, R. & Nebreda, A. R. A novel p34(cdc2)-binding and activating protein that is necessary and sufficient to trigger G(2)/M progression in Xenopus oocytes. Genes Dev. 13, 2177–2189 (1999).
Lenormand, J. L., Dellinger, R. W., Knudsen, K. E., Subramani, S. & Donoghue, D. J. Speedy: a novel cell cycle regulator of the G2/M transition. EMBO J. 18, 1869–1877 (1999).
Karaiskou, A. et al. Differential regulation of Cdc2 and Cdk2 by RINGO and cyclins. J. Biol. Chem. 276, 36028–36034 (2001).
Porter, L. A. et al. Human Speedy: a novel cell cycle regulator that enhances proliferation through activation of Cdk2. J. Cell Biol. 157, 357–366 (2002).
Dinarina, A. et al. Characterization of a new family of cyclin-dependent kinase activators. Biochem. J. 386, 349–355 (2005).
Cheng, A., Gerry, S., Kaldis, P. & Solomon, M. J. Biochemical characterization of Cdk2-Speedy/Ringo A2. BMC Biochem. 6, 19 (2005).
Nebreda, A. R. CDK activation by non-cyclin proteins. Curr. Opin. Cell Biol. 18, 192–198 (2006).
Sagata, N., Oskarsson, M., Copeland, T., Brumbaugh, J. & Vande Woude, G. F. Function of c-mos proto-oncogene product in meiotic maturation in Xenopus oocytes. Nature 335, 519–525 (1988).
Perez, L. H., Antonio, C., Flament, S., Vernos, I. & Nebreda, A. R. Xkid chromokinesin is required for the meiosis I to meiosis II transition in Xenopus laevis oocytes. Nature Cell Biol. 4, 737–742 (2002).
Ciechanover, A. et al. Mechanisms of ubiquitin-mediated, limited processing of the NF-κB precursor protein p105. Biochimie 83, 341–349 (2001).
Price, M. A. & Kalderon, D. Proteolysis of the Hedgehog signaling effector Cubitus interruptus requires phosphorylation by Glycogen Synthase Kinase 3 and Casein Kinase 1. Cell 108, 823–835 (2002).
Jin, J. et al. SCFβ-TRCP links Chk1 signaling to degradation of the Cdc25A protein phosphatase. Genes Dev. 17, 3062–3074 (2003).
Della, N. G., Bowtell, D. D. & Beck, F. Expression of Siah-2, a vertebrate homologue of Drosophila sina, in germ cells of the mouse ovary and testis. Cell Tissue Res. 279, 411–419 (1995).
Moore, J. D., Kirk, J. A. & Hunt, T. Unmasking the S-phase-promoting potential of cyclin B1. Science 300, 987–990 (2003).
Padmanabhan, K. & Richter, J. D. Regulated Pumilio-2 binding controls RINGO/Spy mRNA translation and CPEB activation. Genes Dev. 20, 199–209 (2006).
Kanemori, Y., Uto, K. & Sagata, N. β-TrCP recognizes a previously undescribed nonphosphorylated destruction motif in Cdc25A and Cdc25B phosphatases. Proc. Natl Acad. Sci. USA 102, 6279–6284 (2005).
Watanabe, N. et al. M-phase kinases induce phospho-dependent ubiquitination of somatic Wee1 by SCF β-TrCP. Proc. Natl Acad. Sci. USA 101, 4419–4424 (2004).
Watanabe, N. et al. Cyclin-dependent kinase (CDK) phosphorylation destabilizes somatic Wee1 via multiple pathways. Proc. Natl Acad. Sci. USA 102, 11663–11668 (2005).
Suzuki, H. et al. Homodimer of two F-box proteins βTrCP1 or βTrCP2 binds to IκBα for signal-dependent ubiquitination. J. Biol. Chem. 275, 2877–2884 (2000).
Gutierrez, G. J. & Ronai, Z. Ubiquitin and SUMO systems in the regulation of mitotic checkpoints. Trends Biochem. Sci. 31, 324–332 (2006).
Polekhina, G. et al. Siah ubiquitin ligase is structurally related to TRAF and modulates TNF-α signaling. Nature Struct. Biol. 9, 68–75 (2002).
Rempel, R. E., Sleight, S. B. & Maller, J. L. Maternal Xenopus Cdk2-cyclin E complexes function during meiotic and early embryonic cell cycles that lack a G1 phase. J. Biol. Chem. 270, 6843–6855 (1995).
Murray, A. W. Cell cycle extracts. Methods Cell Biol. 36, 581–605 (1991).
Lorca, T. et al. Fizzy is required for activation of the APC/cyclosome in Xenopus egg extracts. EMBO J. 17, 3565–3575 (1998).
Glotzer, M., Murray, A. W. & Kirschner, M. W. Cyclin is degraded by the ubiquitin pathway. Nature 349, 132–138 (1991).
Schmitt, A. & Nebreda, A. R. Inhibition of Xenopus oocyte meiotic maturation by catalytically inactive protein kinase A. Proc. Natl Acad. Sci. USA 99, 4361–4366 (2002).
Newport, J. W. & Kirschner, M. W. Regulation of the cell cycle during early Xenopus development. Cell 37, 731–742 (1984).
Acknowledgements
We thank J. Moore and T. Hunt (Cancer Research UK Clare Hall Laboratories, South Mimms, UK) for providing advice and reagents to perform DNA replication assays in cyclin-E-depleted extracts; E. Pogge von Strandmann (University of Cologne, Germany) for sending us the Xenopus Siah-2 cDNA; and O. Coux (CNRS-CRBM, France) for providing E1 enzymes, helpful suggestions and for critically reading the manuscript. A.R.N. acknowledges the grant BFU2004-03566 from the Ministerio de Educacion y Ciencia of Spain.
Author information
Authors and Affiliations
Contributions
G.J.G. performed all the experiments presented in this manuscript; most of the work was done during his PhD at the EMBL. A.V. critically contributed to the characterization of the PKA phosphorylation sites on XRINGO. I.F. and G.S. generated some XRINGO mutants and the 9260 and 9261 rabbit antisera. A.C., T.L. and Z.R. provided essential tools to study the regulation of XRINGO by SCFβTrCP and Siah-2. A.R.N. participated in the design of the experiments and the interpretation of the results and wrote the manuscript together with G.J.G.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and Supplementary Information (PDF 1053 kb)
Rights and permissions
About this article
Cite this article
Gutierrez, G., Vögtlin, A., Castro, A. et al. Meiotic regulation of the CDK activator RINGO/Speedy by ubiquitin-proteasome-mediated processing and degradation. Nat Cell Biol 8, 1084–1094 (2006). https://doi.org/10.1038/ncb1472
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1472
This article is cited by
-
Meiosis resumption in human primordial germ cells from induced pluripotent stem cells by in vitro activation and reconstruction of ovarian nests
Stem Cell Research & Therapy (2022)
-
Essential role of the Cdk2 activator RingoA in meiotic telomere tethering to the nuclear envelope
Nature Communications (2016)
-
Expression and clinical significance of SIAH in laryngeal squamous cell carcinoma
Medical Oncology (2013)
-
Evolution of the Cdk-activator Speedy/RINGO in vertebrates
Cellular and Molecular Life Sciences (2012)
-
Siah1 proteins enhance radiosensitivity of human breast cancer cells
BMC Cancer (2010)