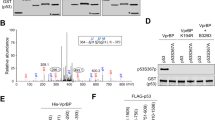
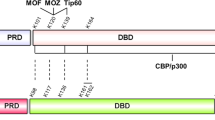
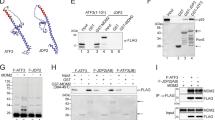
Abstract
The p300–CBP-associated factor (PCAF) is a histone acetyltransferase (HAT) involved in the reversible acetylation of various transcriptional regulators1, including the tumour suppressor p53. It is implicated in many cellular processes, such as transcription, differentiation, proliferation and apoptosis. We observed that knockdown of PCAF expression in HeLa or U2OS cell lines induces stabilization of the oncoprotein Hdm2, a RING finger E3 ligase primarily known for its role in controlling p53 stability2,3. To investigate the molecular basis of this effect, we examined whether PCAF is involved in Hdm2 ubiquitination. Here, we show that PCAF, in addition to its acetyltransferase activity, possesses an intrinsic ubiquitination activity that is critical for controlling Hdm2 expression levels, and thus p53 functions. Our data highlight a regulatory crosstalk between PCAF and Hdm2 activities, which is likely to have a central role in the subtle control of p53 activity after DNA damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Schiltz, R. L. & Nakatani, Y. The PCAF acetylase complex as a potential tumor suppressor. Biochim. Biophys. Acta 1470, M37–M53 (2000).
Vargas, D. A., Takahashi, S. & Ronai, Z. Mdm2: A regulator of cell growth and death. Adv. Cancer Res. 89, 1–34 (2003).
Iwakuma, T. & Lozano, G. MDM2, an introduction. Mol. Cancer Res. 1, 993–1000 (2003).
Bond, G. L., Hu, W. & Levine, A. J. MDM2 is a central node in the p53 pathway: 12 years and counting. Curr. Cancer Drug Targets 5, 3–8 (2005).
Liu, L. et al. p53 sites acetylated in vitro by PCAF and p300 are acetylated in vivo in response to DNA damage. Mol. Cell. Biol. 19, 1202–1209 (1999).
Fang, S., Jensen, J. P., Ludwig, R. L., Vousden, K. H. & Weissman, A. M. Mdm2 is a RING finger-dependent ubiquitin protein ligase for itself and p53. J. Biol. Chem. 275, 8945–8951 (2000).
Linares, L. K., Hengstermann, A., Ciechanover, A., Muller, S. & Scheffner, M. HdmX stimulates Hdm2-mediated ubiquitination and degradation of p53. Proc. Natl Acad. Sci. USA 100, 12009–12014 (2003).
Hershko, A., Ciechanover, A. & Varshavsky, A. The ubiquitin system. Nature Med. 6, 1073–1081 (2000).
Stommel, J. M. & Wahl, G. M. A new twist in the feedback loop: stress-activated MDM2 destabilization is required for p53 activation. Cell Cycle 4, 411–417 (2005).
Lorick, K. L. et al. RING fingers mediate ubiquitin-conjugating enzyme (E2)-dependent ubiquitination. Proc. Natl Acad. Sci. USA 96, 11364–11369 (1999).
Caron, C., Boyault, C. & Khochbin, S. Regulatory cross-talk between lysine acetylation and ubiquitination: role in the control of protein stability. Bioessays 27, 408–415 (2005).
Grossman, S. R. et al. Polyubiquitination of p53 by a ubiquitin ligase activity of p300. Science 300, 342–344 (2003).
Wertz, I. E. et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-κB signalling. Nature 430, 694–699 (2004).
Robinson, P. A. & Ardley, H. C. Ubiquitin-protein ligases. J. Cell Sci. 117, 5191–5194 (2004).
Stommel, J. M. & Wahl, G. M. Accelerated MDM2 auto-degradation induced by DNA-damage kinases is required for p53 activation. EMBO J. 23, 1547–1556 (2004).
Kroll, M. et al. Inducible degradation of IκBα by the proteasome requires interaction with the F-box protein h-βTrCP. J. Biol. Chem. 274, 7941–7945 (1999).
Wang, X., Taplick, J., Geva, N. & Oren, M. Inhibition of p53 degradation by Mdm2 acetylation. FEBS Lett. 561, 195–201 (2004).
Vogelstein, B., Lane, D. & Levine, A. J. Surfing the p53 network. Nature 408, 307–310 (2000).
Michael, D. & Oren, M. The p53–Mdm2 module and the ubiquitin system. Semin. Cancer Biol. 13, 49–58 (2003).
Lakin, N. D. & Jackson, S. P. Regulation of p53 in response to DNA damage. Oncogene 18, 7644–7655 (1999).
Hengstermann, A., Linares, L. K., Ciechanover, A., Whitaker, N. J. & Scheffner, M. Complete switch from Mdm2 to human papillomavirus E6-mediated degradation of p53 in cervical cancer cells. Proc. Natl Acad. Sci. USA 98, 1218–1223 (2001).
Barlev, N. A. et al. Acetylation of p53 activates transcription through recruitment of coactivators/histone acetyltransferases. Mol. Cell 8, 1243–1254 (2001).
You, H., Yamamoto, K. & Mak, T. W. Regulation of transactivation-independent proapoptotic activity of p53 by FOXO3a. Proc. Natl Acad. Sci. USA 103, 9051–9056 (2006).
Brooks, C. L. & Gu, W. p53 Ubiquitination: Mdm2 and beyond. Mol. Cell 21, 307–315 (2006).
Jin, Y., Zeng, S. X., Lee, H. & Lu, H. MDM2 mediates p300/CREB-binding protein-associated factor ubiquitination and degradation. J. Biol. Chem. 279, 20035–20043 (2004).
Jin, Y., Zeng, S. X., Dai, M. S., Yang, X. J. & Lu, H. MDM2 inhibits PCAF (p300/CREB-binding protein-associated factor)-mediated p53 acetylation. J. Biol. Chem. 277, 30838–30843 (2002).
Bode, A. M. & Dong, Z. Post-translational modification of p53 in tumorigenesis. Nature Rev. Cancer 4, 793–805 (2004).
Vousden, K. H. & Prives, C. p53 and prognosis: new insights and further complexity. Cell 120, 7–10 (2005).
Bres, V. et al. Differential acetylation of Tat coordinates its interaction with the co-activators cyclin T1 and PCAF. EMBO J. 21, 6811–6819 (2002).
Sun, X. Z., Nguyen, J. & Momand, J. Purification of recombinant p53 from Sf9 insect cells. Methods Mol. Biol. 234, 17–28 (2003).
Cuvier, O., Hart, C. M., Kas, E. & Laemmli, U. K. Identification of a multicopy chromatin boundary element at the borders of silenced chromosomal domains. Chromosoma 110, 519–531 (2002).
Acknowledgements
We thank V. Ogryzko for Flag–PCAF C-terminal deletion mutants, V. Favaudon for NCS, M. Oren for HA–Hdm2 expression vector, Y. Boublik (Recombinant Protein Platform, CRBM–CNRS) for production of baculovirus-expressed proteins, G. Lledo for enzyme production. We also thank our colleagues for their help, suggestions and criticisms, particularly the members of our groups and V. Gire, M. Piechaczyk and C. Sardet. This work was supported by grants from the Human Frontier Science Program (Young Investigator Program) to M.B., Action Concertée Incitative (ACI; M.B.) and ACI Biologie Cellulaire, Moléculaire et Structurale (O. Coux) from the French Ministère de la Recherche, Sidaction (M.B. and O. Coux), Association pour la Recherche sur le Cancer (ARC; O. Coux), and European contracts n°QLG1-CT-2001-02026 (O. Coux) and 012182 (M.B.). L.L. was supported by fellowships from Sidaction and ANRS (Agence Nationale de Recherches sur le Sida), C.C.B. by the Human Frontier Science Program, and O. Cuvier by a fellowship from the Fondation pour la Recherche Médicale.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5 and S6 (PDF 288 kb)
Rights and permissions
About this article
Cite this article
Linares, L., Kiernan, R., Triboulet, R. et al. Intrinsic ubiquitination activity of PCAF controls the stability of the oncoprotein Hdm2. Nat Cell Biol 9, 331–338 (2007). https://doi.org/10.1038/ncb1545
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1545
This article is cited by
-
p53 regulation by ubiquitin and ubiquitin-like modifications
Genome Instability & Disease (2022)
-
The post-translational modification of Fascin: impact on cell biology and its associations with inhibiting tumor metastasis
Amino Acids (2022)
-
SIRT7 activates p53 by enhancing PCAF-mediated MDM2 degradation to arrest the cell cycle
Oncogene (2020)
-
Structural basis for DNA damage-induced phosphoregulation of MDM2 RING domain
Nature Communications (2020)
-
Structural analysis reveals TLR7 dynamics underlying antagonism
Nature Communications (2020)