Abstract
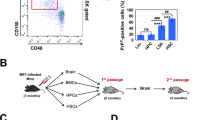
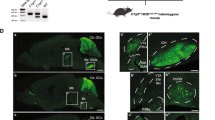
Transplanted bone marrow-derived cells (BMDCs) have been reported to fuse with cells of diverse tissues1,2,3,4,5,6,7,8,9,10,11,12,13, but the extremely low frequency of fusion has led to the view that such events are biologically insignificant. Nonetheless, in mice with a lethal recessive liver disease (tyrosinaemia), transplantation of wild-type BMDCs restored liver function by cell fusion and prevented death3,9, indicating that cell fusion can have beneficial effects. Here we report that chronic inflammation resulting from severe dermatitis or autoimmune encephalitis leads to robust fusion of BMDCs with Purkinje neurons and formation of hundreds of binucleate heterokaryons per cerebellum, a 10–100-fold higher frequency than previously reported8,10,11,14. Single haematopoietic stem-cell transplants showed that the fusogenic cell is from the haematopoietic lineage and parabiosis experiments revealed that fusion can occur without irradiation. Transplantation of rat bone marrow into mice led to activation of dormant rat Purkinje neuron-specific genes in BMDC nuclei after fusion with mouse Purkinje neurons, consistent with nuclear reprogramming. The precise neurological role of these heterokaryons awaits elucidation, but their frequency in brain after inflammation is clearly much higher than previously appreciated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ferrari, G. et al. Muscle regeneration by bone marrow-derived myogenic progenitors. Science 279, 1528–1530 (1998).
Bittner, R. E. et al. Recruitment of bone-marrow-derived cells by skeletal and cardiac muscle in adult dystrophic mdx mice. Anat. Embryol. 199, 391–396 (1999).
Lagasse, E. et al. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nature Med. 6, 1229–1234 (2000).
LaBarge, M. A. & Blau, H. M. Biological progression from adult bone marrow to mononucleate muscle stem cell to multinucleate muscle fiber in response to injury. Cell 111, 589–601. (2002).
Fukada, S. et al. Muscle regeneration by reconstitution with bone marrow or fetal liver cells from green fluorescent protein-gene transgenic mice. J. Cell Sci. 115, 1285–1293 (2002).
Camargo, F. D., Green, R., Capetenaki, Y., Jackson, K. A. & Goodell, M. A. Single hematopoietic stem cells generate skeletal muscle through myeloid intermediates. Nature Med. 9, 1520–1527 (2003).
Corbel, S. Y. et al. Contribution of hematopoietic stem cells to skeletal muscle. Nature Med. 9, 1528–1532 (2003).
Alvarez-Dolado, M. et al. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 425, 968–973 (2003).
Vassilopoulos, G., Wang, P. R. & Russell, D. W. Transplanted bone marrow regenerates liver by cell fusion. Nature (2003).
Weimann, J. M., Charlton, C. A., Brazelton, T. R., Hackman, R. C. & Blau, H. M. Contribution of transplanted bone marrow cells to Purkinje neurons in human adult brains. Proc. Natl Acad. Sci. USA 100, 2088–2093 (2003).
Weimann, J. M., Johansson, C. B., Trejo, A. & Blau, H. M. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nature Cell Biol. 5, 959–966 (2003).
Rizvi, A. Z. et al. Bone marrow-derived cells fuse with normal and transformed intestinal stem cells. Proc. Natl Acad. Sci. USA 103, 6321–6325 (2006).
Herzog, E. L. et al. Lung-specific nuclear reprogramming is accompanied by heterokaryon formation and Y chromosome loss following bone marrow transplantation and secondary inflammation. FASEB J. 21, 2592–2601 (2007).
Massengale, M., Wagers, A. J., Vogel, H. & Weissman, I. L. Hematopoietic cells maintain hematopoietic fates upon entering the brain. J. Exp. Med. 201, 1579–1589 (2005).
Miyamoto, T. The dendritic cell-specific transmembrane protein DC-STAMP is essential for osteoclast fusion and osteoclast bone-resorbing activity. Mod. Rheumatol. Jpn Rheumat. Assoc. 16, 341–342 (2006).
Huppertz, B., Bartz, C. & Kokozidou, M. Trophoblast fusion: fusogenic proteins, syncytins and ADAMs, and other prerequisites for syncytial fusion. Micron 37, 509–517 (2006).
Pajcini, K. V., Pomerantz, J. H., Alkan, O., Doyonnas, R. & Blau, H. M. Myoblasts and macrophages share molecular components that contribute to cell-cell fusion. J. Cell Biol. 180, 1005–1019 (2008).
Sacco, A. et al. IGF-I increases bone marrow contribution to adult skeletal muscle and enhances the fusion of myelomonocytic precursors. J. Cell Biol. 171, 483–492 (2005).
Wagers, A. J., Sherwood, R. I., Christensen, J. L. & Weissman, I. L. Little evidence for developmental plasticity of adult hematopoietic stem cells. Science 297, 2256–2259 (2002).
Pachter, J. S., de Vries, H. E. & Fabry, Z. The blood-brain barrier and its role in immune privilege in the central nervous system. J. Neuropathol. Exp. Neurol. 62, 593–604 (2003).
Yuan, H. et al. Effects of fractionated radiation on the brain vasculature in a murine model: blood-brain barrier permeability, astrocyte proliferation, and ultrastructural changes. Int. J. Rad. Oncol. Biol. Phys. 66, 860–866 (2006).
Wright, D. E., Wagers, A. J., Gulati, A. P., Johnson, F. L. & Weissman, I. L. Physiological migration of hematopoietic stem and progenitor cells. Science 294, 1933–1936 (2001).
Kastenmayer, R. J., Fain, M. A. & Perdue, K. A. A retrospective study of idiopathic ulcerative dermatitis in mice with a C57BL/6 background. J. Am. Assoc. Lab. Anim. Sci. 45, 8–12 (2006).
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nature Neurosci. 10, 1538–1543 (2007).
Fendrick, S. E., Xue, Q. S. & Streit, W. J. Formation of multinucleated giant cells and microglial degeneration in rats expressing a mutant Cu/Zn superoxide dismutase gene. J. Neuroinflam. 4, 9 (2007).
Rock, R. B. et al. Role of microglia in central nervous system infections. Clin. Microbiol. Rev. 17, 942–964, Table of contents (2004).
Doyonnas, R., LaBarge, M. A., Sacco, A., Charlton, C. & Blau, H. M. Hematopoietic contribution to skeletal muscle regeneration by myelomonocytic precursors. Proc. Natl Acad. Sci. USA 101, 13507–13512 (2004).
Kornek, B. et al. Multiple sclerosis and chronic autoimmune encephalomyelitis: a comparative quantitative study of axonal injury in active, inactive, and remyelinated lesions. Am. J. Pathol. 157, 267–276. (2000).
Linker, R. A. et al. EAE in beta-2 microglobulin-deficient mice: axonal damage is not dependent on MHC-I restricted immune responses. Neurobiol. Dis. 19, 218–228 (2005).
Gold, R., Linington, C. & Lassmann, H. Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalomyelitis research. Brain 129, 1953–1971 (2006).
Ousman, S. S. et al. Protective and therapeutic role for αB-crystallin in autoimmune demyelination. Nature 448, 474–479 (2007).
Ildstad, S. T. et al. Cross-species bone marrow transplantation: evidence for tolerance induction, stem cell engraftment, and maturation of T lymphocytes in a xenogeneic stromal environment (rat----mouse). J. Exp. Med. 174, 467–478 (1991).
Ikarashi, K. et al. Bone marrow cells contribute to regeneration of damaged glomerular endothelial cells. Kidney Int. 67, 1925–1933 (2005).
Sacco, T., De Luca, A. & Tempia, F. Properties and expression of Kv3 channels in cerebellar Purkinje cells. Mol. Cell. Neurosci. 33, 170–179 (2006).
Hall, K. U. et al. Phosphorylation-dependent inhibition of protein phosphatase-1 by G-substrate. A Purkinje cell substrate of the cyclic GMP-dependent protein kinase. J. Biol. Chem. 274, 3485–3495 (1999).
Stewart, F. M. et al. Host marrow stem cell potential and engraftability at varying times after low-dose whole-body irradiation. Blood 98, 1246–1251 (2001).
Johansson, C. B. et al. Identification of a neural stem cell in the adult mammalian central nervous system. Cell 96, 25–34 (1999).
Bunster, E. & Meyer, R. K. An improved method of parabiosis. Anat. Rec. 57, 339–343 (1933).
Palermo, A. T., Labarge, M. A., Doyonnas, R., Pomerantz, J. & Blau, H. M. Bone marrow contribution to skeletal muscle: a physiological response to stress. Dev. Biol. 279, 336–344 (2005).
Youssef, S. et al. The HMG-CoA reductase inhibitor, atorvastatin, promotes a Th2 bias and reverses paralysis in central nervous system autoimmune disease. Nature 420, 78–84 (2002).
Acknowledgements
We are grateful to the members of the Blau laboratory for helpful discussions; Mark LaBarge, Timothy Brazelton, Christine Dieterich, Tomas Olsson, Jonas Frisen and Lou Brundin for valuable discussions and critical reading of the manuscript; Corrine Davis for expert assistance with the evaluation of skin pathology; Peggy Kraft and Angelica Trejo for expert technical assistance. This work was supported by fellowships from the Wenner-Gren Foundation and the af Jochnick Foundation, Sweden to C. B. J, the National MS Society and grants from the NIH to L. S and S. Y and a career transitional fellowship from the National Multiple Sclerosis Society (NMSS) to S. Y, a CIHR grant MOP81382 to F. M. V. R, and NIH grants AG009521, HD018179, AG020961, AG024987, the McKnight Foundation, and the Baxter Foundation to H. M. B.
Author information
Authors and Affiliations
Contributions
C. B. J. planned the project, executed all experiments, generated confocal microscopy images, analysed the data, wrote the manuscript and put together the figures; S. Y. participated in the EAE experiment; K. K. carried out the bone marrow transplantations and helped with the parabiosis; C. H. assisted with the immunohistochemistry and generated confocal microscopy images; R. D. contributed to the data analysis and to the writing of the manuscript; S. Y. C. generated single HSC-transplanted mice. L. S. and F. M. V. R. contributed to the writing of the manuscript; H. M. B. planned the project, analysed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and Supplemental Table S1 (PDF 745 kb)
Rights and permissions
About this article
Cite this article
Johansson, C., Youssef, S., Koleckar, K. et al. Extensive fusion of haematopoietic cells with Purkinje neurons in response to chronic inflammation. Nat Cell Biol 10, 575–583 (2008). https://doi.org/10.1038/ncb1720
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1720
This article is cited by
-
Intrinsic signalling factors associated with cancer cell-cell fusion
Cell Communication and Signaling (2023)
-
The impact and outcomes of cancer-macrophage fusion
BMC Cancer (2023)
-
Cell fusion upregulates PD-L1 expression for evasion from immunosurveillance
Cancer Gene Therapy (2023)
-
Bone marrow mesenchymal stromal cells for diabetes therapy: touch, fuse, and fix?
Stem Cell Research & Therapy (2022)
-
Optimization study of plasmonic cell fusion
Scientific Reports (2022)