Abstract
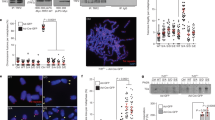
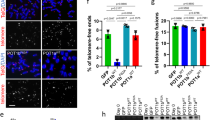
The telomere-capping complex shelterin protects functional telomeres and prevents the initiation of unwanted DNA-damage-response pathways. At the end of cellular replicative lifespan, uncapped telomeres lose this protective mechanism and DNA-damage signalling pathways are triggered that activate p53 and thereby induce replicative senescence. Here, we identify a signalling pathway involving p53, Siah1 (a p53-inducible E3 ubiquitin ligase) and TRF2 (telomere repeat binding factor 2; a component of the shelterin complex). Endogenous Siah1 and TRF2 were upregulated and downregulated, respectively, during replicative senescence with activated p53. Experimental manipulation of p53 expression demonstrated that p53 induces Siah1 and represses TRF2 protein levels. The p53-dependent ubiquitylation and proteasomal degradation of TRF2 are attributed to the E3 ligase activity of Siah1. Knockdown of Siah1 stabilized TRF2 and delayed the onset of cellular replicative senescence, suggesting a role for Siah1 and TRF2 in p53-regulated senescence. This study reveals that p53, a downstream effector of telomere-initiated damage signalling, also functions upstream of the shelterin complex.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Collado, M., Blasco, M. A. & Serrano, M. Cellular senescence in cancer and aging. Cell 130, 223–233 (2007).
Deng, Y., Chan, S. S. & Chang, S. Telomere dysfunction and tumour suppression: the senescence connection. Nat. Rev. Cancer 8, 450–458 (2008).
Begus-Nahrmann, Y. et al. p53 deletion impairs clearance of chromosomal-instable stem cells in aging telomere-dysfunctional mice. Nat. Genet. 41, 1138–1143 (2009).
Cicalese, A. et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell 138, 1083–1095 (2009).
Marion, R. M. et al. A p53-mediated DNA damage response limits reprogramming to ensure iPS cell genomic integrity. Nature 460, 1149–1153 (2009).
Cosme-Blanco, W. et al. Telomere dysfunction suppresses spontaneous tumorigenesis in vivo by initiating p53-dependent cellular senescence. EMBO Rep. 8, 497–503 (2007).
Feldser, D. M. & Greider, C. W. Short telomeres limit tumor progression in vivo by inducing senescence. Cancer Cell 11, 461–469 (2007).
Herbig, U., Jobling, W. A., Chen, B. P., Chen, D. J. & Sedivy, J. M. Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53 and p21CIP1, but not p16INK4a. Mol. Cell 14, 501–513 (2004).
Ju, Z. et al. Telomere dysfunction induces environmental alterations limiting hematopoietic stem cell function and engraftment. Nat. Med. 13, 742–747 (2007).
Nalapareddy, K., Jiang, H., Guachalla Gutierrez, L. M. & Rudolph, K. L. Determining the influence of telomere dysfunction and DNA damage on stem and progenitor cell aging: what markers can we use? Exp. Gerontol. 43, 998–1004 (2008).
Marion, R. M. et al. Telomeres acquire embryonic stem cell characteristics in induced pluripotent stem cells. Cell Stem Cell 4, 141–154 (2009).
van Steensel, B., Smogorzewska, A. & de Lange, T. TRF2 protects human telomeres from end-to-end fusions. Cell 92, 401–413 (1998).
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005).
Stansel, R. M., de Lange, T. & Griffith, J. D. T-loop assembly in vitro involves binding of TRF2 near the 3′ telomeric overhang. EMBO J. 20, 5532–5540 (2001).
Denchi, E. L. & de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature 448, 1068–1071 (2007).
Lechel, A. et al. The cellular level of telomere dysfunction determines induction of senescence or apoptosis in vivo. EMBO Rep. 6, 275–281 (2005).
Stagno D'Alcontres, M., Mendez-Bermudez, A., Foxon, J. L., Royle, N. J. & Salomoni, P. Lack of TRF2 in ALT cells causes PML-dependent p53 activation and loss of telomeric DNA. J. Cell Biol. 179, 855–867 (2007).
Buscemi, G. et al. The shelterin protein TRF2 inhibits Chk2 activity at telomeres in the absence of DNA damage. Curr. Biol. 19, 874–879 (2009).
Bilaud, T. et al. Telomeric localization of TRF2, a novel human telobox protein. Nat. Genet. 17, 236–239 (1997).
Zhu, X. D., Kuster, B., Mann, M., Petrini, J. H. & de Lange, T. Cell-cycle-regulated association of RAD50/MRE11/NBS1 with TRF2 and human telomeres. Nat. Genet. 25, 347–352 (2000).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Stewart, S. A. et al. Erosion of the telomeric single-strand overhang at replicative senescence. Nat. Genet. 33, 492–496 (2003).
Fujita, K. et al. p53 isoforms Δ133p53 and p53β are endogenous regulators of replicative cellular senescence. Nat. Cell Biol. 11, 1135–1142 (2009).
Matsuzawa, S. I. & Reed, J. C. Siah-1, SIP and Ebi collaborate in a novel pathway for β-catenin degradation linked to p53 responses. Mol. Cell 7, 915–926 (2001).
Tanikawa, J. et al. p53 suppresses the c-Myb-induced activation of heat shock transcription factor 3. J. Biol. Chem. 275, 15578–15585 (2000).
Bourdon, J. C. et al. p53 isoforms can regulate p53 transcriptional activity. Genes Dev. 19, 2122–2137 (2005).
Hu, G. & Fearon, E. R. Siah-1 N-terminal RING domain is required for proteolysis function, and C-terminal sequences regulate oligomerization and binding to target proteins. Mol. Cell. Biol. 19, 724–732 (1999).
Rodriguez, M. S., Desterro, J. M., Lain, S., Lane, D. P. & Hay, R. T. Multiple C-terminal lysine residues target p53 for ubiquitin-proteasome-mediated degradation. Mol. Cell. Biol. 20, 8458–8467 (2000).
Katoh, S. et al. High precision NMR structure and function of the RING-H2 finger domain of EL5, a rice protein whose expression is increased upon exposure to pathogen-derived oligosaccharides. J. Biol. Chem. 278, 15341–15348 (2003).
Lorick, K. L. et al. RING fingers mediate ubiquitin-conjugating enzyme (E2)-dependent ubiquitination. Proc. Natl Acad. Sci. USA 96, 11364–11369 (1999).
Karlseder, J., Smogorzewska, A. & de Lange, T. Senescence induced by altered telomere state, not telomere loss. Science 295, 2446–2449 (2002).
Jacobs, J. J. & de Lange, T. Significant role for p16INK4a in p53-independent telomere-directed senescence. Curr. Biol. 14, 2302–2308 (2004).
Smogorzewska, A. & de Lange, T. Different telomere damage signaling pathways in human and mouse cells. EMBO J. 21, 4338–4348 (2002).
Collado, M. et al. Tumour biology: senescence in premalignant tumours. Nature 436, 642 (2005).
Batchelor, E., Loewer, A. & Lahav, G. The ups and downs of p53: understanding protein dynamics in single cells. Nat. Rev. Cancer 9, 371–377 (2009).
Zhang, T., Brazhnik, P. & Tyson, J. J. Exploring mechanisms of the DNA-damage response: p53 pulses and their possible relevance to apoptosis. Cell Cycle 6, 85–94 (2007).
Winter, M. et al. Control of HIPK2 stability by ubiquitin ligase Siah-1 and checkpoint kinases ATM and ATR. Nat. Cell Biol. 10, 812–824 (2008).
Kamura, T. et al. Cytoplasmic ubiquitin ligase KPC regulates proteolysis of p27Kip1 at G1 phase. Nat. Cell Biol. 6, 1229–1235 (2004).
Lin, D. I. et al. Phosphorylation-dependent ubiquitination of cyclin D1 by the SCF(FBX4-αB crystallin) complex. Mol. Cell 24, 355–366 (2006).
Dimitrova, Y. N. et al. Direct ubiquitination of β-catenin by Siah-1 and regulation by the exchange factor TBL1. J. Biol. Chem. 285, 13507–13516 (2010).
Henderson, B. R. Nuclear-cytoplasmic shuttling of APC regulates β-catenin subcellular localization and turnover. Nat. Cell Biol. 2, 653–660 (2000).
Frew, I. J. et al. Normal p53 function in primary cells deficient for Siah genes. Mol. Cell. Biol. 22, 8155–8164 (2002).
Wright, W. E. & Shay, J. W. Telomere dynamics in cancer progression and prevention: fundamental differences in human and mouse telomere biology. Nat. Med. 6, 849–851 (2000).
Blasco, M. A. Telomeres and human disease: ageing, cancer and beyond. Nat. Rev. Genet. 6, 611–622 (2005).
Munoz, P., Blanco, R., Flores, J. M. & Blasco, M. A. XPF nuclease-dependent telomere loss and increased DNA damage in mice overexpressing TRF2 result in premature aging and cancer. Nat. Genet. 37, 1063–1071 (2005).
Roperch, J. P. et al. SIAH-1 promotes apoptosis and tumor suppression through a network involving the regulation of protein folding, unfolding, and trafficking: identification of common effectors with p53 and p21Waf1. Proc. Natl Acad. Sci. USA 96, 8070–8073 (1999).
Telerman, A. & Amson, R. The molecular programme of tumour reversion: the steps beyond malignant transformation. Nat. Rev. Cancer 9, 206–216 (2009).
Munoz, P., Blanco, R. & Blasco, M. A. Role of the TRF2 telomeric protein in cancer and ageing. Cell Cycle 5, 718–721 (2006).
Bischoff, F. Z. et al. Spontaneous abnormalities in normal fibroblasts from patients with Li-Fraumeni cancer syndrome: aneuploidy and immortalization. Cancer Res. 50, 7979–7984 (1990).
Robles, A. I. et al. Schedule-dependent synergy between the heat shock protein 90 inhibitor 17-(dimethylaminoethylamino)-17-demethoxygeldanamycin and doxorubicin restores apoptosis to p53-mutant lymphoma cell lines. Clin. Cancer Res. 12, 6547–6556 (2006).
Sengupta, S. et al. BLM helicase-dependent transport of p53 to sites of stalled DNA replication forks modulates homologous recombination. EMBO J. 22, 1210–1222 (2003).
Buolamwini, J. K. et al. Small molecule antagonists of the MDM2 oncoprotein as anticancer agents. Curr. Cancer Drug Targets 5, 57–68 (2005).
Yang, Q. et al. Functional diversity of human protection of telomeres 1 isoforms in telomere protection and cellular senescence. Cancer Res. 67, 11677–11686 (2007).
Conze, D. B., Wu, C. J., Thomas, J. A., Landstrom, A. & Ashwell, J. D. Lys63-linked polyubiquitination of IRAK-1 is required for interleukin-1 receptor- and toll-like receptor-mediated NF-κB activation. Mol. Cell Biol. 28, 3538–3547 (2008).
Yang, Q., Zheng, Y. L. & Harris, C. C. POT1 and TRF2 cooperate to maintain telomeric integrity. Mol. Cell. Biol. 25, 1070–1080 (2005).
Chang, W., Dynek, J. N. & Smith, S. TRF1 is degraded by ubiquitin-mediated proteolysis after release from telomeres. Genes Dev. 17, 1328–1333 (2003).
Michishita, E., Park, J. Y., Burneskis, J. M., Barrett, J. C. & Horikawa, I. Evolutionarily conserved and nonconserved cellular localizations and functions of human SIRT proteins. Mol. Biol. Cell 16, 4623–4635 (2005).
Shevchenko, A., Tomas, H., Havlis, J., Olsen, J. V. & Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 1, 2856–2860 (2006).
Jeram, S. M., Srikumar, T., Pedrioli, P. G. & Raught, B. Using mass spectrometry to identify ubiquitin and ubiquitin-like protein conjugation sites. Proteomics 9, 922–934 (2009).
Acknowledgements
We thank M. Tainsky, B. Vogelstein and T. de Lange for cells and reagents. We also thank K. Kumamoto for carrying out Nutlin-3a treatment, A. Robles for carrying out doxorubicin treatment of lymphoblast cells, M. Yoneda for technical assistance and E. Spillare for continuous support. This research was supported in part by the Intramural Research Program of the NIH, NCI. B.V. was supported by the grants from the Grant Agency of the Czech Republic (number 301/08/1468) and the Internal Grant Agency of Health of Czech Republic (number NS/9812-4). J.C.B. was supported by Breast Cancer Campaign, Cancer-Research UK and the Institut National de la Sante et de la Recherche Medicale. D.L. is a Gibb fellow of Cancer-Research UK.
Author information
Authors and Affiliations
Contributions
K.F., I.H., A.M.M. and L.M.M.J. performed experiments. B.V., J.-C.B. and D.P.L. provided essential reagents and suggestions. K.F., I.H., E.A., L.M.M.J. and C.C.H. coordinated the study and wrote the manuscript. C.C.H. was responsible for the overall project. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1468 kb)
Rights and permissions
About this article
Cite this article
Fujita, K., Horikawa, I., Mondal, A. et al. Positive feedback between p53 and TRF2 during telomere-damage signalling and cellular senescence. Nat Cell Biol 12, 1205–1212 (2010). https://doi.org/10.1038/ncb2123
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2123