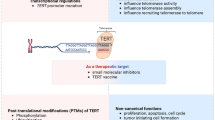
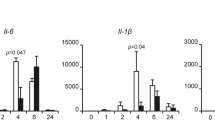
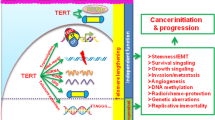
Abstract
Although elongation of telomeres is thought to be the prime function of reactivated telomerase in cancers, this activity alone does not account for all of the properties that telomerase reactivation attributes to human cancer cells. Here, we uncover a link between telomerase and NF-κB, a master regulator of inflammation. We observe that while blocking NF-κB signalling can inhibit effects of telomerase overexpression on processes relevant to transformation, increasing NF-κB activity can functionally substitute for reduced telomerase activity. Telomerase directly regulates NF-κB-dependent gene expression by binding to the NF-κB p65 subunit and recruitment to a subset of NF-κB promoters such as those of IL-6 and TNF-α, cytokines that are critical for inflammation and cancer progression. As NF-κB can transcriptionally upregulate telomerase levels, our findings suggest that a feed-forward regulation between them could be the key mechanistic basis for the coexistence of chronic inflammation and sustained telomerase activity in human cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Blackburn, E. H. et al. Recognition and elongation of telomeres by telomerase. Genome 31, 553–560 (1989).
Counter, C. M. et al. Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization. Proc. Natl Acad. Sci. USA 95, 14723–14728 (1998).
Hayflick, L. The limited in vitro lifetime of human diploid cell strains. Exp. Cell Res. 37, 614–636 (1965).
Verdun, R. E. & Karlseder, J. Replication and protection of telomeres. Nature 447, 924–931 (2007).
Shay, J. W. & Wright, W. E. Senescence and immortalization: role of telomeres and telomerase. Carcinogenesis 26, 867–874 (2005).
Harley, C. B. Telomerase and cancer therapeutics. Nat. Rev. Cancer 8, 167–179 (2008).
Chang, S. & DePinho, R. A. Telomerase extracurricular activities. Proc. Natl Acad. Sci. USA 99, 12520–12522 (2002).
Gordon, D. M. & Santos, J. H. The emerging role of telomerase reverse transcriptase in mitochondrial DNA metabolism. J. Nucl. Acidshttp://dx.doi.org/10.4061/2010/390791 (2010).
Maida, Y. et al. An RNA-dependent RNA polymerase formed by TERT and the RMRP RNA. Nature 461, 230–235 (2009).
Martinez, P. & Blasco, M. A. Telomeric and extra-telomeric roles for telomerase and the telomere-binding proteins. Nat. Rev. Cancer 11, 161–176 (2011).
Sarin, K. Y. et al. Conditional telomerase induction causes proliferation of hair follicle stem cells. Nature 436, 1048–1052 (2005).
Smith, L. L., Coller, H. A. & Roberts, J. M. Telomerase modulates expression of growth-controlling genes and enhances cell proliferation. Nat. Cell Biol. 5, 474–479 (2003).
Stewart, S. A. et al. Telomerase contributes to tumorigenesis by a telomere length-independent mechanism. Proc. Natl Acad. Sci. USA 99, 12606–12611 (2002).
Li, S., Crothers, J., Haqq, C. M. & Blackburn, E. H. Cellular and gene expression responses involved in the rapid growth inhibition of human cancer cells by RNA interference-mediated depletion of telomerase RNA. J. Biol. Chem. 280, 23709–23717 (2005).
Artandi, S. E. et al. Constitutive telomerase expression promotes mammary carcinomas in aging mice. Proc. Natl Acad. Sci. USA 99, 8191–8196 (2002).
Blasco, M. A., Rizen, M., Greider, C. W. & Hanahan, D. Differential regulation of telomerase activity and telomerase RNA during multi-stage tumorigenesis. Nat. Genet. 12, 200–204 (1996).
Cosme-Blanco, W. et al. Telomere dysfunction suppresses spontaneous tumorigenesis in vivo by initiating p53-dependent cellular senescence. EMBO Rep. 8, 497–503 (2007).
Gonzalez-Suarez, E. et al. Increased epidermal tumors and increased skin wound healing in transgenic mice overexpressing the catalytic subunit of telomerase, mTERT, in basal keratinocytes. EMBO J. 20, 2619–2630 (2001).
Gizard, F. et al. Telomerase activation in atherosclerosis and induction of telomerase reverse transcriptase expression by inflammatory stimuli in macrophages. Arterioscler Thromb. Vasc. Biol. 31, 245–252 (2011).
Mitchell, J. R., Wood, E. & Collins, K. A telomerase component is defective in the human disease dyskeratosis congenita. Nature 402, 551–555 (1999).
Shkreli, M. et al. Reversible cell-cycle entry in adult kidney podocytes through regulated control of telomerase and Wnt signaling. Nature Med. 18, 111–119 (2011).
Park, J. I. et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 460, 66–72 (2009).
Indran, I. R., Hande, M. P. & Pervaiz, S. hTERT overexpression alleviates intracellular ROS production, improves mitochondrial function, and inhibits ROS-mediated apoptosis in cancer cells. Cancer Res. 71, 266–276 (2011).
Mukherjee, S., Firpo, E. J., Wang, Y. & Roberts, J. M. Separation of telomerase functions by reverse genetics. Proc. Natl Acad. Sci. USA 108, E1363–E1371 (2011).
Masutomi, K. et al. The telomerase reverse transcriptase regulates chromatin state and DNA damage responses. Proc. Natl Acad. Sci. USA 102, 8222–8227 (2005).
Joseph, I. et al. The telomerase inhibitor imetelstat depletes cancer stem cells in breast and pancreatic cancer cell lines. Cancer Res. 70, 9494–9504 (2010).
Nitta, E. et al. Telomerase reverse transcriptase protects ATM-deficient hematopoietic stem cells from ROS-induced apoptosis through a telomere-independent mechanism. Blood 117, 4169–4180 (2011).
Ren, J. G. et al. Expression of telomerase inhibits hydroxyl radical-induced apoptosis in normal telomerase negative human lung fibroblasts. FEBS Lett. 488, 133–138 (2001).
Perkins, N. D. The diverse and complex roles of NF-κB subunits in cancer. Nat. Rev. Cancer 12, 121–132 (2012).
Oeckinghaus, A., Hayden, M. S. & Ghosh, S. Crosstalk in NF-κB signaling pathways. Nature Immunol. 12, 695–708 (2011).
Hayden, M. S. & Ghosh, S. Shared principles in NF-κB signaling. Cell 132, 344–362 (2008).
Basseres, D. S., Ebbs, A., Levantini, E. & Baldwin, A. S. Requirement of the NF-κB subunit p65/RelA for K-Ras-induced lung tumorigenesis. Cancer Res. 70, 3537–3546 (2010).
Loercher, A. et al. Nuclear factor-κB is an important modulator of the altered gene expression profile and malignant phenotype in squamous cell carcinoma. Cancer Res. 64, 6511–6523 (2004).
DeNardo, D. G., Johansson, M. & Coussens, L. M. Inflaming gastrointestinal oncogenic programming. Cancer Cell 14, 7–9 (2008).
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
Pikarsky, E. et al. NF-κB functions as a tumour promoter in inflammation-associated cancer. Nature 431, 461–466 (2004).
Jones, R. L. et al. Nuclear NF-κB/p65 expression and response to neoadjuvant chemotherapy in breast cancer. J. Clin. Pathol. 64, 130–135 (2011).
Teo, H. et al. Telomere-independent Rap1 is an IKK adaptor and regulates NF-κB-dependent gene expression. Nat. Cell Biol. 12, 758–767 (2010).
Ben-Neriah, Y. & Karin, M. Inflammation meets cancer, with NF-κB as the matchmaker. Nature Immunol. 12, 715–723 (2011).
Wu, Z. H. et al. ATM- and NEMO-dependent ELKS ubiquitination coordinates TAK1-mediated IKK activation in response to genotoxic stress. Mol. Cell 40, 75–86 (2010).
Perkins, N. D. Integrating cell-signalling pathways with NF-κB and IKK function. Nat. Rev. Mol. Cell Biol. 8, 49–62 (2007).
Saccani, S., Marazzi, I., Beg, A. A. & Natoli, G. Degradation of promoter-bound p65/RelA is essential for the prompt termination of the nuclear factor κB response. J. Exp. Med. 200, 107–113 (2004).
Chew, J. et al. WIP1 phosphatase is a negative regulator of NF-κB signalling. Nat. Cell Biol. 11, 659–666 (2009).
Cabannes, E., Khan, G., Aillet, F., Jarrett, R. F. & Hay, R. T. Mutations in the IkBa gene in Hodgkin’s disease suggest a tumour suppressor role for IκBα. Oncogene 18, 3063–3070 (1999).
Ngo, V. N. et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 470, 115–119 (2011).
Barbie, D. A. et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature 462, 108–112 (2009).
Van Antwerp, D. J., Martin, S. J., Kafri, T., Green, D. R. & Verma, I. M. Suppression of TNF- α-induced apoptosis by NF-κB. Science 274, 787–789 (1996).
Seimiya, H. et al. Telomere shortening and growth inhibition of human cancer cells by novel synthetic telomerase inhibitors MST-312, MST-295, and MST-1991. Mol. Cancer Ther. 1, 657–665 (2002).
Barkett, M. & Gilmore, T. D. Control of apoptosis by Rel/NF-κB transcription factors. Oncogene 18, 6910–6924 (1999).
Pfeffer, K. et al. Mice deficient for the 55 kd tumor necrosis factor receptor are resistant to endotoxic shock, yet succumb to L. monocytogenes infection. Cell 73, 457–467 (1993).
Dejager, L. & Libert, C. Tumor necrosis factor α mediates the lethalhepatotoxic effects of poly(I:C) in D-galactosamine-sensitized mice. Cytokine 42, 55–61 (2008).
Yin, L., Hubbard, A. K. & Giardina, C. NF- κB regulates transcription of the mouse telomerase catalytic subunit. J. Biol. Chem. 275, 36671–36675 (2000).
Vanden Berghe, W., De Bosscher, K., Boone, E., Plaisance, S. & Haegeman, G. The nuclear factor-κB engages CBP/p300 and histone acetyltransferase activity for transcriptional activation of the interleukin-6 gene promoter. J. Biol. Chem. 274, 32091–32098 (1999).
Rentoukas, E. et al. Connection between telomerase activity in PBMC and markers of inflammation and endothelial dysfunction in patients with metabolic syndrome. PLoS One 7, e35739 (2012).
Goytisolo, F. A. et al. Short telomeres result in organismal hypersensitivity to ionizing radiation in mammals. J. Exp. Med. 192, 1625–1636 (2000).
Ping, L., Asai, A., Okada, A., Isobe, K. & Nakajima, H. Dramatic increase of telomerase activity during dendritic cell differentiation and maturation. J. Leukoc. Biol. 74, 270–276 (2003).
Bodnar, A. G., Kim, N. W., Effros, R. B. & Chiu, C. P. Mechanism of telomerase induction during T cell activation. Exp. Cell Res. 228, 58–64 (1996).
Ogoshi, M., Takashima, A. & Taylor, R. S. Mechanisms regulating telomerase activity in murine T cells. J. Immunol. 158, 622–628 (1997).
Eaton, K. A., Opp, J. S., Gray, B. M., Bergin, I. L. & Young, V. B. Ulcerative typhlocolitis associated with Helicobacter mastomyrinus in telomerase-deficient mice. Vet. Pathol. 48, 713–725 (2011).
Chu, C., Qu, K., Zhong, F. L., Artandi, S. E. & Chang, H. Y. Genomic maps of long noncoding RNA occupancy reveal principles of RNA-chromatin interactions. Mol. Cell 44, 667–678 (2011).
Gilbert, L. A. & Hemann, M. T. DNA damage-mediated induction of a chemoresistant niche. Cell 143, 355–366 (2010).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Li, S. et al. Rapid inhibition of cancer cell growth induced by lentiviral delivery and expression of mutant-template telomerase RNA and anti-telomerase short-interfering RNA. Cancer Res. 64, 4833–4840 (2004).
Tergaonkar, V., Correa, R. G., Ikawa, M. & Verma, I. M. Distinct roles of IκB proteins in regulating constitutive NF-κB activity. Nat. Cell Biol. 7, 921–923 (2005).
Fujita, P. A. et al. The UCSC Genome Browser database: update 2011. Nucleic Acids Res. 39, D876-882 (2011).
Xu, H. et al. A signal-noise model for significance analysis of ChIP-seq with negative control. Bioinformatics 26, 1199–1204 (2010).
Kent, W. J. et al. The human genome browser at UCSC. Genome Res. 12, 996–1006 (2002).
Zhang, Z., Chang, C. W., Goh, W. L., Sung, W. K. & Cheung, E. CENTDIST: discovery of co-associated factors by motif distribution. Nucleic Acids Res. 39, W391–W399 (2011).
Simonet, T. et al. The human TTAGGG repeat factors 1 and 2 bind to a subset of interstitial telomeric sequences and satellite repeats. Cell Res. 21, 1028–1038 (2011).
McLean, C. Y. et al. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 28, 495–501 (2010).
Acknowledgements
We thank the Agency for Science Technology and Research, Singapore (A*Star) for funding and support to the V.T. laboratory.
Author information
Authors and Affiliations
Contributions
A.G., S.C.L., S.L., E.L. and V.T. designed experiments and planned the project. G.S., Z.Z., G.L. and W-K.S. carried our genome-wide analyses and were guided by E.L. A.G., E.M.S., E.K., S.C.L., M.W. and T.D.Y. carried out all experiments and animal work. J.Z. and W.J.C. provided patient samples and carried out experiments with patient material. A.G. and V.T. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2114 kb)
Rights and permissions
About this article
Cite this article
Ghosh, A., Saginc, G., Leow, S. et al. Telomerase directly regulates NF-κB-dependent transcription. Nat Cell Biol 14, 1270–1281 (2012). https://doi.org/10.1038/ncb2621
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2621
This article is cited by
-
The regulations of telomerase reverse transcriptase (TERT) in cancer
Cell Death & Disease (2024)
-
Genomic alterations in thyroid cancer: biological and clinical insights
Nature Reviews Endocrinology (2024)
-
Regulation of telomerase towards tumor therapy
Cell & Bioscience (2023)
-
The hTERT-p50 homodimer inhibits PLEKHA7 expression to promote gastric cancer invasion and metastasis
Oncogene (2023)
-
CCL18 signaling from tumor-associated macrophages activates fibroblasts to adopt a chemoresistance-inducing phenotype
Oncogene (2023)