Abstract
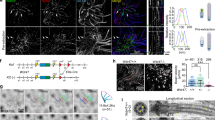
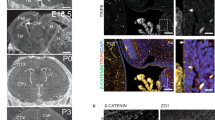
The transition zone is a specialized compartment found at the base of cilia, adjacent to the centriole distal end, where axonemal microtubules are heavily crosslinked to the surrounding membrane to form a barrier that gates the ciliary compartment. A number of ciliopathy molecules have been found to associate with the transition zone, but factors that directly recognize axonemal microtubules to specify transition zone assembly at the cilia base remain unclear. Here, through quantitative centrosome proteomics, we identify an axoneme-associated protein, CEP162 (KIAA1009), tethered specifically at centriole distal ends to promote transition zone assembly. CEP162 interacts with core transition zone components, and mediates their association with microtubules. Loss of CEP162 arrests ciliogenesis at the stage of transition zone assembly. Abolishing its centriolar tethering, however, allows CEP162 to stay on the growing end of the axoneme and ectopically assemble transition zone components at cilia tips. This generates extra-long cilia with strikingly swollen tips that actively release ciliary contents into the extracellular environment. CEP162 is thus an axoneme-recognition protein pre-tethered at centriole distal ends before ciliogenesis to promote and restrict transition zone formation specifically at the cilia base.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Sorokin, S. Centrioles and the formation of rudimentary cilia by fibroblasts and smooth muscle cells. J. Cell Biol. 15, 363–377 (1962).
Sorokin, S. P. Reconstructions of centriole formation and ciliogenesis in mammalian lungs. J. Cell Sci. 3, 207–230 (1968).
Eggenschwiler, J. T. & Anderson, K. V. Cilia and developmental signaling. Annu. Rev. Cell Dev. Biol. 23, 345–373 (2007).
Rohatgi, R. & Snell, W. J. The ciliary membrane. Curr. Opin. Cell Biol. 22, 541–546 (2010).
Schmidt, K. N. et al. Cep164 mediates vesicular docking to the mother centriole during early steps of ciliogenesis. J. Cell Biol. 199, 1083–1101 (2012).
Tanos, B. E. et al. Centriole distal appendages promote membrane docking, leading to cilia initiation. Gen. Dev. 27, 163–168 (2013).
Gilula, N. B. & Satir, P. The ciliary necklace. A ciliary membrane specialization. J. Cell Biol. 53, 494–509 (1972).
Ishikawa, H. & Marshall, W. F. Ciliogenesis: building the cell’s antenna. Nat. Rev. Mol. Cell Biol. 12, 222–234 (2011).
Pedersen, L. B. & Rosenbaum, J. L. Intraflagellar transport (IFT) role in ciliary assembly, resorption and signalling. Curr. Top. Dev. Biol. 85, 23–61 (2008).
Hu, Q. et al. A septin diffusion barrier at the base of the primary cilium maintains ciliary membrane protein distribution. Science 329, 436–439 (2010).
Kee, H. L. et al. A size-exclusion permeability barrier and nucleoporins characterize a ciliary pore complex that regulates transport into cilia. Nat. Cell Biol. 14, 431–437 (2012).
Anderson, R. G. The three-dimensional structure of the basal body from the rhesus monkey oviduct. J. Cell Biol. 54, 246–265 (1972).
Chih, B. et al. A ciliopathy complex at the transition zone protects the cilia as a privileged membrane domain. Nat. Cell Biol. 14, 61–72 (2012).
Reiter, J. F., Blacque, O. E. & Leroux, M. R. The base of the cilium: roles for transition fibres and the transition zone in ciliary formation, maintenance and compartmentalization. EMBO Rep. 13, 608–618 (2012).
Sang, L. et al. Mapping the NPHP-JBTS-MKS protein network reveals ciliopathy disease genes and pathways. Cell 145, 513–528 (2011).
Williams, C. L. et al. MKS and NPHP modules cooperate to establish basal body/transition zone membrane associations and ciliary gate function during ciliogenesis. J. Cell Biol. 192, 1023–1041 (2011).
Garcia-Gonzalo, F. R. et al. A transition zone complex regulates mammalian ciliogenesis and ciliary membrane composition. Nat. Gene. 43, 776–784 (2011).
Winkelbauer, M. E., Schafer, J. C., Haycraft, C. J., Swoboda, P. & Yoder, B. K. The C. elegans homologs of nephrocystin-1 and nephrocystin-4 are cilia transition zone proteins involved in chemosensory perception. J. Cell Sci. 118, 5575–5587 (2005).
Czarnecki, P. G. & Shah, J. V. The ciliary transition zone: from morphology and molecules to medicine. Trends Cell Biol. 22, 201–210 (2012).
Craige, B. et al. CEP290 tethers flagellar transition zone microtubules to the membrane and regulates flagellar protein content. J. Cell Biol. 190, 927–940 (2010).
Wang, W. J., Soni, R. K., Uryu, K. & Tsou, M. F. The conversion of centrioles to centrosomes: essential coupling of duplication with segregation. J. Cell Biol. 193, 727–739 (2011).
Ong, S. E. et al. Stable isotope labeling by amino acids in cell culture, SILAC, as a simple and accurate approach to expression proteomics. Mol. Cell. Proteom. 1, 376–386 (2002).
Spektor, A., Tsang, W. Y., Khoo, D. & Dynlacht, B. D. Cep97 and CP110 suppress a cilia assembly program. Cell 130, 678–690 (2007).
Singla, V., Romaguera-Ros, M., Garcia-Verdugo, J. M. & Reiter, J.F. Ofd1, a human disease gene, regulates the length and distal structure of centrioles. Dev. Cell 18, 410–424 (2010).
Tsang, W. Y. et al. CP110 suppresses primary cilia formation through its interaction with CEP290, a protein deficient in human ciliary disease. Dev. Cell 15, 187–197 (2008).
Otto, E. A. et al. Candidate exome capture identifies mutation of SDCCAG8 as the cause of a retinal-renal ciliopathy. Nat. Genet. 42, 840–850 (2010).
Tsang, W. Y. et al. Cep76, a centrosomal protein that specifically restrains centriole reduplication. Dev. Cell 16, 649–660 (2009).
Jakobsen, L. et al. Novel asymmetrically localizing components of human centrosomes identified by complementary proteomics methods. EMBO J. 30, 1520–1535 (2011).
Leon, A., Omri, B., Gely, A., Klein, C. & Crisanti, P. QN1/KIAA1009: a new essential protein for chromosome segregation and mitotic spindle assembly. Oncogene 25, 1887–1895 (2006).
Fry, A. M. et al. C-Nap1, a novel centrosomal coiled-coil protein and candidate substrate of the cell cycle-regulated protein kinase Nek2. J. Cell Biol. 141, 1563–1574 (1998).
Graser, S. et al. Cep164, a novel centriole appendage protein required for primary cilium formation. J. Cell Biol. 179, 321–330 (2007).
Snell, W. J. Mating in Chlamydomonas: a system for the study of specific cell adhesion. I. Ultrastructural and electrophoretic analyses of flagellar surface components involved in adhesion. J. Cell Biol. 68, 48–69 (1976).
Baldari, C. T. & Rosenbaum, J. Intraflagellar transport: it’s not just for cilia anymore. Curr. Opin. Cell Biol. 22, 75–80 (2010).
Habedanck, R., Stierhof, Y. D., Wilkinson, C. J. & Nigg, E. A. The Polo kinase Plk4 functions in centriole duplication. Nat. Cell Biol. 7, 1140–1146 (2005).
Tsou, M. F. & Stearns, T. Mechanism limiting centrosome duplication to once per cell cycle. Nature 442, 947–951 (2006).
Valente, E. M. et al. Mutations in CEP290, which encodes a centrosomalprotein, cause pleiotropic forms of Joubert syndrome. Nat. Genet. 38, 623–625 (2006).
Sakaguchi, T., Kikuchi, Y., Kuroiwa, A., Takeda, H. & Stainier, D. Y. The yolk syncytial layer regulates myocardial migration by influencing extracellular matrix assembly in zebrafish. Development 133, 4063–4072 (2006).
Huang, C. J., Tu, C. T., Hsiao, C. D., Hsieh, F. J. & Tsai, H. J. Germ-line transmission of a myocardium-specific GFP transgene reveals critical regulatory elementsin the cardiac myosin light chain 2 promoter of zebrafish. Dev. Dyn. 228, 30–40 (2003).
Kimmel, C. B., Ballard, W. W., Kimmel, S. R., Ullmann, B. & Schilling, T. F. Stages of embryonic development of the zebrafish. Dev. Dyn. 203, 253–310 (1995).
Long, S., Ahmad, N. & Rebagliati, M. The zebrafish nodal-related gene southpaw is required for visceral and diencephalic left–right asymmetry. Development 130, 2303–2316 (2003).
Gao, C., Wang, G., Amack, J. D. & Mitchell, D. R. Oda16/Wdr69 is essential for axonemal dynein assembly and ciliary motility during zebrafish embryogenesis. Dev. Dyn. 239, 2190–2197 (2010).
Robu, M. E. et al. p53 activation by knockdown technologies. PLoS Gene. 3, e78 (2007).
Acknowledgements
We are grateful to K. Anderson and her laboratory at Memorial Sloan-Kettering Cancer Center, USA, for reagents and antibodies. We thank L. Gunther, G. Perumal and F. Macaluso at the Analytical Imaging Center of Albert Einstein College of Medicine for assistance with transmission and scanning electron microscopy; K. Uryu at Rockefeller university and N. Lampen at MSKCC for assisting with the usage of electron microscopes; F. Foley at SUNY Upstate and D. Gutierrez at MSKCC for technical assistance and zebrafish management; and A. Hall, Z. Bao and C. Haynes at MSKCC for comments on the manuscript. This work was supported by the National Institutes of Health grants HL095690 to J.D.A. and GM088253 to M-F.B.T.
Author information
Authors and Affiliations
Contributions
W-J.W. and M-F.B.T. designed experiments and analysed data. W-J.W. performed most of the experiments. W-J.W., R.S. and J.M.A. did the quantitative centrosome proteomics. W-J.W. and R.S. prepared purified centrosomes. J.M.A. carried out SILAC mass spectrometry and analysed the data. The zebrafish works were carried out by H.G.T. and J.D.M. M.G.G. helped with initial zebrafish experiments. G.S.P., F.P.M. and W-J.W. performed the correlative light and scanning electron microscopy. M-F.B.T. and W-J.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 393 kb)
Supplementary Table 1
Supplementary Information (XLSX 56 kb)
Supplementary Table 2
Supplementary Information (XLS 41 kb)
Ciliary contents are actively released from cilia tips modified with CEP162.
RPE1 cells expressing GFP-tagged CEP162tN1C1C2 were serum-starved for 24 h, and imaged by time-lapse fluorescence microscopy (2 min time interval). Images shown in movies were generated from maximum intensity projections of a z-stack. (MOV 697 kb)
Bursts of cilia tips modified by CEP162.
Images were collected and processed from RPE1 cells expressing GFP-tagged CEP162tN1C1C2 as described in Supplementary Video S1. (MOV 578 kb)
Rights and permissions
About this article
Cite this article
Wang, WJ., Tay, H., Soni, R. et al. CEP162 is an axoneme-recognition protein promoting ciliary transition zone assembly at the cilia base. Nat Cell Biol 15, 591–601 (2013). https://doi.org/10.1038/ncb2739
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2739
This article is cited by
-
Mycoplasma hyopneumoniae J elicits an antioxidant response and decreases the expression of ciliary genes in infected swine epithelial cells
Scientific Reports (2020)
-
CEP120 interacts with C2CD3 and Talpid3 and is required for centriole appendage assembly and ciliogenesis
Scientific Reports (2019)
-
M-Phase Phosphoprotein 9 regulates ciliogenesis by modulating CP110-CEP97 complex localization at the mother centriole
Nature Communications (2018)
-
Super-resolution architecture of mammalian centriole distal appendages reveals distinct blade and matrix functional components
Nature Communications (2018)
-
Microtubule asters anchored by FSD1 control axoneme assembly and ciliogenesis
Nature Communications (2018)