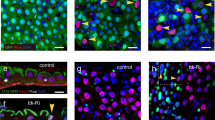
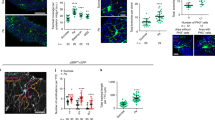
Abstract
Ageing results in loss of tissue homeostasis across taxa1. In the intestine of Drosophila melanogaster, ageing is correlated with an increase in intestinal stem cell (ISC) proliferation, a block in terminal differentiation of progenitor cells, activation of inflammatory pathways, and increased intestinal permeability2. However, causal relationships between these phenotypes remain unclear. Here, we demonstrate that ageing results in altered localization and expression of septate junction proteins in the posterior midgut, which is quite pronounced in differentiated enterocytes (ECs) at tricellular junctions (TCJs). Acute loss of the TCJ protein Gliotactin (Gli) in ECs results in increased ISC proliferation and a block in differentiation in intestines from young flies, demonstrating that compromised TCJ function is sufficient to alter ISC behaviour in a non-autonomous manner. Blocking the Jun N-terminal kinase signalling pathway is sufficient to suppress changes in ISC behaviour, but has no effect on loss of intestinal barrier function, as a consequence of Gli depletion. Our work demonstrates a pivotal link between TCJs, stem cell behaviour, and intestinal homeostasis and provides insights into causes of age-onset and gastrointestinal diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jones, D. L. & Rando, T. A. Emerging models and paradigms for stem cell ageing. Nat. Cell Biol. 13, 506–512 (2011).
Li, H. & Jasper, H. Gastrointestinal stem cells in health and disease: from flies to humans. Dis. Models Mech. 9, 487–499 (2016).
Marchiando, A. M., Graham, W. V. & Turner, J. R. Epithelial barriers in homeostasis and disease. Annu. Rev. Pathol. 5, 119–144 (2010).
Micchelli, C. A. & Perrimon, N. Evidence that stem cells reside in the adult Drosophila midgut epithelium. Nature 439, 475–479 (2006).
Ohlstein, B. & Spradling, A. The adult Drosophila posterior midgut is maintained by pluripotent stem cells. Nature 439, 470–474 (2006).
Biteau, B., Hochmuth, C. E. & Jasper, H. JNK activity in somatic stem cells causes loss of tissue homeostasis in the aging Drosophila gut. Cell Stem Cell 3, 442–455 (2008).
Jiang, H. et al. Cytokine/Jak/Stat signaling mediates regeneration and homeostasis in the Drosophila midgut. Cell 137, 1343–1355 (2009).
Rera, M. et al. Modulation of longevity and tissue homeostasis by the Drosophila PGC-1 homolog. Cell Metab. 14, 623–634 (2011).
Rera, M., Clark, R. I. & Walker, D. W. Intestinal barrier dysfunction links metabolic and inflammatory markers of aging to death in Drosophila. Proc. Natl Acad. Sci. USA 109, 21528–21533 (2012).
Tepass, U. & Hartenstein, V. The development of cellular junctions in the Drosophila embryo. Dev. Biol. 161, 563–596 (1994).
Fristrom, D. K. Septate junctions in imaginal disks of Drosophila: a model for the redistribution of septa during cell rearrangement. J. Cell Biol. 94, 77–87 (1982).
Schulte, J., Tepass, U. & Auld, V. J. Gliotactin, a novel marker of tricellular junctions, is necessary for septate junction development in Drosophila. J. Cell Biol. 161, 991–1000 (2003).
Byri, S. et al. The triple-repeat protein anakonda controls epithelial tricellular junction formation in Drosophila. Dev. Cell 33, 535–548 (2015).
Hildebrandt, A. et al. Bark beetle controls epithelial morphogenesis by septate junction maturation in Drosophila. Dev. Biol. 400, 237–247 (2015).
Auld, V. J., Fetter, R. D., Broadie, K. & Goodman, C. S. Gliotactin, a novel transmembrane protein on peripheral glia, is required to form the blood-nerve barrier in Drosophila. Cell 81, 757–767 (1995).
Schulte, J. et al. Gliotactin and Discs large form a protein complex at the tricellular junction of polarized epithelial cells in Drosophila. J. Cell Sci. 119, 4391–4401 (2006).
Padash-Barmchi, M., Charish, K., Que, J. & Auld, V. J. Gliotactin and Discs large are co-regulated to maintain epithelial integrity. J. Cell Sci. 126, 1134–1143 (2013).
Fontana, L., Partridge, L. & Longo, V. D. Extending healthy life span–from yeast to humans. Science 328, 321–326 (2010).
Osterwalder, T., Yoon, K. S., White, B. H. & Keshishian, H. A conditional tissue-specific transgene expression system using inducible GAL4. Proc. Natl Acad. Sci. USA 98, 12596–12601 (2001).
Roman, G., Endo, K., Zong, L. & Davis, R. L. P[Switch], a system for spatial and temporal control of gene expression in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 98, 12602–12607 (2001).
Choi, Y. J. et al. Age-related upregulation of Drosophila caudal gene via NF-κB in the adult posterior midgut. Biochim. Biophys. Acta 1780, 1093–1100 (2008).
Park, J. S., Kim, Y. S. & Yoo, M. A. The role of p38b MAPK in age-related modulation of intestinal stem cell proliferation and differentiation in Drosophila. Aging 1, 637–651 (2009).
Buchon, N., Broderick, N. A., Chakrabarti, S. & Lemaitre, B. Invasive and indigenous microbiota impact intestinal stem cell activity through multiple pathways in Drosophila. Genes Dev. 23, 2333–2344 (2009).
Buchon, N., Broderick, N. A. & Lemaitre, B. Gut homeostasis in a microbial world: insights from Drosophila melanogaster. Nat. Rev. Microbiol. 11, 615–626 (2013).
Guo, L., Karpac, J., Tran, S. L. & Jasper, H. PGRP-SC2 promotes gut immune homeostasis to limit commensal dysbiosis and extend lifespan. Cell 156, 109–122 (2014).
Clark, R. I. et al. Distinct shifts in microbiota composition during Drosophila aging impair intestinal function and drive mortality. Cell Rep. 12, 1656–1667 (2015).
Biteau, B., Karpac, J., Hwangbo, D. & Jasper, H. Regulation of Drosophila lifespan by JNK signaling. Exp. Gerontol. 46, 349–354 (2011).
Staley, B. K. & Irvine, K. D. Warts and Yorkie mediate intestinal regeneration by influencing stem cell proliferation. Curr. Biol. 20, 1580–1587 (2010).
Meier, J. & Sturm, A. The intestinal epithelial barrier: does it become impaired with age? Dig. Dis. 27, 240–245 (2009).
Ren, W. Y. et al. Age-related changes in small intestinal mucosa epithelium architecture and epithelial tight junction in rat models. Aging Clin. Exp. Res. 26, 183–191 (2014).
Tran, L. & Greenwood-VanMeerveld, B. Age-associated remodeling of the intestinal epithelial barrier. J. Gerontol. A Biol. Sci. Med. Sci. 68, 1045–1056 (2013).
McGuire, S. E., Mao, Z. & Davis, R. L. Spatiotemporal gene expression targeting with the TARGET and gene-switch systems in Drosophila. Sci. STKE 2004, pl6 (2004).
Martin-Blanco, E. et al. Puckered encodes a phosphatase that mediates a feedback loop regulating JNK activity during dorsal closure in Drosophila. Genes Dev. 12, 557–570 (1998).
Cruz, C., Glavic, A., Casado, M. & de Celis, J. F. A gain-of-function screen identifying genes required for growth and pattern formation of the Drosophila melanogaster wing. Genetics 183, 1005–1026 (2009).
Mitchell-Jordan, S. et al. Features of endogenous cardiomyocyte chromatin revealed by super-resolution STED microscopy. J. Mol. Cell. Cardiol. 53, 552–558 (2012).
Carpenter, A. E. CellProfiler: image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 7, R100 (2006).
Bakula, M. The persistence of a microbial flora during postembryogenesis of Drosophila melanogaster. J. Invertebr. Pathol. 14, 365–374 (1969).
Acknowledgements
The authors thank H. Jasper (The Buck Institute for Research on Aging, USA), V. Auld (University of British Columbia, Canada), J. F. de Celis (CBMSO, Spain), M. Furuse (Kobe University, Japan), the Vienna Drosophila RNAi Center (VDRC), and Bloomington Stock Center for reagents, M. Cilluffo from the Brain Research Institute at UCLA EM core facility, and the Jones and Walker laboratories for comments on the manuscript. In addition, we would like to thank Y. Wu and E. Stefani for their sharing their time, expertise, and STED technology. This work was supported by the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at the University of California, Los Angeles and the Rose Hills Foundation (D.L.J.), and the NIH: AG028092 (D.L.J.), AG040288 (D.L.J. and D.W.W.), AG049157 (D.W.W.), and a training grant that supports the UCLA-Caltech Medical Scientist Training Program, GM08042 (S.L.).
Author information
Authors and Affiliations
Contributions
M.R.-D. designed, performed and analysed experiments and wrote the manuscript. C.L.K., R.I.C., J.M.S., D.M.W., V.S., S.L. and C.D’A. designed, performed and analysed experiments. D.W.W. designed and analysed experiments. D.L.J. designed and analysed experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Localization of septate junction (SJ) proteins in the intestinal tract in young and aged flies.
(A) Schematic representation of a Drosophila melanogaster midgut (B–I) Localization of SJ proteins in posterior midgut and hindgut of young flies. (B) Nervana:GFP, (C) Neurexin IV:GFP, (D) Neuroglian:GFP, (E) Snakeskin, (F) Scribble:GFP, (G) Discs large, (H) Coracle, (I) Mesh. Images are representative of at least 5 midguts. Scale bars, 0.1 mm. (J–S) No changes in SJ protein levels were observed in hindguts from aged flies. STED (J–O) and confocal (P,Q) images comparing SJ proteins localization of 10do hindguts to 45do, scale bars, 1 μm and 5 μm. (R,S) STED images showed the AJ protein Arm localization is not affected by aging, scale bars, 1 μm. More than 14 hindguts per condition (10 ECs were observed per hindgut). Samples were dissected and stained in parallel under same conditions, pictures taken at same laser intensity.
Supplementary Figure 2 Verification of Gli reagents.
(A–C) GLI:GFP and anti-GLI co-localization (wing discs). GliRNAi efficiency demonstrated by immunofluorescence (IF) microscopy (wing discs) and qPCR (posterior midguts). (A–A′′) Wing disc peripodial membrane showing the co-localization of Gli-GFP (green) with Gli (red), DAPI (blue); Scale bars, 10 μm. (B,C) Gli protein is localized at the TCJ between wing cells (red signal in B;) in wild type discs. Gli is strongly reduced in discs expressing GliRNAi (SalPE-GAL4 UAS-GFP UAS-GliRNAi, C;). Scale bars, 50 μm. (D) RT-qPCR of posterior midguts 5966GSGAL4/UAS-GliRNAi flies RU+ and RU− showing a decrease of Gli expression after 5 and 9 days post GliRNAi expression. n = 75 females posterior midguts per condition. Bars are the mean +/− S.E.M (two tailed, unpaired Student’s t-test). ∗∗∗ = P < 0.001, ∗∗ = P < 0.01 represent a statistically significant difference. (E) Flies fed with EtOH (light blue) (5966GSGAL4/+, RU−, n = 270) show no statistical difference in number of flies that have lost barrier function, when compared to those fed RU (blue) (5966GSGAL4/ + fed, RU+, n = 268). Fisher’s exact test; two tailed. (F,G) EM images of SJ (arrowheads) between adjacent ECs in guts from 23do 5966GSGAL4/UAS-GliRNAi flies (12 RU+) (G) compared to control flies (F) 5966GSGAL4/UAS-GliRNAi (15 RU−). For each midgut 9 EC/EC Septate Junctions were observed per experimental condition. Scale bars, 0.1 μm. (H) Lifespan curves of 5966GSGAL4/UAS-GliRNAi [RU+, red (n = 265) and RU−, grey (n = 240)] female flies. T50 was significantly lower in RU+ d30 compared with RU− d37. Data analyzed with non parametric Log-Rank (Mantel-Cox) test; ∗∗∗ = P < 0.001, represent a statistically significant difference.
Supplementary Figure 3 Effects of Gli depletion in the posterior midgut.
(A–F) 5966GSGAL4; esg-GFP flies (RU+ or RU−) crossed with RNAi lines targeting Gli or Dlg. (A–D) Multiple GliRNAi lines induce an increase of ISC/EB cells, when expressed (RU+) (B;D) compared to RU− controls (A;C), similar to results presented in Fig. 4D. (E,F) Depletion of Dlg (dlgRNAi, RU+) (E) induces an increase in ISC/EBs compared to controls (RU−) (F). (G–K) Changes in ISC/EB number after Gli depletion are reversible (5966GSGAL4; esg-GFP UAS-GliRNAiRU+, RU−). (G) Graphical summary showing the statistical significance in changes in ISC/EB number depicted in H–K. 9do RU− UAS-GliRNAi n = 18, 9do RU+ UAS-GliRNAi n = 15, recovery experiment n = 19, 20do RU− UAS-GliRNAi n = 21, 20do RU+ UAS-GliRNAi n = 20. Each data point is an average proportion calculated from 4 independent images per midgut and bars are the mean +/− S.E.M of those averages (ONE-way ANOVA/Tukey’s multiple comparisons test) ∗∗ = P < 0.01, ∗∗∗ = P < 0.001, ∗∗∗∗ = P < 0.0001 represents a statistically significant difference. (H) Intestine from 5966GSGAL4; esg-GFP UAS-GliRNAi (fed RU+) for 9 days. (I) Flies were shifted onto food containing EtOH (RU−) to re-initiate Gli expression for 11 days. Re-expression of Gli resulted in resumption of normal ISC behavior and morphology compared to guts from 20do RU− 5966GSGAL4; esg-GFP UAS-GliRNAi controls (J) or guts from 20do 5966GSGAL4; esg-GFP GliRNAi flies fed RU+ (K). Scale bars, 10 μm. (L) Graphical summary showing that depletion of Gli from EEs using Rab3-GAL4, tubGAL80TS for 6d at 29 °C (red) did not result in any changes in ISC behavior, compared to control flies (y, w1118; grey). Each data point is an average proportion calculated from 4 independent images per midgut and bars are the median with interquartile range of those averages (Kruskal-Wallis/Dunn multiple comparisons test). Ctrl n = 18; UAS-GliRNAi n = 20. (M,N) No significant differences were detected in AMP expression, after 2, 5, 9 and 21 days post GliRNAi expression. ∗∗ = P < 0.01, represent statistically significant difference. (M) Drosomycin, (N) Diptericin. n = 6 replicates of 5. Boxplots display the first and third quartile, with horizontal bar at the median and whiskers showing the most extreme data point, which is no more than 1.5 times the interquartile range from the box.
Supplementary Figure 4 Neither changes in bacteria nor death of ECs induces changes in ISC behavior downstream of Gli.
(A–F) Epistatic analysis between Gli and apoptosis inhibitors. (A) ISC/EB counts in flies of indicated genotypes. RU− GliRNAi n = 19, RU+ GliRNAi n = 25, GliRNAi dIAP n = 25, dIAP n = 25, GliRNAi P35 n = 17, P35 n = 15. Each data point is an average proportion calculated from 4 independent images per midgut and bars are the mean +/− S.E.M (ONE-way ANOVA/Tukey’s multiple comparisons test). (B) Mitosis counts in midguts from flies of indicated genotypes. RU− GliRNAi n = 19, RU+ GliRNAi n = 25, GliRNAi dIAP n = 25, dIAP n = 25, GliRNAi P35 n = 17, P35 n = 15. Each data point is an average proportion calculated from 4 independent images per midgut and bars are the median with interquartile range (Kruskal-Wallis/Dunn multiple comparisons test) ∗∗∗∗ = P < 0.0001, represent statistically significant difference. (C–F) IF images of midguts from flies of indicated genotypes stained with DAPI (nuclei, blue), GFP (esg + cells, green) and PH3 (mitotic cells, red, arrowheads) following 9 days of incubation in RU+ or RU−. Co-expression of GliRNAi with dIAP (5966GSGAL4/UAS-GliRNAi; UAS-dIAP, RU+) or P35 (5966GSGAL4/UAS-GliRNAi; UAS-P35, RU+ (A,B;E,F) does not rescue the effect on ISCs produced by depletion of Gli (5966GSGAL4/UAS-GliRNAiUAS-LacZ, RU+) (A,B,D). Compare to control flies fed EtOH (RU−) (5966GSGAL4/UAS-GliRNAiUAS-LacZ, RU−) (A–C). (C–F) Images are representative of at least 15 midguts, 4 images taken per midgut. (G–H′) Under axenic conditions, reduction of Gli expression in ECs still initiates JNK pathway activation, as reported by puc-lacZ expression (G,H: red or G’,H’: grey) in ECs 2 days after reducing Gli expression (H–H’), compared to RU− controls (G–G’). ISC/EBs marked by esg-GFP (green), cell nuclei marked by DAPI (blue). (I) MRS plates showing absence of bacterial colonies from cultured fly homogenates of axenic flies at 7do. (J) RT-qPCR of posterior midguts from 5966GSGAL4/UAS-GliRNAi flies (RU+ and RU−) showing an increase of puc expression after 2, 5, 9 days post GliRNAi expression. n = 75 females posterior midguts per condition. Bars are the mean +/− S.E.M (two tailed, unpaired Student’s t-test). ∗∗∗ = P < 0.001, ∗∗ = P < 0.01, ∗ = P < 0.05 represent statistically significant difference. (K) Lifespan curves of 5966GSGAL4RU+ female flies crossed with yw118 (grey, n = 290), UAS-BskDN (green, n = 277), UAS-GliRNAi flies (red, n = 259) and GliRNAi UAS-BskDN (blue, n = 264). ∗∗∗ = P < 0.001, non parametric Log-rank (Mantel-Cox) test. Scale bars, 10 μm.
Supplementary information
Supplementary Information
Supplementary Information (PDF 8017 kb)
Supplementary Table 1
Supplementary Information (XLSX 587 kb)
Supplementary Table 2
Supplementary Information (XLSX 320 kb)
Rights and permissions
About this article
Cite this article
Resnik-Docampo, M., Koehler, C., Clark, R. et al. Tricellular junctions regulate intestinal stem cell behaviour to maintain homeostasis. Nat Cell Biol 19, 52–59 (2017). https://doi.org/10.1038/ncb3454
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3454
This article is cited by
-
Basal stem cell progeny establish their apical surface in a junctional niche during turnover of an adult barrier epithelium
Nature Cell Biology (2023)
-
A framework for intestinal barrier dysfunction in aging
Nature Aging (2023)
-
Sexual identity of enterocytes regulates autophagy to determine intestinal health, lifespan and responses to rapamycin
Nature Aging (2022)
-
white regulates proliferative homeostasis of intestinal stem cells during ageing in Drosophila
Nature Metabolism (2021)
-
Claudin-9 constitutes tight junctions of folliculo-stellate cells in the anterior pituitary gland
Scientific Reports (2021)