Abstract
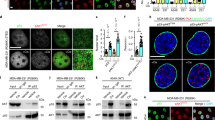
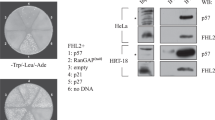
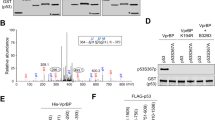
Transcriptional activity of p53, a central regulatory switch in a network controlling cell proliferation and apoptosis, is modulated by protein stability and post-translational modifications including phosphorylation and acetylation. Here we demonstrate that the human serine/threonine kinase homeodomain-interacting protein kinase-2 (HIPK2) colocalizes and interacts with p53 and CREB-binding protein (CBP) within promyelocytic leukaemia (PML) nuclear bodies. HIPK2 is activated by ultraviolet (UV) radiation and selectively phosphorylates p53 at Ser 46, thus facilitating the CBP-mediated acetylation of p53 at Lys 382, and promoting p53-dependent gene expression. Accordingly, the kinase function of HIPK2 mediates the increased expression of p53 target genes, which results in growth arrest and the enhancement of UV-induced apoptosis. Interference with HIPK2 expression by antisense oligonucleotides impairs UV-induced apoptosis. Our results imply that HIPK2 is a novel regulator of p53 effector functions involved in cell growth, proliferation and apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Momand, J., Zambetti, G. P., Olson, D. C., George, D. & Levine, A. J. The mdm-2 oncogene product forms a complex with the p53 protein and inhibits p53-mediated transactivation. Cell 69, 1237–1245 (1992).
Oliner, J. D. et al. Oncoprotein MDM2 conceals the activation domain of tumour suppressor p53. Nature 362, 857–860 (1993).
Haupt, Y., Maya, R., Kazaz, A. & Oren, M. Mdm2 promotes the rapid degradation of p53. Nature 387, 296–299 (1997).
Kubbutat, M. H., Jones, S. N. & Vousden, K. H. Regulation of p53 stability by Mdm2. Nature 387, 299–303 (1997).
Oren, M. Regulation of the p53 tumor suppressor protein. J. Biol. Chem. 274, 36031–36034 (1999).
Yu, J. et al. Identification and classification of p53-regulated genes. Proc. Natl Acad. Sci. USA 96, 14517–14522 (1999).
Zhao, R. et al. Analysis of p53-regulated gene expression patterns using oligonucleotide arrays. Genes Dev. 14, 981–993 (2000).
Ashcroft, M., Taya, Y. & Vousden, K. H. Stress signals utilize multiple pathways to stabilize p53. Mol. Cell. Biol. 20, 3224–3233 (2000).
Shieh, S. Y., Ikeda, M., Taya, Y. & Prives, C. DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 91, 325–334 (1997).
Unger, T. et al. Critical role for Ser20 of human p53 in the negative regulation of p53 by Mdm2. EMBO J. 18, 1805–1814 (1999).
Fuchs, S. Y., Fried, V. A. & Ronai, Z. Stress-activated kinases regulate protein stability. Oncogene 17, 1483–1490 (1998).
Appella, E. & Anderson, C. W. Post-translational modifications and activation of p53 by genotoxic stresses. Eur. J. Biochem. 268, 2764–2772 (2001).
Hirao, A. et al. DNA damage-induced activation of p53 by the checkpoint kinase Chk2 Science 287, 1824–1827 (2000).
Shieh, S. Y., Ahn, J., Tamai, K., Taya, Y. & Prives, C. The human homologs of checkpoint kinases Chk1 and Cds1 (Chk2) phosphorylate p53 at multiple DNA damage-inducible sites. Genes Dev. 14, 289–300 (2000).
Lakin, N. D., Hann, B. C. & Jackson, S. P. The ataxia-telangiectasia related protein ATR mediates DNA-dependent phosphorylation of p53. Oncogene 18, 3989–3995 (1999).
Burma, S. et al. DNA-dependent protein kinase-independent activation of p53 in response to DNA damage. J. Biol. Chem. 274, 17139–17143 (1999).
Nakagawa, K., Taya, Y., Tamai, K. & Yamaizumi, M. Requirement of ATM in phosphorylation of the human p53 protein at serine 15 following DNA double-strand breaks. Mol. Cell. Biol. 19, 2828–2834 (1999).
de Stanchina, E. et al. E1A signaling to p53 involves the p19(ARF) tumor suppressor. Genes Dev. 12, 2434–2442 (1998).
Bulavin, D. V. et al. Phosphorylation of human p53 by p38 kinase coordinates N-terminal phosphorylation and apoptosis in response to UV radiation. EMBO J. 18, 6845–6854 (1999).
Oda, K. et al. p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by Ser-46-phosphorylated p53. Cell 102, 849–862 (2000).
Liu, L. et al. p53 sites acetylated in vitro by PCAF and p300 are acetylated in vivo in response to DNA damage. Mol. Cell. Biol. 19, 1202–1209 (1999).
Ito, A. et al. p300/CBP-mediated p53 acetylation is commonly induced by p53-activating agents and inhibited by MDM2. EMBO. J. 20, 1331–1340 (2001).
Gu, W. & Roeder, R. G. Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 90, 595–606 (1997).
Sakaguchi et al. DNA damage activates p53 through a phosphorylation–acetylation cascade. Genes Dev. 12, 2831–2841 (1998).
Luo, J., Su, F., Chen, D., Shiloh, A. & Gu, W. Deacetylation of p53 modulates its effect on cell growth and apoptosis. Nature 408, 377–381 (2000).
Ferbeyre, G. et al. PML is induced by oncogenic ras and promotes premature senescence. Genes Dev. 14, 2015–2027 (2000).
Fogal, V. et al. Regulation of p53 activity in nuclear bodies by a specific PML isoform. EMBO J. 19, 6185–6195 (2000).
Guo, A. et al. The function of PML in p53-dependent apoptosis. Nature Cell Biol. 10, 730–736 (2000).
Pearson, M. et al. PML regulates p53 acetylation and premature senescence induced by oncogenic Ras. Nature 406, 207–210 (2000).
Hofmann, T. G., Mincheva, A., Lichter, P., Dröge, W. & Schmitz, M. L. Human homeodomain-interacting protein kinase-2 (HIPK2) is a member of the DYRK family of protein kinases and maps to chromosome 7q32–q34. Biochimie 82, 1123–1127 (2000).
Kim, Y. H., Choi, C. Y., Lee, S. J., Conti, M. A. & Kim, Y. Homeodomain-interacting protein kinases, a novel family of co-repressors for homeodomain transcription factors. J. Biol. Chem. 273, 25875–25879 (1998).
Trost, M., Kochs, G. & Haller, O. Characterization of a novel serine/threonine kinase associated with nuclear bodies. J. Biol. Chem. 275, 7373–7377 (2000).
LaMorte, V. J., Dyck, J. A., Ochs, R. L. & Evans, R. M. Localization of nascent RNA and CREB binding protein with the PML-containing nuclear body. Proc. Natl Acad. Sci. USA 95, 4991–4996 (1998).
Doucas, V., Tini, M., Egan, D. A. & Evans, R. M. Modulation of CREB binding protein function by the promyelocytic (PML) oncoprotein suggests a role for nuclear bodies in hormone signaling. Proc. Natl Acad. Sci. USA 96, 2627–2632 (1999).
el-Deiry, W. S. et al. WAF1, a potential mediator of p53 tumor suppression. Cell 75, 817–825 (1993).
Gottifredi, V. & Prives, C. P53 and PML: new partners in tumor suppression. Trends Cell Biol. 11, 184–187 (2001).
Maul, G. G., Negorev, D., Bell, P. & Ishov, A. M. Properties and assembly mechanisms of ND10, PML bodies, or PODs. J. Struct. Biol. 129, 278–287 (2000).
Boisvert, F.M., Kruhlak, M. J., Box, A. K., Hendzel, M. J. & Bazett-Jones, D. P. The transcription coactivator CBP is a dynamic component of the promyelocytic leukemia nuclear body. J. Cell Biol. 152, 1099–1106 (2001).
Boisvert, F. M., Hendzel, M. J. & Bazett-Jones, D. P. Promyelocytic leukemia (PML) nuclear bodies are protein structures that do not accumulate RNA. J. Cell Biol. 148, 283–292 (2000).
Kögel, D., Plottner, O., Landsberg, G., Christian, S. & Scheidtmann, K. H. Cloning and characterization of Dlk, a novel serine/threonine kinase that is tightly associated with chromatin and phosphorylates core histones. Oncogene 17, 2645–2654 (1998).
Sanchez-Prieto, R., Rojas, J. M., Taya, Y. & Gutkind, J. S. A role for the p38 mitogen-activated protein kinase pathway in the transcriptional activation of p53 on genotoxic stress by chemotherapeutic agents. Cancer Res. 60, 2464–2472 (2000).
Higashimoto, Y. et al. Human p53 is phosphorylated on serines 6 and 9 in response to DNA damage-inducing agents. J. Biol. Chem. 275, 23199–23203 (2000).
Bean, L.-J. & Stark, G.-R. Phosphorylation of serines 15 and 37 is necessary for efficient accumulation of p53 following irradiation with UV. Oncogene 20, 1076–1084 (2001).
Sternsdorf, T. et al. PIC-1/SUMO-1-modified PML-retinoic acid receptor alpha mediates arsenic trioxide-induced apoptosis in acute promyelocytic leukemia. Mol. Cell. Biol. 19, 5170–5178 (1999).
Kern, S. E. et al. Oncogenic forms of p53 inhibit p53-regulated gene expression. Science 256, 827–830 (1992).
Delerive, P. et al. Peroxisome proliferator-activated receptor alpha negatively regulates the vascular inflammatory gene response by negative cross-talk with transcription factors NF-κB and AP-1. J. Biol. Chem. 274, 32048–32054 (1999).
Hehner, S. P. et al. Mixed-lineage kinase 3 delivers CD3/CD28-derived signals into the IκB kinase complex. Mol. Cell. Biol. 20, 2556–2568 (2000).
Ausubel, F. M. et al. (eds) Short Protocols in Molecular Biology (Greene Publishing Associates, New York, NY, 1992).
Acknowledgements
We are grateful to S. Soddu for sharing results before publication, E. Appella and L. Florin for helpful comments on the manuscript, T. Hamid and N. Stephan for excellent technical assistance, P. Gutwein for help with microscopy, and all colleagues who generously provided plasmids and reagents: W. Gu, G. Haegeman, M. Oren, B. Vogelstein, G. del Sal, G. Blandino and A. J. Fornace Jr. Our laboratories are supported by grants from the EU (QLK3-CT-2000-00463), Deutsche Krebshilfe, Deutsche Forschungsgemeinschaft (Schm 1417/3-1), Universität Heidelberg and Fonds der chemischen Industrie. The HPI is supported by the Freie und Hansestadt Hamburg and the Bundesministerium für Gesundheit. Correspondence and requests for materials should be addressed to M.L.S.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary figures
Figure S1 HIPK2-mediated phosphorylation of p53. (PDF 175 kb)
Figure S2 UV-radiation does not alter endogenous HIPK2 levels.
Rights and permissions
About this article
Cite this article
Hofmann, T., Möller, A., Sirma, H. et al. Regulation of p53 activity by its interaction with homeodomain-interacting protein kinase-2. Nat Cell Biol 4, 1–10 (2002). https://doi.org/10.1038/ncb715
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb715
This article is cited by
-
Low glucose metabolite 3-phosphoglycerate switches PHGDH from serine synthesis to p53 activation to control cell fate
Cell Research (2023)
-
Deciphering the acetylation code of p53 in transcription regulation and tumor suppression
Oncogene (2022)
-
Sirt6 attenuates chondrocyte senescence and osteoarthritis progression
Nature Communications (2022)
-
DNA damage response revisited: the p53 family and its regulators provide endless cancer therapy opportunities
Experimental & Molecular Medicine (2022)
-
miR-4653-3p overexpression is associated with a poor prognosis of pancreatic ductal adenocarcinoma via HIPK2 downregulation
Scientific Reports (2022)