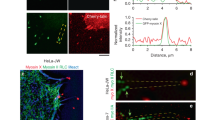
Abstract
Filopodia are thin cellular protrusions that are important in cell motility and neuronal growth cone guidance. The actin filaments that make up the core of a filopodium undergo continuous retrograde flow towards the cell body1,2. Surface receptors or particles can couple to this retrograde flow3,4 and can also move forward to the tips of filopodia4,5, although the molecular basis of forward transport is unknown. We report here that myosin-X (Myo10 or M10), the founding member of a novel class of myosins6, localizes to the tips of filopodia and undergoes striking forward and rearward movements within filopodia, which we term intrafilopodial motility. The movements of the GFP–M10 puncta correspond to forward and rearward movements of phase-dense granules along the filopodia. Finally, overexpressing full-length M10 (but not truncated forms of M10) causes an increase in the number and length of filopodia, indicating that M10 or its cargo may function in filopodial dynamics. The localization and movements of M10 strongly suggest that it functions as a motor for intrafilopodial motility.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Mitchison, T. & Kirschner, M. Neuron 1, 761–772 (1988).
Forscher, P. & Smith, S. J. J. Cell Biol. 107, 1505–1516 (1988).
Felsenfeld, D. P., Choquet, D. & Sheetz, M. P. Nature 383, 438–440 (1996).
Grabham, P. W., Foley, M., Umeojiako, A. & Goldberg, D. J. J. Cell Sci. 113, 3003–3012 (2000).
Sheetz, M. P., Baumrind, N. L., Wayne, D. B. & Pearlman, A. L. Cell 61, 231–241 (1990).
Berg, J. S., Derfler, B. H., Pennisi, C. M., Corey, D. P. & Cheney, R. E. J. Cell Sci. 113, 3439–3451 (2000).
Mallavarapu, A. & Mitchison, T. J. Cell Biol. 146, 1097–1106 (1999).
Sheetz, M. P., Wayne, D. B. & Pearlman, A. L. Cell Motil. Cytoskeleton 22, 160–169 (1992).
Lin, C. H., Espreafico, E. M., Mooseker, M. S. & Forscher, P. Neuron 16, 769–782 (1996).
Isakoff, S. J. et al. EMBO J. 17, 5374–5387 (1998).
Yonemura, S. et al. J. Cell Biol. 140, 885–895 (1998).
Chen, L. C., Wang, F., Meng, H., Corey, D. P. & Sellers, J. R. Mol. Biol. Cell 10s, 152a (1999).
Homma, K., Saito, J., Ikebe, R. & Ikebe, M. J. Biol. Chem. 276, 34348–34354 (2001).
Mitchison, T. J. Cell Motil. Cytoskeleton 22, 135–151 (1992).
Ramirez-Weber, F. A. & Kornberg, T. B. Cell 97, 599–607 (1999).
Bryant, P. J. Curr. Biol. 9, R655–R657 (1999).
Tsakraklides, V. et al. J. Cell Sci. 112, 2853–2865 (1999).
Tang, N. & Ostap, E. M. Curr. Biol. 11, 1131–1135 (2001).
Moores, S. L., Sabry, J. H. & Spudich, J. A. Proc. Natl Acad. Sci. USA 93, 443–446 (1996).
Wei, Q. & Adelstein, R. S. Mol. Biol. Cell 11, 3617–3627 (2000).
Berg, J. S., Powell, B. C. & Cheney, R. E. Mol. Biol. Cell 12, 780–794 (2001).
Titus, M. A. Curr. Biol. 9, 1297–1303 (1999).
Tuxworth, R. I. et al. Curr. Biol. 11, 318–329 (2001).
Cox, D., Berg, J., Dale, B., Cheney, R. & Greenberg, S., Molecular Biology of the Cell 11, 375a (2000).
Nobes, C. D. & Hall, A. Cell 81, 53–62 (1995).
Borisy, G. G. & Svitkina, T. M. Curr. Opin. Cell Biol. 12, 104–112 (2000).
Lee, W. L., Bezanilla, M. & Pollard, T. D. J. Cell Biol. 151, 789–800 (2000).
Lanier, L. M. et al. Neuron 22, 313–325 (1999).
Wu, D. Y. & Goldberg, D. J. J. Cell Biol. 123, 653–664 (1993).
Rosenbaum, J. L., Cole, D. G. & Diener, D. R. J. Cell Biol. 144, 385–388 (1999).
Acknowledgements
The authors thank A. Stuart for use of the 63 × phase lens used in this work and D. Moore (Lineberger Comprehensive Cancer Center Biostatistics Core Facility) for help with statistical analysis. We also thank T. Salmon, D. Kashatus, and O. Rodriguez for helpful discussions. This work was supported by National Institutes of Health grant DC03299 to R.E.C.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Movie 1
This example demonstrates numerous instances of filopodial extension and retraction with GFP:M10 localized to the tip. (MOV 577 kb)
Movie 2
This example demonstrates extension and retraction of filopodia with GFP:M10 at the tip, as well as several instances of intrafilopodial motility within filopodia that remain attached to the substrate. (MOV 1397 kb)
Movie 3
This example demonstrates extension and retraction of filopodia with GFP:M10 at the tip, as well as several instances of intrafilopodial motility within filopodia that remain attached to the substrate. (MOV 1620 kb)
Rights and permissions
About this article
Cite this article
Berg, J., Cheney, R. Myosin-X is an unconventional myosin that undergoes intrafilopodial motility. Nat Cell Biol 4, 246–250 (2002). https://doi.org/10.1038/ncb762
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb762
This article is cited by
-
Optogenetic manipulation of cellular communication using engineered myosin motors
Nature Cell Biology (2021)
-
Targeting the cytoskeleton against metastatic dissemination
Cancer and Metastasis Reviews (2021)
-
Phenotypic analysis of Myo10 knockout (Myo10tm2/tm2) mice lacking full-length (motorized) but not brain-specific headless myosin X
Scientific Reports (2019)
-
Myosin IIA and formin dependent mechanosensitivity of filopodia adhesion
Nature Communications (2019)