Abstract
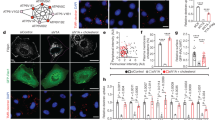
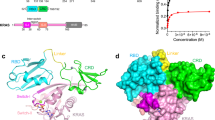
Current models evoke the plasma membrane (PM) as the exclusive platform from which Ras regulates signalling. We developed a fluorescent probe that reports where and when Ras is activated in living cells. We show that oncogenic H-Ras and N-Ras engage Raf-1 on the Golgi and that endogenous Ras and unpalmitoylated H-Ras are activated in response to mitogens on the Golgi and endoplasmic reticulum (ER), respectively. We also demonstrate that H-Ras that is restricted to the ER can activate the Erk pathway and transform fibroblasts, and that Ras localized on different membrane compartments differentially engages various signalling pathways. Thus, Ras signalling is not limited to the PM, but also proceeds on the endomembrane.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Di Fiore, P. P. & Gill, G. N. Endocytosis and mitogenic signalling. Curr. Opin. Cell Biol. 11, 483–488 (1999).
Vieira, A. V., Lamaze, C. & Schmid, S. L. Control of EGF receptor signalling by clathrin-mediated endocytosis. Science 274, 2086–2089 (1996).
Willingham, M. C., Pastan, I., Shih, T. Y. & Scolnick, E. M. Localization of the src gene product of the Harvey strain of MSV to plasma membrane of transformed cells by electron microscopic immunocytochemistry. Cell 19, 1005–1014 (1980).
Willumsen, B. M., Norris, K., Papageorge, A. G., Hubbert, N. L. & Lowy, D. R. Harvey murine sarcoma virus p21 ras protein: biological and biochemical significance of the cysteine nearest the carboxy terminus. EMBO J. 3, 2581–2585 (1984).
Lowenstein, E. J. et al. The SH2 and SH3 Domain-containing Protein GRB2 Links Receptor Tyrosine Kinases to ras Signalling. Cell 70, 431–442 (1992).
Leevers, S. J., Paterson, H. F. & Marshall, C. J. Requirement for Ras in Raf activation is overcome by targeting Raf to the plasma membrane. Nature 369, 411–414 (1994).
Stokoe, D., Macdonald, S. G., Cadwallader, K., Symons, M. & Hancock, J. F. Activation of raf as a result of recruitment to the plasma membrane. Science 264, 1463–1467 (1994).
Clarke, S. Protein isoprenylation and methylation at carboxyl terminal cysteine residues. Annu. Rev. Biochem. 61, 355–386 (1992).
Hancock, J. F., Paterson, H. & Marshall, C. J. A polybasic domain or palmitoylation is required in addition to the CAAX motif to localize p21ras to the plasma membrane. Cell 63, 133–139 (1990).
Choy, E. et al. Endomembrane trafficking of ras: the CAAX motif targets proteins to the ER and Golgi. Cell 98, 69–80 (1999).
Schmidt, W. K., Tam, A., Fujimura-Kamada, K. & Michaelis, S. Endoplasmic reticulum membrane localization of Rce1p and Ste24p, yeast proteases involved in carboxyl-terminal CAAX protein processing and amino-terminal α-factor cleavage. Proc. Natl Acad. Sci. USA 95, 11175–11180 (1998).
Dai, Q. et al. Mammalian prenylcysteine carboxyl methyltransferase is in the endoplasmic reticulum. J. Biol. Chem. 273, 15030–15034 (1998).
Apolloni, A., Prior, I. A., Lindsay, M., Parton, R. G. & Hancock, J. F. H-ras but not K-ras traffics to the plasma membrane through the exocytic pathway. Mol. Cell Biol. 20, 2475–2487 (2000).
Herrmann, C., Horn, G., Spaargaren, M. & Wittinghofer, A. Differential interaction of the ras family GTP-binding proteins H-Ras, Rap1A, and R-Ras with the putative effector molecules Raf kinase and Ral-guanine nucleotide exchange factor. J. Biol. Chem. 271, 6794–6800 (1996).
Bastiaens, P. I., Majoul, I. V., Verveer, P. J., Soling, H. D. & Jovin, T. M. Imaging the intracellular trafficking and state of the AB5 quaternary structure of cholera toxin. EMBO J. 15, 4246–4253 (1996).
Sorkin, A., McClure, M., Huang, F. & Carter, R. Interaction of EGF receptor and grb2 in living cells visualized by fluorescence resonance energy transfer (FRET) microscopy. Curr. Biol. 10, 1395–1398 (2000).
Chen, H. et al. Epsin is an EH-domain-binding protein implicated in clathrin-mediated endocytosis. Nature 394, 793–797 (1998).
Punnonen, E. L., Ryhanen, K. & Marjomaki, V. S. At reduced temperature, endocytic membrane traffic is blocked in multivesicular carrier endosomes in rat cardiac myocytes. Eur. J. Cell Biol. 75, 344–352 (1998).
Bjorge, J. D., Jakymiw, A. & Fujita, D. J. Selected glimpses into the activation and function of Src kinase. Oncogene 19, 5620–5635 (2000).
Cadwaller, K. A., Paterson, H., Macdonald, S. G. & Hancock, J. F. N-terminally myristoylated ras proteins require palmitoylation or a polybasic domain for plasma membrane localization. Mol. Cell. Biol. 14, 4722–4730 (1994).
Swift, A. M. & Machamer, C. E. A Golgi retention signal in a membrane-spanning domain of coronavirus E1 protein. J. Cell Biol. 115, 19–30 (1991).
Willumsen, B. M., Christensen, A., Hubbert, N. L., Papageorge, A. G. & Lowy, D. R. The p21 ras C-terminus is required for transformation and membrane association. Nature 310, 583–586 (1984).
Voice, J. K., Klemke, R. L., Le, A. & Jackson, J. H. Four human ras homologs differ in their abilities to activate Raf-1, induce transformation, and stimulate cell motility. J. Biol. Chem. 274, 17164–17170 (1999).
Yan, J., Roy, S., Apolloni, A., Lane, A. & Hancock, J. F. Ras isoforms vary in their ability to activate Raf-1 and phosphoinositide 3-kinase. J. Biol. Chem. 273, 24052–24056 (1998).
Walsh, A. B. & Bar-Sagi, D. Differential activation of the Rac pathway by Ha-Ras and K-Ras. J. Biol. Chem. 276, 15609–15615 (2001).
Mochizuki, N. et al. Spatio-temporal images of growth-factor-induced activation of Ras and Rap1. Nature 411, 1065–1068 (2001).
Marshall, C. J. Specificity of receptor tyrosine kinase signalling: transient versus sustained extracellular signal-regulated kinase activation. Cell 80, 179–185 (1995).
Ebinu, J. O. et al. RasGRP, a Ras guanyl nucleotide- releasing protein with calcium- and diacylglycerol-binding motifs. Science 280, 1082–1086 (1998).
Lorenzo, P. S. et al. Phorbol esters modulate the Ras exchange factor RasGRP3. Cancer Res. 61, 943–949 (2001).
Liao, F., Shin, H. S. & Rhee, S. G. In vitro tyrosine phosphorylation of PLC-γ1 and PLC-γ2 by src- family protein tyrosine kinases. Biochem. Biophys. Res. Commun. 191, 1028–1033 (1993).
Hart, K. C., Robertson, S. C. & Donoghue, D. J. Activation of H-ras61L-specific signalling pathways does not require posttranslational processing of H-ras. Exp. Cell Res. 257, 89–100 (2000).
Klinghoffer, R. A., Sachsenmaier, C., Cooper, J. A. & Soriano, P. Src family kinases are required for integrin but not PDGFR signal transduction. EMBO J. 18, 2459–2471 (1999).
Oancea, E. & Meyer, T. Protein kinase C as a molecular machine for decoding calcium and diacylglycerol signals. Cell 95, 307–318 (1998).
Foos, G., Garcia-Ramirez, J. J., Galang, C. K. & Hauser, C. A. Elevated expression of Ets2 or distinct portions of Ets2 can reverse Ras-mediated cellular transformation. J. Biol. Chem. 273, 18871–18880 (1998).
Cox, A. D. & Der, C. J. Biological assays for cellular transformation. Methods Enzymol. 238, 277–294 (1994).
Acknowledgements
We thank P. DeCamilli, C. Hauser, J. Lippincott-Schwartz, D. Littman and C. Machamer for plasmids, A. Kenworthy for insightful discussions and M. Feoktistov for her excellent technical assistance. We also thank the late and ever-inspiring T. Morimoto for his assistance, advice and unwavering encouragement. This work was supported by National Institutes of Health grants AI36224 and GM55279 and by the Burroughs Wellcome Fund to M.R.P., by NIH grants CA67771 and CA76092 to A.D.C. and a General Clinical Research Center NIH grant, NCRR (M01RR00096).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
41556_2002_BFncb783_MOESM1_ESM.mov
Movie 1 Rapid Activation of Palmitoylation-deficient H-Ras on Endomembrane in Response to EGF Receptor Stimulation. (MOV 1480 kb)
Rights and permissions
About this article
Cite this article
Chiu, V., Bivona, T., Hach, A. et al. Ras signalling on the endoplasmic reticulum and the Golgi. Nat Cell Biol 4, 343–350 (2002). https://doi.org/10.1038/ncb783
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb783
This article is cited by
-
Computer-based design of sensors that can monitor endogenous Ras activity
Nature Biotechnology (2024)
-
CD40 induces selective routing of Ras isoforms to subcellular compartments
Journal of Cell Communication and Signaling (2023)
-
The role of ral signaling and post translational modifications (PTMs) of Ras in cancer
Genome Instability & Disease (2022)
-
Post-translational modification of KRAS: potential targets for cancer therapy
Acta Pharmacologica Sinica (2021)
-
Spatial regulation and generation of diversity in signaling pathways
Journal of Biosciences (2021)