Abstract
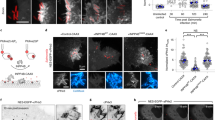
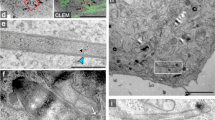
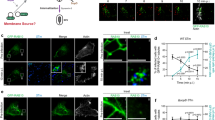
Salmonella invades mammalian cells by inducing membrane ruffling and macropinocytosis through actin remodelling. Because phosphoinositides are central to actin assembly, we have studied the dynamics of phosphatidylinositol-4,5-bisphosphate (PtdIns(4,5)P2) in HeLa cells during invasion by Salmonella typhimurium. Here we show that the outermost parts of the ruffles induced by invasion show a modest enrichment in PtdIns(4,5)P2, but that PtdIns(4,5)P2 is virtually absent from the invaginating regions. Rapid disappearance of PtdIns(4,5)P2 requires the expression of the Salmonella phosphatase SigD (also known as SopB). Deletion of SigD markedly delays fission of the invaginating membranes, indicating that elimination of PtdIns(4,5)P2 may be required for rapid formation of Salmonella-containing vacuoles. Heterologous expression of SigD is sufficient to promote the disappearance of PtdIns(4,5)P2, to reduce the rigidity of the membrane skeleton, and to induce plasmalemmal invagination and fission. Hydrolysis of PtdIns(4,5)P2 may be a common and essential feature of membrane fission during several internalization processes including invasion, phagocytosis and possibly endocytosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Parker, C., Asokan, K. & Guard-Petter, J. Egg contamination by Salmonella serovar enteritidis following vaccination with δ-aroA Salmonella serovar typhimurium. FEMS Microbiol. Lett. 195, 73–78 (2001).
Dworkin, M. S., Shoemaker, P. C., Goldoft, M. J. & Kobayashi, J. M. Reactive arthritis and Reiter's syndrome following an outbreak of gastroenteritis caused by Salmonella enteritidis. Clin. Infect. Dis. 33, 1010–1014 (2001).
Galan, J. E. & Zhou, D. Striking a balance: modulation of the actin cytoskeleton by Salmonella. Proc. Natl Acad. Sci. USA 97, 8754–8761 (2000).
Friebel, A. et al. SopE and SopE2 from Salmonella typhimurium activate different sets of Rho GTPases of the host cell. J Biol Chem 276, 34035–40. (2001).
Zhou, D., Chen, L. M., Hernandez, L., Shears, S. B. & Galan, J. E. A Salmonella inositol polyphosphatase acts in conjunction with other bacterial effectors to promote host cell actin cytoskeleton rearrangements and bacterial internalization. Mol. Microbiol. 39, 248–259 (2001).
Marcus, S. L., Wenk, M. R., Steele-Mortimer, O. & Finlay, B. B. A synaptojanin-homologous region of Salmonella typhimurium SigD is essential for inositol phosphatase activity and Akt activation. FEBS Lett. 494, 201–207 (2001).
Fu, Y. & Galan, J. E. A salmonella protein antagonizes Rac-1 and Cdc42 to mediate host-cell recovery after bacterial invasion. Nature 401, 293–297 (1999).
Stebbins, C. E. & Galan, J. E. Modulation of host signaling by a bacterial mimic: structure of the Salmonella effector SptP bound to Rac1. Mol. Cell 6, 1449–1460 (2000).
Honda, A. et al. Phosphatidylinositol 4-phosphate 5-kinase α is a downstream effector of the small G protein ARF6 in membrane ruffle formation. Cell 99, 521–532 (1999).
Rohatgi, R. et al. The interaction between N-WASP and the Arp2/3 complex links Cdc42- dependent signals to actin assembly. Cell 97, 221–231 (1999).
Chen, F. et al. Cdc42 is required for PIP2-induced actin polymerization and early development but not for cell viability. Curr. Biol. 10, 758–765 (2000).
Sechi, A. S. & Wehland, J. The actin cytoskeleton and plasma membrane connection: PtdIns(4,5)P2 influences cytoskeletal protein activity at the plasma membrane. J. Cell Sci. 113, 3685–3695 (2000).
Norris, F. A., Wilson, M. P., Wallis, T. S., Galyov, E. E. & Majerus, P. W. SopB, a protein required for virulence of Salmonella dublin, is an inositol phosphate phosphatase. Proc. Natl Acad. Sci. USA 95, 14057–14059 (1998).
Stauffer, T. P., Ahn, S. & Meyer, T. Receptor-induced transient reduction in plasma membrane PtdIns(4,5)P2 concentration monitored in living cells. Curr. Biol. 8, 343–346 (1998).
Varnai, P. & Balla, T. Visualization of phosphoinositides that bind pleckstrin homology domains: calcium- and agonist-induced dynamic changes and relationship to myo-[3H]inositol-labeled phosphoinositide pools. J. Cell Biol. 143, 501–510 (1998).
Raucher, D. et al. Phosphatidylinositol 4,5-bisphosphate functions as a second messenger that regulates cytoskeleton-plasma membrane adhesion. Cell 100, 221–228 (2000).
Teruel, M. N., Blanpied, T. A., Shen, K., Augustine, G. J. & Meyer, T. A versatile microporation technique for the transfection of cultured CNS neurons. J. Neurosci. Methods 93, 37–48 (1999).
Criss, A. K., Ahlgren, D. M., Jou, T. S., McCormick, B. A. & Casanova, J. E. The GTPase Rac1 selectively regulates Salmonella invasion at the apical plasma membrane of polarized epithelial cells. J. Cell Sci. 114, 1331–1341 (2001).
Tolias, K. & Carpenter, C. L. In vitro interaction of phosphoinositide-4-phosphate 5-kinases with Rac. Methods Enzymol. 325, 190–200 (2000).
Weernink, P. A. et al. Control of cellular phosphatidylinositol 4,5-bisphosphate levels by adhesion signals and rho GTPases in NIH 3T3 fibroblasts involvement of both phosphatidylinositol-4-phosphate 5-kinase and phospholipase C. Eur. J. Biochem. 267, 5237–5246 (2000).
Jepson, M. A., Kenny, B. & Leard, A. D. Role of sipA in the early stages of Salmonella typhimurium entry into epithelial cells. Cell Microbiol. 3, 417–426 (2001).
Steele-Mortimer, O. et al. Activation of Akt/protein kinase B in epithelial cells by the Salmonella typhimurium effector sigD. J. Biol. Chem. 275, 37718–37724 (2000).
Kontos, C. D. et al. Tyrosine 1101 of Tie2 is the major site of association of p85 and is required for activation of phosphatidylinositol 3-kinase and Akt. Mol. Cell Biol. 18, 4131–4140 (1998).
Watton, S. J. & Downward, J. Akt/PKB localisation and 3′ phosphoinositide generation at sites of epithelial cell–matrix and cell–cell interaction. Curr. Biol. 9, 433–436 (1999).
Takahashi, A. et al. Production of β-defensin-2 by human colonic epithelial cells induced by Salmonella enteritidis flagella filament structural protein. FEBS Lett. 508, 484–488 (2001).
Gewirtz, A. T. et al. Salmonella typhimurium induces epithelial IL-8 expression via Ca2+- mediated activation of the NF-κB pathway. J. Clin. Invest. 105, 79–92 (2000).
Ruschkowski, S., Rosenshine, I. & Finlay, B. B. Salmonella typhimurium induces an inositol phosphate flux in infected epithelial cells. FEMS Microbiol. Lett. 74, 121–126 (1992).
Pace, J., Hayman, M. J. & Galan, J. E. Signal transduction and invasion of epithelial cells by S. typhimurium. Cell 72, 505–514 (1993).
Oancea, E., Teruel, M. N., Quest, A. F. & Meyer, T. Green fluorescent protein (GFP)-tagged cysteine-rich domains from protein kinase C as fluorescent indicators for diacylglycerol signaling in living cells. J. Cell Biol. 140, 485–498 (1998).
Botelho, R. J. et al. Localized biphasic changes in phosphatidylinositol-4,5-bisphosphate at sites of phagocytosis. J. Cell Biol. 151, 1353–1368 (2000).
Chen, P., Xie, H. & Wells, A. Mitogenic signaling from the EGF receptor is attenuated by a phospholipase C-γ/protein kinase C feedback mechanism. Mol. Biol. Cell 7, 871–881 (1996).
Eckmann, L. et al. D-myo-Inositol 1,4,5,6-tetrakisphosphate produced in human intestinal epithelial cells in response to Salmonella invasion inhibits phosphoinositide 3-kinase signaling pathways. Proc. Natl Acad. Sci. USA 94, 14456–60. (1997).
Feng, Y., Wente, S. R. & Majerus, P. W. Overexpression of the inositol phosphatase SopB in human 293 cells stimulates cellular chloride influx and inhibits nuclear mRNA export. Proc. Natl Acad. Sci. USA 98, 875–879 (2001).
Balla, T., Bondeva, T. & Varnai, P. How accurately can we image inositol lipids in living cells? Trends Pharmacol. Sci. 21, 238–241 (2000).
Surprenant, A., Rassendren, F., Kawashima, E., North, R. A. & Buell, G. The cytolytic P2Z receptor for extracellular ATP identified as a P2X receptor (P2X7). Science 272, 735–738 (1996).
Laux, T. et al. GAP43, MARCKS, and CAP23 modulate PtdIns(4,5)P2 at plasmalemmal rafts, and regulate cell cortex actin dynamics through a common mechanism. J. Cell Biol. 149, 1455–1472 (2000).
Sheetz, M. P. Cell control by membrane-cytoskeleton adhesion. Nature Rev. Mol. Cell Biol. 2, 392–396 (2001).
A-Hassan E. et al. Relative microelastic mapping of living cells by atomic force microscopy. Biophys. J. 74, 1564–1578 (1998).
Galyov, E. E. et al. A secreted effector protein of Salmonella dublin is translocated into eukaryotic cells and mediates inflammation and fluid secretion in infected ileal mucosa. Mol. Microbiol. 25, 903–912 (1997).
Jost, M., Simpson, F., Kavran, J. M., Lemmon, M. A. & Schmid, S. L. Phosphatidylinositol-4,5-bisphosphate is required for endocytic coated vesicle formation. Curr. Biol. 8, 1399–1402 (1998).
Garcia-del Portillo, F., Pucciarelli, M. G., Jefferies, W. A. & Finlay, B. B. Salmonella typhimurium induces selective aggregation and internalization of host cell surface proteins during invasion of epithelial cells. J. Cell Sci. 107, 2005–2020 (1994).
Acknowledgements
This work was supported by the Arthritis Society of Canada and the Canadian Institutes for Health Research. M.T. is the recipient of a Hospital for Sick Children Restracomp Fellowship. O.V.V. is the recipient of a postdoctoral fellowship from PRAXIS XX1. B.B.F and S.G. are recipients of Canadian Institutes for Health Research (CIHR) Distinguished Scientist Awards. S.G. is the current holder of the Pitblado Chair in Cell Biology at The Hospital for Sick Children and is cross-appointed to the Department of Biochemistry, University of Toronto. C.M.Y. is the holder of a Canada Research Chair.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Movie 1
Live imaging of HeLa cells during invasion by wildtype Salmonella enterica (serovar Typhimurium). (MOV 2714 kb)
Movie 2
Live imaging of HeLa cells during invasion by mutant Salmonella lacking SopB/SigD. (MOV 2988 kb)
Movie 3
Live imaging of HeLa cells during invasion by mutant Salmonella lacking SopE and SopE2 (MOV 2899 kb)
Rights and permissions
About this article
Cite this article
Terebiznik, M., Vieira, O., Marcus, S. et al. Elimination of host cell PtdIns(4,5)P2 by bacterial SigD promotes membrane fission during invasion by Salmonella. Nat Cell Biol 4, 766–773 (2002). https://doi.org/10.1038/ncb854
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb854
This article is cited by
-
Strategies adopted by Salmonella to survive in host: a review
Archives of Microbiology (2023)
-
Kir2.1-mediated membrane potential promotes nutrient acquisition and inflammation through regulation of nutrient transporters
Nature Communications (2022)
-
Kinase-independent synthesis of 3-phosphorylated phosphoinositides by a phosphotransferase
Nature Cell Biology (2022)
-
Delivering the pain: an overview of the type III secretion system with special consideration for aquatic pathogens
Veterinary Research (2021)
-
Salmonella effector SopD promotes plasma membrane scission by inhibiting Rab10
Nature Communications (2021)