Abstract
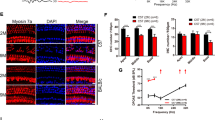
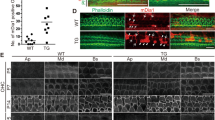
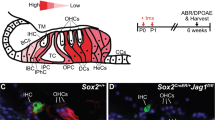
Maintenance of the post-mitotic state in the post-natal mammalian brain is an active process that requires the cyclin-dependent kinase inhibitors (CKIs) p19Ink4d (Ink4d) and p27Kip1 (Kip1)1. In animals with targeted deletions of both Ink4d and Kip1, terminally differentiated, post-mitotic neurons are observed to re-enter the cell cycle, divide and undergo apoptosis. However, when either Ink4d or Kip1 alone are deleted, the post-mitotic state is maintained, suggesting a redundant role for these genes in mature neurons1. In the organ of Corti — the auditory sensory epithelium of mammals — sensory hair cells and supporting cells become post-mitotic during embryogenesis2 and remain quiescent for the life of the animal. When lost as a result of environmental insult or genetic abnormality, hair cells do not regenerate, and this loss is a common cause of deafness in humans3. Here, we report that targeted deletion of Ink4d alone is sufficient to disrupt the maintenance of the post-mitotic state of sensory hair cells in post-natal mice. In Ink4d−/− animals, hair cells are observed to aberrantly re-enter the cell cycle and subsequently undergo apoptosis, resulting in progressive hearing loss. Our results identify a novel mechanism underlying a non-syndromic form of progressive hearing loss in mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zindy, F. et al. Post-natal neuronal proliferation in mice lacking Ink4d and Kip1 inhibitors of cyclin-dependent kinases. Proc. Natl Acad. Sci. USA 96, 13462–13467 (1999).
Ruben, R.J. Development of the inner ear of the mouse: a radioautographic study of terminal mitoses. Acta Otolaryngol Suppl. 1–44 (1967).
Schuknecht, H.F. Pathology Of The Ear (Lea and Febiger, Philadelphia, 1993).
Chen, P., Johnson, J.E., Zoghbi, H.Y. & Segil, N. The role of Math1 in inner ear development: Uncoupling the establishment of the sensory primordium from hair cell fate determination. Development 129, 2495–2505 (2002).
Chen, P. & Segil, N. p27(Kip1) links cell proliferation to morphogenesis in the developing organ of Corti. Development 126, 1581–1590 (1999).
Schrott, A., Puel, J.L. & Rebillard, G. Cochlear origin of 2f1-f2 distortion products assessed by using 2 types of mutant mice. Hear. Res. 52, 245–253 (1991).
Zheng, Q.Y., Johnson, K.R. & Erway, L.C. Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hear. Res. 130, 94–107 (1999).
Jimenez, A.M., Stagner, B.B., Martin, G.K. & Lonsbury-Martin, B.L. Age-related loss of distortion product otoacoustic emissions in four mouse strains. Hear. Res. 138, 91–105 (1999).
Ohlemiller, K.K. Reduction in sharpness of frequency tuning but not endocochlear potential in aging and noise-exposed BALB/cJ Mice. J. Assoc. Res. Otolaryngol. 3, 444–456 (2002).
Lowenheim, H. et al. Gene disruption of p27(Kip1) allows cell proliferation in the post-natal and adult organ of corti. Proc. Natl Acad. Sci. USA 96, 4084–4088 (1999).
Kouroku, Y. et al. Detection of activated Caspase-3 by a cleavage site-directed antiserum during naturally occurring DRG neurons apoptosis. Biochem. Biophys. Res. Commun. 247, 780–784 (1998).
Liu, D.X. & Greene, L.A. Neuronal apoptosis at the G1/S cell cycle checkpoint. Cell Tissue Res. 305, 217–228 (2001).
Herrup, K. & Yang, Y. Pictures in molecular medicine: contemplating Alzheimer's disease as cancer: a loss of cell-cycle control. Trends Mol. Med. 7, 527 (2001).
Nguyen, M.D., Mushynski, W.E. & Julien, J.P. Cycling at the interface between neurodevelopment and neurodegeneration. Cell Death Differ. 9, 1294–1306 (2002).
Morton, N.E. Genetic epidemiology of hearing impairment. Ann. NY Acad. Sci. 630, 16–31 (1991).
Corwin, J.T. & Cotanche, D.A. Regeneration of sensory hair cells after acoustic trauma. Science 240, 1772–1774 (1988).
Ryals, B.M. & Rubel, E.W. Hair cell regeneration after acoustic trauma in adult Coturnix quail. Science 240, 1774–1776 (1988).
Chardin, S. & Romand, R. Regeneration and mammalian auditory hair cells. Science 267, 707–711 (1995).
Zindy, F., van Deursen, J., Grosveld, G., Sherr, C.J. & Roussel, M.F. Ink4d-deficient mice are fertile despite testicular atrophy. Mol. Cell. Biol. 20, 372–378 (2000).
Fero, M.L. et al. A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27(Kip1)-deficient mice. Cell 85, 733–744 (1996).
Harlow, E. & Lane, D. Antibodies: A Laboratory Manual (Cold Spring Harbor Laboratory, Cold Spring Harbor, NY, 1988).
White, P.M. & Anderson, D.J. In vivo transplantation of mammalian neural crest cells into chick hosts reveals a new autonomic sublineage restriction. Development 126, 4351–4363 (1999).
Acknowledgements
We thank J. Zuo for helpful discussion, A.Groves, A. Bass and J. Johnson for comments on the manuscript, W. den Besten for developing the PCR protocol used for genotyping Ink4d mice, J. Llamos, W. Macmurra, S. Portillo and C. Meunier for assistance in animal care and genotyping, and B. Welch for graphics assistance. We also thank J. Roberts and M. Fero for the p27Kip1-mutant mice, and C. Petit and A. El-Amraoui for the anti-Myosin VIIa antibody used in this study. This work was supported by grants from the National Institutes of Health (to N.S., P.C. and M.F.R.), the American Lebanese-Syrian-associated Charities (ALSAC) of St Jude Children's Research Hospital (to M.F.R.) and the Oberkotter Foundation (to N.S.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Chen, P., Zindy, F., Abdala, C. et al. Progressive hearing loss in mice lacking the cyclin-dependent kinase inhibitor Ink4d. Nat Cell Biol 5, 422–426 (2003). https://doi.org/10.1038/ncb976
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb976
This article is cited by
-
Renewed proliferation in adult mouse cochlea and regeneration of hair cells
Nature Communications (2019)
-
The miR-183/ItgA3 axis is a key regulator of prosensory area during early inner ear development
Cell Death & Differentiation (2017)
-
Rescue from early-onset hearing loss in a mouse model lacking the cyclin-dependent kinase inhibitor p19Ink4d
Cell Death & Disease (2016)
-
Segregating neural and mechanosensory fates in the developing ear: patterning, signaling, and transcriptional control
Cell and Tissue Research (2015)
-
Age-related hearing loss or presbycusis
European Archives of Oto-Rhino-Laryngology (2010)