Abstract
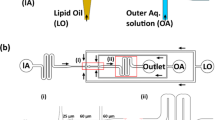
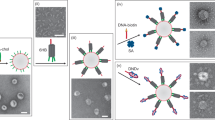
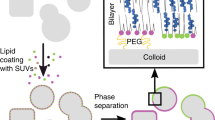
Eukaryotic subcellular membrane systems, such as the nuclear envelope or endoplasmic reticulum, present a rich array of architecturally and compositionally complex supramolecular targets that are as yet inaccessible. Here we describe layer-by-layer phospholipid membrane assembly on microfluidic droplets, a route to structures with defined compositional asymmetry and lamellarity. Starting with phospholipid-stabilized water-in-oil droplets trapped in a static droplet array, lipid monolayer deposition proceeds as oil/water-phase boundaries pass over the droplets. Unilamellar vesicles assembled layer-by-layer support functional insertion both of purified and of in situ expressed membrane proteins. Synthesis and chemical probing of asymmetric unilamellar and double-bilayer vesicles demonstrate the programmability of both membrane lamellarity and lipid-leaflet composition during assembly. The immobilized vesicle arrays are a pragmatic experimental platform for biophysical studies of membranes and their associated proteins, particularly complexes that assemble and function in multilamellar contexts in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
van Meer, G., Voelker, D. R. & Feigenson, G. W. Membrane lipids: where they are and how they behave. Nature Rev. Mol. Cell Biol. 9, 112–124 (2008).
Rosenbaum, D. M., Rasmussen, S. G. F. & Kobilka, B. K. The structure and function of G-protein-coupled receptors. Nature 459, 356–363 (2009).
Peter, B. et al. BAR domains as sensors of membrane curvature: the amphiphysin BAR structure. Science 303, 495–499 (2004).
Hatzakis, N. S. et al. How curved membranes recruit amphipathic helices and protein anchoring motifs. Nature Chem. Biol. 5, 835–841 (2009).
Liu, A. P. & Fletcher, D. A. Biology under construction: in vitro reconstitution of cellular function. Nature Rev. Mol. Cell Biol. 10, 644–650 (2009).
Bangham, A. D., Standish, M. M. & Watkins, J. C. Diffusion of univalent ions across lamellae of swollen phospholipids. J. Mol. Biol. 13, 238–252 (1965).
Jahn, A., Vreeland, W. N., Gaitan, M. & Locascio, L. E. Controlled vesicle self-assembly in microfluidic channels with hydrodynamic focusing. J. Am. Chem. Soc. 126, 2674–2675 (2004).
Shum, H. C., Lee, D., Yoon, I., Kodger, T. & Weitz, D. A. Double emulsion templated monodisperse phospholipid vesicles. Langmuir 24, 7651–7653 (2008).
Stachowiak, J. C. et al. Unilamellar vesicle formation and encapsulation by microfluidic jetting. Proc. Natl Acad. Sci. USA 105, 4697–4702 (2008).
Ota, S., Yoshizawa, S. & Takeuchi, S. Microfluidic formation of monodisperse, cell-sized, and unilamellar vesicles. Angew. Chem. Int. Ed. 48, 6533–6537 (2009).
Richmond, D. L. et al. Forming giant vesicles with controlled membrane composition, asymmetry, and contents. Proc. Natl Acad. Sci. USA 108, 9431–9436 (2011).
Hu, P. C., Li, S. & Malmstadt, N. Microfluidic fabrication of asymmetric giant lipid vesicles. ACS Appl. Mater. Interfaces 3, 1434–1440 (2011).
Pautot, S., Frisken, B. J. & Weitz, D. A. Production of unilamellar vesicles using an inverted emulsion. Langmuir 19, 2870–2879 (2003).
Pautot, S., Frisken, B. & Weitz, D. Engineering asymmetric vesicles. Proc. Natl Acad. Sci. USA 100, 10718–10721 (2003).
Noireaux, V. & Libchaber, A. A vesicle bioreactor as a step toward an artificial cell assembly. Proc. Natl Acad. Sci. USA 101, 17669–17674 (2004).
Matosevic, S. & Paegel, B. M. Stepwise synthesis of giant unilamellar vesicles on a microfluidic assembly line. J. Am. Chem. Soc. 133, 2798–2800 (2011).
Blodgett, K. Films built by depositing successive monomolecular layers on a solid surface. J. Am. Chem. Soc. 57, 1007–1022 (1935).
Hase, M., Yamada, A., Hamada, T. & Yoshikawa, K. Transport of a cell-sized phospholipid micro-container across water/oil interface. Chem. Phys. Lett. 426, 441–444 (2006).
Huebner, A. et al. Static microdroplet arrays: a microfluidic device for droplet trapping, incubation and release for enzymatic and cell-based assays. Lab Chip 9, 692–698 (2009).
Anna, S., Bontoux, N. & Stone, H. Formation of dispersions using ‘flow focusing’ in microchannels. Appl. Phys. Lett. 82, 364–366 (2003).
Baret, J-C., Kleinschmidt, F., Harrak El, A., & Griffiths, A. D. Kinetic aspects of emulsion stabilization by surfactants: a microfluidic analysis. Langmuir 25, 6088–6093 (2009).
Song, L. et al. Structure of staphylococcal alpha-hemolysin, a heptameric transmembrane pore. Science 274, 1859–1866 (1996).
Shimizu, Y. et al. Cell-free translation reconstituted with purified components. Nature Biotechnol. 19, 751–755 (2001).
Tawfik, D. S. & Griffiths, A. Man-made cell-like compartments for molecular evolution. Nature Biotechnol. 16, 652–656 (1998).
Andes-Koback, M. & Keating, C. D. Complete budding and asymmetric division of primitive model cells to produce daughter vesicles with different interior and membrane compositions. J. Am. Chem. Soc. 133, 9545–9555 (2011).
McIntyre, J. C. & Sleight, R. G. Fluorescence assay for phospholipid membrane asymmetry. Biochemistry 30, 11819–11827 (1991).
Morris, S. J., Bradley, D. & Blumenthal, R. The use of cobalt ions as a collisional quencher to probe surface charge and stability of fluorescently labeled bilayer vesicles. Biochim. Biophys. Acta 818, 365–372 (1985).
Heider, E. C., Barhoum, M., Edwards, K., Gericke, K-H. & Harris, J. M. Structural characterization of individual vesicles using fluorescence microscopy. Anal. Chem. 83, 4909–4915 (2011).
Kantak, C., Beyer, S., Yobas, L., Bansal, T. & Trau, D. A. ‘microfluidic pinball’ for on-chip generation of layer-by-layer polyelectrolyte microcapsules. Lab Chip 11, 1030–1035 (2011).
Antonin, W., Ellenberg, J. & Dultz, E. Nuclear pore complex assembly through the cell cycle: regulation and membrane organization. FEBS Lett. 582, 2004–2016 (2008).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nature Chem. Biol. 9, 671–675 (2012).
Acknowledgements
This work was supported by a National Institutes of Health Pathway to Independence Career Development Award (GM083155) and a National Science Foundation CAREER Award (1255250) to B.M.P.
Author information
Authors and Affiliations
Contributions
B.M.P. and S.M. conceived and designed the experiments, analysed the resulting data and co-authored the paper. S.M. executed all experimental work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 756 kb)
Supplementary movie
Supplementary movie (MP4 23121 kb)
Rights and permissions
About this article
Cite this article
Matosevic, S., Paegel, B. Layer-by-layer cell membrane assembly. Nature Chem 5, 958–963 (2013). https://doi.org/10.1038/nchem.1765
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1765
This article is cited by
-
Multiple interactions between microfluidic droplets and on-chip pneumatic valves
Microfluidics and Nanofluidics (2022)
-
Cell-derived extracellular vesicles and membranes for tissue repair
Journal of Nanobiotechnology (2021)
-
Cell membrane coating integrity affects the internalization mechanism of biomimetic nanoparticles
Nature Communications (2021)
-
Dynamic self-assembly of compartmentalized DNA nanotubes
Nature Communications (2021)
-
New opportunities for creating man-made bioarchitectures utilizing microfluidics
Biomedical Microdevices (2019)