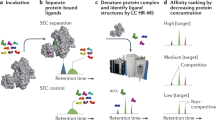
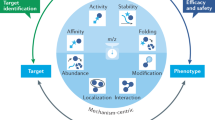
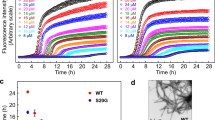
Abstract
The search for therapeutic agents that bind specifically to precursor protein conformations and inhibit amyloid assembly is an important challenge. Identifying such inhibitors is difficult because many protein precursors of aggregation are partially folded or intrinsically disordered, which rules out structure-based design. Furthermore, inhibitors can act by a variety of mechanisms, including specific or nonspecific binding, as well as colloidal inhibition. Here we report a high-throughput method based on ion mobility spectrometry–mass spectrometry (IMS–MS) that is capable of rapidly detecting small molecules that bind to amyloid precursors, identifying the interacting protein species and defining the mode of inhibition. Using this method we have classified a variety of small molecules that are potential inhibitors of human islet amyloid polypeptide (hIAPP) aggregation or amyloid-beta 1-40 aggregation as specific, nonspecific, colloidal or non-interacting. We also demonstrate the ability of IMS–MS to screen for inhibitory small molecules in a 96-well plate format and use this to discover a new inhibitor of hIAPP amyloid assembly.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sipe, J. D. et al. Amyloid fibril protein nomenclature: 2012 recommendations from the Nomenclature Committee of the International Society of Amyloidosis. Amyloid 19, 167–170 (2012).
Grimster, N. P. et al. Aromatic sulfonyl fluorides covalently kinetically stabilize transthyretin to prevent amyloidogenesis while affording a fluorescent conjugate. J. Am. Chem. Soc. 135, 5656–5668 (2013).
Klabunde, T. et al. Rational design of potent human transthyretin amyloid disease inhibitors. Nature Struct. Mol. Biol. 7, 312–321 (2000).
Connelly, S., Choi, S., Johnson, S. M., Kelly, J. W. & Wilson, I. A. Structure-based design of kinetic stabilizers that ameliorate the transthyretin amyloidoses. Curr. Opin. Struct. Biol. 20, 54–62 (2010).
Hamrang, Z., Rattray, N. J. W. & Pluen, A. Proteins behaving badly: emerging technologies in profiling biopharmaceutical aggregation. Trends Biotech. 31, 448–458 (2013).
Meier, J. J. et al. Inhibition of human IAPP fibril formation does not prevent β-cell death: evidence for distinct actions of oligomers and fibrils of human IAPP. Am. J. Physiol. Endocrinol. Metab. 291, E1317–E1324 (2006).
Aitken, J. F., Loomes, K. M., Konarkowska, B. & Cooper, G. J. S. Suppression by polycyclic compounds of the conversion of human amylin into insoluble amyloid. Biochem. J. 374, 779–784 (2003).
Harroun, T. A., Bradshaw, J. P. & Ashley, R. H. Inhibitors can arrest the membrane activity of human islet amyloid polypeptide independently of amyloid formation. FEBS Lett. 507, 200–204 (2001).
Woods, L. A. et al. Ligand binding to distinct states diverts aggregation of an amyloid-forming protein. Nature Chem. Biol. 7, 730–739 (2011).
Young, L. M., Cao, P., Raleigh, D. P., Ashcroft, A. E. & Radford, S. E. Ion mobility spectrometry–mass spectrometry defines the oligomeric intermediates in amylin amyloid formation and the mode of action of inhibitors. J. Am. Chem. Soc. 136, 660–670 (2014).
Hyung, S. J. et al. Insights into antiamyloidogenic properties of the green tea extract (–)-epigallocatechin-3-gallate toward metal-associated amyloid-β species. Proc. Natl Acad. Sci. USA 110, 3743–3748 (2013).
Westermark, P. Amyloid in the islets of Langerhans: thoughts and some historical aspects. Ups. J. Med. Sci. 116, 81–89 (2011).
Selkoe, D. J. Alzheimer's disease: genes, proteins, and therapy. Physiol. Rev. 81, 741–766 (2001).
Feng, B. Y. et al. Small-molecule aggregates inhibit amyloid polymerization. Nature Chem. Biol. 4, 197–199 (2008).
Bieschke, J. et al. EGCG remodels mature α-synuclein and amyloid-β fibrils and reduces cellular toxicity. Proc. Natl Acad. Sci. USA 107, 7710–7715 (2010).
Meng, F., Abedini, A., Plesner, A., Verchere, C. B. & Raleigh, D. P. The flavanol (–)-epigallocatechin 3-gallate inhibits amyloid formation by islet amyloid polypeptide, disaggregates amyloid fibrils, and protects cultured cells against IAPP-induced toxicity. Biochemistry 49, 8127–8133 (2010).
Howlett, D., Cutler, P., Heales, S. & Camilleri, P. Hemin and related porphyrins inhibit β-amyloid aggregation. FEBS Lett. 417, 249–251 (1997).
Meng, F. & Raleigh, D. P. Inhibition of glycosaminoglycan-mediated amyloid formation by islet amyloid polypeptide and proIAPP processing intermediates. J. Mol. Biol. 406, 491–502 (2010).
Porat, Y., Mazor, Y., Efrat, S. & Gazit, E. Inhibition of islet amyloid polypeptide fibril formation: a potential role for heteroaromatic interactions. Biochemistry 43, 14454–14462 (2004).
Ehrnhoefer, D. E. et al. EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nature Struct. Mol. Biol. 15, 558–566 (2008).
Daubenfeld, T., Bouin, A. P. & van der Rest, G. A deconvolution method for the separation of specific versus nonspecific interactions in noncovalent protein–ligand complexes analyzed by ESI-FT-ICR mass spectrometry. J. Am. Soc. Mass Spectrom. 17, 1239–1248 (2006).
Sun, N., Sun, J., Kitova, E. N. & Klassen, J. S. Identifying nonspecific ligand binding in electrospray ionization mass spectrometry using the reporter molecule method. J. Am. Soc. Mass Spectrom. 20, 1242–1250 (2009).
Sun, J., Kitova, E. N., Wang, W. & Klassen, J. S. Method for distinguishing specific from nonspecific protein–ligand complexes in nanoelectrospray ionization mass spectrometry. Anal. Chem. 78, 3010–3018 (2006).
Wang, W., Kitova, E. N. & Klassen, J. S. Influence of solution and gas phase processes on protein–carbohydrate binding affinities determined by nanoelectrospray Fourier transform ion cyclotron resonance mass spectrometry. Anal. Chem. 75, 4945–4955 (2003).
Meng, F. et al. The sulfated triphenyl methane derivative acid fuchsin is a potent inhibitor of amyloid formation by human islet amyloid polypeptide and protects against the toxic effects of amyloid formation. J. Mol. Biol. 400, 555–566 (2010).
Dupuis, N. F., Wu, C., Shea, J. E. & Bowers, M. T. Human islet amyloid polypeptide monomers form ordered beta-hairpins: a possible direct amyloidogenic precursor. J. Am. Chem. Soc. 131, 18283–18292 (2009).
Cheng, B. et al. Silibinin inhibits the toxic aggregation of human islet amyloid polypeptide. Biochem. Biophys. Res. Commun. 419, 495–499 (2012).
Khurana, R., Uversky, V. N., Nielsen, L. & Fink, A. L. Is Congo red an amyloid-specific dye? J. Biol. Chem. 276, 22715–22721 (2001).
Kim, Y. S., Randolph, T. W., Manning, M. C., Stevens, F. J. & Carpenter, J. F. Congo red populates partially unfolded states of an amyloidogenic protein to enhance aggregation and amyloid fibril formation. J. Biol. Chem. 278, 10842–10850 (2003).
Porat, Y., Abramowitz, A. & Gazit, E. Inhibition of amyloid fibril formation by polyphenols: structural similarity and aromatic interactions as a common inhibition mechanism. Chem. Biol. Drug. Des. 67, 27–37 (2006).
Thomas, T., Nadackal, G. T. & Thomas, K. Aspirin and diabetes: inhibition of amylin aggregation by nonsteroidal anti-inflammatory drugs. Exp. Clin. Endocrinol. Diabetes 111, 8–11 (2003).
Woods, L. A. et al. Ligand binding to distinct states diverts aggregation of an amyloid-forming protein. Nature Chem. Biol. 7, 730–739 (2011).
Cao, P. & Raleigh, D. P. Analysis of the inhibition and remodeling of islet amyloid polypeptide amyloid fibers by flavanols. Biochemistry 51, 2670–2683 (2012).
Palhano, F. L., Lee, J., Grimster, N. P. & Kelly, J. W. Toward the molecular mechanism(s) by which EGCG treatment remodels mature amyloid fibrils. J. Am. Chem. Soc. 135, 7503–7510 (2013).
Tenidis, K. et al. Identification of a penta- and hexapeptide of islet amyloid polypeptide (IAPP) with amyloidogenic and cytotoxic properties. J. Mol. Biol. 295, 1055–1071 (2000).
Petkova, A. T. et al. A structural model for Alzheimer's β-amyloid fibrils based on experimental constraints from solid state NMR. Proc. Natl Acad. Sci. USA 99, 16742–16747 (2002).
Young, L. et al. Monitoring oligomer formation from self-aggregating amylin peptides using ESI-IMS-MS. Int. J. Ion Mobil. Spectrom. 16, 29–39 (2013).
Aisen, P. et al. Alzhemed: a potential treatment for Alzheimer's disease. Curr. Alzheimer Res. 4, 473–478 (2007).
Gervais, F. et al. Targeting soluble Aβ peptide with tramiprosate for the treatment of brain amyloidosis. Neurobiol. Aging 28, 537–547 (2007).
Necula, M., Kayed, R., Milton, S. & Glabe, C. G. Small molecule inhibitors of aggregation indicate that amyloid β oligomerization and fibrillization pathways are independent and distinct. J. Biol. Chem. 282, 10311–10324 (2007).
Valler, M. J. & Green, D. Diversity screening versus focussed screening in drug discovery. Drug Discov. Today 5, 286–293 (2000).
De Felice, F. G. et al. Targeting the neurotoxic species in Alzheimer's disease: inhibitors of Aβ oligomerization. FASEB J. 18, 1366–1372 (2004).
Ladiwala, A. R. A. et al. Resveratrol selectively remodels soluble oligomers and fibrils of amyloid Aβ into off-pathway conformers. J. Biol. Chem. 285, 24228–24237 (2010).
Yang, F. et al. Curcumin inhibits formation of amyloid β oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J. Biol. Chem. 280, 5892–5901 (2005).
Scherzer-Attali, R. et al. Complete phenotypic recovery of an Alzheimer's disease model by a quinone–tryptophan hybrid aggregation inhibitor. PLoS ONE 5, e11101 (2010).
Rapid Overlay of Chemical Structures (OpenEye, Scientific Software, Santa Fe, New Mexico, 2007).
Marek, P., Woys, A. M., Sutton, K., Zanni, M. T. & Raleigh, D. P. Efficient microwave-assisted synthesis of human islet amyloid polypeptide designed to facilitate the specific incorporation of labeled amino acids. Org. Lett. 12, 4848–4851 (2010).
Giles, K. et al. Applications of a travelling wave-based radio-frequency-only stacked ring ion guide. Rapid Commun. Mass Spectrom. 18, 2401–2414 (2004).
Platt, G. W., Routledge, K. E., Homans, S. W. & Radford, S. E. Fibril growth kinetics reveal a region of beta2-microglobulin important for nucleation and elongation of aggregation. J. Mol. Biol. 378, 251–263 (2008).
Maestro, version 9.3, Schrödinger Release 2014–2 (Schrödinger LLC, New York, 2014).
Acknowledgements
L.M.Y. is funded by a Biotechnology and Biological Sciences Research Council (BBSRC) CASE studentship (Grant Number BB/I015361/1) sponsored by Micromass UK Ltd/Waters Corporation. J.C.S. is funded by a BBSRC CASE studentship (Grant Number BB/H014713/1) sponsored by Avacta Analytical. R.A.M. is funded by a BBSRC studentship (Grant Number BB/F01614X/1). The Synapt HDMS mass spectrometer was purchased with funds from the BBSRC through its Research Equipment Initiative scheme (BB/E012558/1). S.E.R. acknowledges funding from the European Research Council under the European Union's Seventh Framework Programme (FP7/2007-2013; 322408). D.P.R. acknowledges support from the US National Institutes of Health (GM078114). We thank D. Walsh (Brigham & Women's Hospital) and S. Linse (Lund University) for provision of the Aβ40 peptide and vector and J. R. Ault (University of Leeds) for setting up the automated ESI-MS analyses. We also acknowledge all members of the Ashcroft, Radford and Raleigh groups for helpful discussions.
Author information
Authors and Affiliations
Contributions
L.M.Y. and J.C.S. contributed equally to this work. L.M.Y., J.C.S., S.E.R. and A.E.A. conceived and designed the experiments. L.M.Y., J.C.S. and R.A.M. performed the experiments. C.H.R. and R.J.F. designed and prepared the screening library. L.H.T. and D.P.R. prepared the hIAPP. L.M.Y. analysed the data. All authors discussed the results, and wrote and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 8855 kb)
Rights and permissions
About this article
Cite this article
Young, L., Saunders, J., Mahood, R. et al. Screening and classifying small-molecule inhibitors of amyloid formation using ion mobility spectrometry–mass spectrometry. Nature Chem 7, 73–81 (2015). https://doi.org/10.1038/nchem.2129
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2129
This article is cited by
-
Five-mer peptides prevent short-term spatial memory deficits in Aβ25-35-induced Alzheimer’s model mouse by suppressing Aβ25-35 aggregation and resolving its aggregate form
Alzheimer's Research & Therapy (2023)
-
Advances in mass spectrometry to unravel the structure and function of protein condensates
Nature Protocols (2023)
-
Tuning the rate of aggregation of hIAPP into amyloid using small-molecule modulators of assembly
Nature Communications (2022)
-
Islet amyloid polypeptide aggregation exerts cytotoxic and proinflammatory effects on the islet vasculature in mice
Diabetologia (2022)
-
Plasmonic nanoparticle amyloid corona for screening Aβ oligomeric aggregate-degrading drugs
Nature Communications (2021)