Abstract
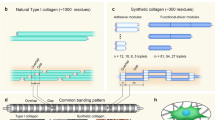
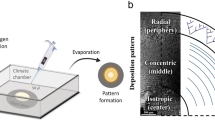
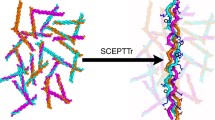
Sticky-ended DNA duplexes can associate spontaneously into long double helices; however, such self-assembly is much less developed with proteins. Collagen is the most prevalent component of the extracellular matrix and a common clinical biomaterial. As for natural DNA, the ~103-residue triple helices (~300 nm) of natural collagen are recalcitrant to chemical synthesis. Here we show how the self-assembly of short collagen-mimetic peptides (CMPs) can enable the fabrication of synthetic collagen triple helices that are nearly a micrometre in length. Inspired by the mathematics of tessellations, we derive rules for the design of single CMPs that self-assemble into long triple helices with perfect symmetry. Sticky ends thus created are uniform across the assembly and drive its growth. Enacting this design yields individual triple helices that, in length, match or exceed those in natural collagen and are remarkably thermostable, despite the absence of higher-order association. The symmetric assembly of CMPs provides an enabling platform for the development of advanced materials for medicine and nanotechnology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cohen, S. N., Chang, A. C. Y., Boyer, H. W. & Helling, R. B. Construction of biologically functional bacterial plasmids in-vitro. Proc. Natl Acad. Sci. USA 70, 3240–3244 (1973).
Gibson, D. G. et al. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nature Methods 6, 343–345 (2009).
Khalil, A. S. & Collins, J. J. Synthetic biology: applications come of age. Nature Rev. Genet. 11, 367–379 (2010).
Seeman, N. C. Nanomaterials based on DNA. Annu. Rev. Biochem. 79, 65–87 (2010).
Zimenkov, Y. et al. Rational design of a reversible pH-responsive switch for peptide self-assembly. J. Am. Chem. Soc. 128, 6770–6771 (2006).
Banwell, E. F. et al. Rational design and application of responsive α-helical peptide hydrogels. Nature Mater. 8, 596–600 (2009).
Brinckmann, J. Collagens at a glance. Top. Curr. Chem. 247, 1–6 (2005).
Meyers, M. A., Chen, P.-Y., Lin, A. Y.-M. & Seki, Y. Biological materials: structure and mechanical properties. Prog. Mater. Sci. 53, 1–206 (2008).
Chattopadhyay, S. & Raines, R. T. Collagen-based biomaterials for wound healing. Biopolymers 101, 821–833 (2014).
Ricard-Blum, S. The collagen family. Cold Spring Harb. Perspect. Biol. 3, a004978 (2011).
Fields, G. B. Synthesis and biological applications of collagen-model triple-helical peptides. Org. Biomol. Chem. 8, 1237–1258 (2010).
Shoulders, M. D. & Raines, R. T. Collagen structure and stability. Annu. Rev. Biochem. 78, 929–958 (2009).
Siebler, C., Erdmann, R. S. & Wennemers, H. From azidoproline to functionalizable collagen. Chimia 67, 891–895 (2013).
Kotch, F. W. & Raines, R. T. Self-assembly of synthetic collagen triple helices. Proc. Natl Acad. Sci. USA 103, 3028–3033 (2006).
Rele, S. et al. D-Periodic collagen-mimetic microfibers. J. Am. Chem. Soc. 129, 14780–14787 (2007).
Gottlieb, D., Morin, S. A., Jin, S. & Raines, R. T. Self-assembled collagen-like peptide fibers as templates for metallic nanowires. J. Mater. Chem. 18, 3865–3870 (2008).
Cejas, M. A. et al. Thrombogenic collagen-mimetic peptides: self-assembly of triple helix-based fibrils driven by hydrophobic interactions. Proc. Natl Acad. Sci. USA 105, 8513–8518 (2008).
Yamazaki, C. M. et al. A collagen-mimetic triple helical supramolecule that evokes integrin-dependent cell responses. Biomaterials 31, 1925–1934 (2010).
O'Leary, L. E. R., Fallas, J. A., Bakota, E. L., Kang, M. K. & Hartgerink, J. D. Multi-hierarchical self-assembly of a collagen mimetic peptide from triple helix to nanofibre and hydrogel. Nature Chem. 3, 821–828 (2011).
Xu, F. et al. Compositional control of higher order assembly using synthetic collagen peptides. J. Am. Chem. Soc. 134, 47–50 (2012).
Sarkar, B., O'Leary, L. E. R. & Hartgerink, J. D. Self-assembly of fiber-forming collagen mimetic peptides controlled by triple-helical nucleation. J. Am. Chem. Soc. 136, 14417–14424 (2014).
Persikov, A. V., Ramshaw, J. A. M. & Brodsky, B. Prediction of collagen stability from amino acid sequence. J. Biol. Chem. 280, 19343–19349 (2005).
Fallas, J. A., Gauba, V. & Hartgerink, J. D. Solution structure of an ABC collagen heterotrimer reveals a single-register helix stabilized by electrostatic interactions. J. Biol. Chem. 284, 26851–26859 (2009).
Coxeter, H. S. M. Crystal symmetry and its generalizations. Trans. R. Soc. Can. 51, 1–13 (1957).
Emmer, M. & Schattschneider, D. M. C. Escher's Legacy: A Centennial Celebration (Springer, 2003).
Crick, F. H. C. & Watson, J. D. Structure of small viruses. Nature 177, 473–475 (1956).
Padilla, J. E., Colovos, C. & Yeates, T. O. Nanohedra: using symmetry to design self assembling protein cages, layers, crystals, and filaments. Proc. Natl Acad. Sci. USA 98, 2217–2221 (2001).
Tanrikulu, I. C. & Raines, R. T. Optimal interstrand bridges for collagen-like biomaterials. J. Am. Chem. Soc. 136, 13490–13493 (2014).
Gauba, V. & Hartgerink, J. D. Surprisingly high stability of collagen ABC heterotrimer: evaluation of side chain charge pairs. J. Am. Chem. Soc. 129, 15034–15041 (2007).
Smulders, M. M. J. et al. How to distinguish isodesmic from cooperative supramolecular polymerisation. Chem. Eur. J. 16, 362–367 (2010).
Gelman, R. A., Williams, B. R. & Piez, K. A. Collagen fibril formation: evidence for a multistep process. J. Biol. Chem. 254, 180–186 (1979).
Bai, H. Y., Xu, K., Xu, Y. J. & Matsui, H. Fabrication of Au nanowires of uniform length and diameter using a monodisperse and rigid biomolecular template: collagen-like triple helix. Angew. Chem. Int. Ed. 46, 3319–3322 (2007).
Persikov, A. V., Ramshaw, J. A., Kirkpatrick, A. & Brodsky, B. Amino acid propensities for the collagen triple-helix. Biochemistry 39, 14960–14967 (2000).
Chen, Y.-S., Chen, C.-C. & Horng, J.-C. Thermodynamic and kinetic consequences of substituting glycine at different positions in a Pro-Hyp-Gly repeat collagen model peptide. Biopolymers 96, 60–68 (2011).
Jiang, T., Xu, C., Zuo, X. & Conticello, V. P. Structurally homogeneous nanosheets from self-assembly of a collagen-mimetic peptide. Angew. Chem. Int. Ed. 53, 8367–8371 (2014).
McGuinness, K., Khan, I. J. & Nanda, V. Morphological diversity and polymorphism of self-assembling collagen peptides controlled by length of hydrophobic domains. ACS Nano 8, 12514–12523 (2014).
Wess, T. J. Collagen fibril form and function. Adv. Protein Chem. 70, 341–374 (2005).
Rad-Malekshahi, M., Lempsink, L., Amidi, M., Hennink, W. E. & Mastrobattista, E. Biomedical applications of self-assembling peptides. Bioconjugate Chem. 27, 3–18 (2016).
Kaur, P. et al. Three-dimensional directed self-assembly of peptide nanowires into micrometer-sized crystalline cubes with nanoparticle joints. Angew. Chem. Int. Ed. 49, 8375–8378 (2010).
Durchschlag, H. & Zipper, P. Calculation of the partial volume of organic compounds and polymers. Ultracentrifugation 94, 20–39 (1994).
Acknowledgements
We thank A. J. Ellison, B. M. Hoover, R. Biswas and S. Chattopadhyay for help with solution- and solid-phase peptide synthesis, S. A. Morin for help with nanocharacterization, M. D. Shoulders, F. W. Kotch and E. Emrah for discussions on the CMP assembly and R. W. Newberry for reviewing the manuscript. This study would not have been possible without expert advice and assistance from M. D. Boersma and N. Porcaro (UW Biotechnology Center) and from D. R. McCaslin (UW BIF). A.F. and S.J. were supported by Grant DMR-0832760 (National Science Foundation). This work was supported by Grant R01 AR044276 (National Institutes of Health).
Author information
Authors and Affiliations
Contributions
I.C.T and R.T.R. conceived the project and planned the experiments. I.C.T. designed, synthesized and characterized the peptides in solution, and computed their association landscapes. A.F. imaged the peptide assemblies. All the authors analysed the data. I.C.T. and R.T.R. wrote the paper. All the authors proofread, commented on and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 22777 kb)
Rights and permissions
About this article
Cite this article
Tanrikulu, I., Forticaux, A., Jin, S. et al. Peptide tessellation yields micrometre-scale collagen triple helices. Nature Chem 8, 1008–1014 (2016). https://doi.org/10.1038/nchem.2556
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2556
This article is cited by
-
Design of synthetic collagens that assemble into supramolecular banded fibers as a functional biomaterial testbed
Nature Communications (2022)
-
Supramolecular tessellations by the exo-wall interactions of pagoda[4]arene
Nature Communications (2021)