Abstract
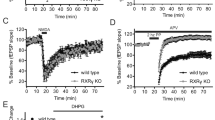
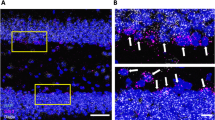
Liver X receptors (Lxrα and Lxrβ) are ligand-dependent nuclear receptors critical for ventral midbrain neurogenesis in vivo. However, no endogenous midbrain Lxr ligand has so far been identified. Here we used LC/MS and functional assays to identify cholic acid as a new Lxr ligand. Moreover, 24(S),25-epoxycholesterol (24,25-EC) was found to be the most potent and abundant Lxr ligand in the developing mouse midbrain. Both Lxr ligands promoted neural development in an Lxr-dependent manner in zebrafish in vivo. Notably, each ligand selectively regulated the development of distinct midbrain neuronal populations. Whereas cholic acid increased survival and neurogenesis of Brn3a-positive red nucleus neurons, 24,25-EC promoted dopaminergic neurogenesis. These results identify an entirely new class of highly selective and cell type–specific regulators of neurogenesis and neuronal survival. Moreover, 24,25-EC promoted dopaminergic differentiation of embryonic stem cells, suggesting that Lxr ligands may thus contribute to the development of cell replacement and regenerative therapies for Parkinson's disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chawla, A., Repa, J.J., Evans, R.M. & Mangelsdorf, D.J. Nuclear receptors and lipid physiology: opening the X-files. Science 294, 1866–1870 (2001).
Bensinger, S.J. & Tontonoz, P. Integration of metabolism and inflammation by lipid-activated nuclear receptors. Nature 454, 470–477 (2008).
Alberti, S., Steffensen, K.R. & Gustafsson, J.A. Structural characterisation of the mouse nuclear oxysterol receptor genes LXRα and LXRβ. Gene 243, 93–103 (2000).
Edwards, P.A., Kennedy, M.A. & Mak, P.A. LXRs: oxysterol-activated nuclear receptors that regulate genes controlling lipid homeostasis. Vascul. Pharmacol. 38, 249–256 (2002).
Zelcer, N. & Tontonoz, P. Liver X receptors as integrators of metabolic and inflammatory signaling. J. Clin. Invest. 116, 607–614 (2006).
Hu, X., Li, S., Wu, J., Xia, C. & Lala, D.S. Liver X receptors interact with corepressors to regulate gene expression. Mol. Endocrinol. 17, 1019–1026 (2003).
Janowski, B.A., Willy, P.J., Devi, T.R., Falck, J.R. & Mangelsdorf, D.J. An oxysterol signalling pathway mediated by the nuclear receptor LXRα. Nature 383, 728–731 (1996).
Janowski, B.A. et al. Structural requirements of ligands for the oxysterol liver X receptors LXRα and LXRβ. Proc. Natl. Acad. Sci. USA 96, 266–271 (1999).
Viennois, E. et al. Targeting liver X receptors in human health: deadlock or promising trail? Expert Opin. Ther. Targets 15, 219–232 (2011).
Björkhem, I. Are side-chain oxidized oxysterols regulators also in vivo? J. Lipid Res. 50, S213–S218 (2009).
Lehmann, J.M. et al. Activation of the nuclear receptor LXR by oxysterols defines a new hormone response pathway. J. Biol. Chem. 272, 3137–3140 (1997).
Fu, X. et al. 27-hydroxycholesterol is an endogenous ligand for liver X receptor in cholesterol-loaded cells. J. Biol. Chem. 276, 38378–38387 (2001).
Zhao, C. & Dahlman-Wright, K. Liver X receptor in cholesterol metabolism. J. Endocrinol. 204, 233–240 (2010).
Björkhem, I. & Diczfalusy, U. Oxysterols: friends, foes, or just fellow passengers? Arterioscler. Thromb. Vasc. Biol. 22, 734–742 (2002).
Schroepfer, G.J. Oxysterols: modulators of cholesterol metabolism and other processes. Physiol. Rev. 80, 361–554 (2000).
Laffitte, B.A. et al. LXRs control lipid-inducible expression of the apolipoprotein E gene in macrophages and adipocytes. Proc. Natl. Acad. Sci. USA 98, 507–512 (2001).
Venkateswaran, A. et al. Control of cellular cholesterol efflux by the nuclear oxysterol receptor LXRα. Proc. Natl. Acad. Sci. USA 97, 12097–12102 (2000).
Joseph, S.B., Castrillo, A., Laffitte, B.A., Mangelsdorf, D.J. & Tontonoz, P. Reciprocal regulation of inflammation and lipid metabolism by liver X receptors. Nat. Med. 9, 213–219 (2003).
Hannedouche, S. et al. Oxysterols direct immune cell migration via EBI2. Nature 475, 524–527 (2011).
Valledor, A.F. et al. Activation of liver X receptors and retinoid X receptors prevents bacterial-induced macrophage apoptosis. Proc. Natl. Acad. Sci. USA 101, 17813–17818 (2004).
Wang, L. et al. Liver X receptors in the central nervous system: from lipid homeostasis to neuronal degeneration. Proc. Natl. Acad. Sci. USA 99, 13878–13883 (2002).
Sacchetti, P. et al. Liver X receptors and oxysterols promote ventral midbrain neurogenesis in vivo and in human embryonic stem cells. Cell Stem Cell 5, 409–419 (2009).
Andersson, S., Gustafsson, N., Warner, M. & Gustafsson, J.A. Inactivation of liver X receptor β leads to adult-onset motor neuron degeneration in male mice. Proc. Natl. Acad. Sci. USA 102, 3857–3862 (2005).
Zelcer, N. et al. Attenuation of neuroinflammation and Alzheimer's disease pathology by liver x receptors. Proc. Natl. Acad. Sci. USA 104, 10601–10606 (2007).
Fitz, N.F. et al. Liver X receptor agonist treatment ameliorates amyloid pathology and memory deficits caused by high-fat diet in APP23 mice. J. Neurosci. 30, 6862–6872 (2010).
Kele, J. et al. Neurogenin 2 is required for the development of ventral midbrain dopaminergic neurons. Development 133, 495–505 (2006).
Wang, Y. et al. Targeted lipidomic analysis of oxysterols in the embryonic central nervous system. Mol. Biosyst. 5, 529–541 (2009).
Mano, N. et al. Presence of protein-bound unconjugated bile acids in the cytoplasmic fraction of rat brain. J. Lipid Res. 45, 295–300 (2004).
Gan, X. et al. Dual mechanisms of ABCA1 regulation by geranylgeranyl pyrophosphate. J. Biol. Chem. 276, 48702–48708 (2001).
Russek-Blum, N. et al. Dopaminergic neuronal cluster size is determined during early forebrain patterning. Development 135, 3401–3413 (2008).
Chen, Y.C., Priyadarshini, M. & Panula, P. Complementary developmental expression of the two tyrosine hydroxylase transcripts in zebrafish. Histochem. Cell Biol. 132, 375–381 (2009).
Zetterström, R.H. et al. Dopamine neuron agenesis in Nurr1-deficient mice. Science 276, 248–250 (1997).
Bello, E.P. et al. Cocaine supersensitivity and enhanced motivation for reward in mice lacking dopamine D2 autoreceptors. Nat. Neurosci. 14, 1033–1038 (2011).
Makishima, M. et al. Identification of a nuclear receptor for bile acids. Science 284, 1362–1365 (1999).
Parks, D.J. et al. Bile acids: natural ligands for an orphan nuclear receptor. Science 284, 1365–1368 (1999).
Wang, H., Chen, J., Hollister, K., Sowers, L.C. & Forman, B.M. Endogenous bile acids are ligands for the nuclear receptor FXR/BAR. Mol. Cell 3, 543–553 (1999).
Song, C., Hiipakka, R.A. & Liao, S. Selective activation of liver X receptor α by 6α-hydroxy bile acids and analogs. Steroids 65, 423–427 (2000).
Song, C. & Liao, S. Cholestenoic acid is a naturally occurring ligand for liver X receptor α. Endocrinology 141, 4180–4184 (2000).
Goodwin, B.J., Zuercher, W.J. & Collins, J.L. Recent advances in liver X receptor biology and chemistry. Curr. Top. Med. Chem. 8, 781–791 (2008).
Gray, P.A. et al. Mouse brain organization revealed through direct genome-scale TF expression analysis. Science 306, 2255–2257 (2004).
Makishima, M. et al. Vitamin D receptor as an intestinal bile acid sensor. Science 296, 1313–1316 (2002).
Tint, G.S., Yu, H., Shang, Q., Xu, G. & Patel, S.B. The use of the Dhcr7 knockout mouse to accurately determine the origin of fetal sterols. J. Lipid Res. 47, 1535–1541 (2006).
Zhang, C.L., Zou, Y., He, W., Gage, F.H. & Evans, R.M. A role for adult TLX-positive neural stem cells in learning and behaviour. Nature 451, 1004–1007 (2008).
Lee, S., Lee, B., Lee, J.W. & Lee, S.K. Retinoid signaling and neurogenin2 function are coupled for the specification of spinal motor neurons through a chromatin modifier CBP. Neuron 62, 641–654 (2009).
Környei, Z. et al. Astroglia-derived retinoic acid is a key factor in glia-induced neurogenesis. FASEB J. 21, 2496–2509 (2007).
Berrodin, T.J. et al. Identification of 5,6-epoxycholesterol as a novel modulator of liver x receptor activity. Mol. Pharmacol. 78, 1046–1058 (2010).
Venteclef, N. et al. GPS2-dependent corepressor/SUMO pathways govern anti-inflammatory actions of LRH-1 and LXRβ in the hepatic acute phase response. Genes Dev. 24, 381–395 (2010).
Huang, P., Chandra, V. & Rastinejad, F. Structural overview of the nuclear receptor superfamily: insights into physiology and therapeutics. Annu. Rev. Physiol. 72, 247–272 (2010).
Clare, K., Hardwick, S.J., Carpenter, K.L., Weeratunge, N. & Mitchinson, M.J. Toxicity of oxysterols to human monocyte-macrophages. Atherosclerosis 118, 67–75 (1995).
Jakobsson, T. et al. GPS2 is required for cholesterol efflux by triggering histone demethylation, LXR recruitment, and coregulator assembly at the ABCG1 locus. Mol. Cell 34, 510–518 (2009).
Boergesen, M. et al. Genome-wide profiling of liver X receptor, retinoid X receptor, and peroxisome proliferator-activated receptor in mouse liver reveals extensive sharing of binding sites. Mol. Cell. Biol. 32, 852–867 (2012).
Archer, A. et al. The Liver X-Receptor (Lxr) governs lipid homeostasis in zebrafish during development. Open J. Endocrine Metab. Diseases. 2, 74–81 (2012).
Acknowledgements
Work in Karolinska was supported by Hjärnfonden, European Union (Strokemap and NeuroStemCell projects), Swedish Foundation for Strategic Research (SRL Program), Karolinska Institutet (Strategic Research Program and Thematic Center in Stem Cells and Regenerative Medicine) and the Swedish Research Council (DBRM, 2008:2811, 2011-3116 and 3318). The authors are grateful to L. Jansson-Sjöstrand and J. Söderlund for technical assistance, A. Nanni for secretarial help and S. Yang for help with artwork. Work in Swansea was supported by funding from the UK Research Councils Biotechnology and Biological Sciences Research Council (BB/C515771/2, BB/C511356/1 and BB/I001735/1 to W.J.G. and BB/H001018/1 to Y.W.). S.T. was supported by the Swedish Medical Research Council and the Onassis Foundation.
Author information
Authors and Affiliations
Contributions
S.T. performed most of the experiments. Y.W. performed the HPLC and LC/MSn experiments. S.S.K. performed some of the zebrafish experiments. P.S. contributed to some luciferase assay and primary culture experiments. K.M.S. contributed to some luciferase assay experiments. K.B., J.K., M.G. and K.K. contributed to the HPLC and LC/MSn experiments. C.S. contributed to some of the mESC experiments. E.M.T. contributed to some of the ChIP experiments. J.-Å.G. and K.R.S. provided the Lxrα−/− Lxrβ−/− mice. P.E., J.S. and W.J.G. contributed to draw conclusions from experiments, supervision and provided intellectual input. E.A. contributed to experimental design, supervision, conclusions and coordinated experimental approaches and wrote the manuscript together with S.T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results (PDF 1666 kb)
Rights and permissions
About this article
Cite this article
Theofilopoulos, S., Wang, Y., Kitambi, S. et al. Brain endogenous liver X receptor ligands selectively promote midbrain neurogenesis. Nat Chem Biol 9, 126–133 (2013). https://doi.org/10.1038/nchembio.1156
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1156
This article is cited by
-
Role of Brain Liver X Receptor in Parkinson’s Disease: Hidden Treasure and Emerging Opportunities
Molecular Neurobiology (2024)
-
Microbial-derived metabolites as a risk factor of age-related cognitive decline and dementia
Molecular Neurodegeneration (2022)
-
Key role for lipids in cognitive symptoms of schizophrenia
Translational Psychiatry (2020)
-
Activation of LXRβ Signaling in the Amygdala Confers Anxiolytic Effects Through Rebalancing Excitatory and Inhibitory Neurotransmission upon Acute Stress
Neurotherapeutics (2020)
-
Optimization of mass spectrometry settings for steroidomic analysis in young and old killifish
Analytical and Bioanalytical Chemistry (2020)