Abstract
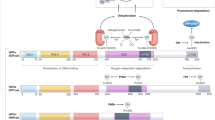
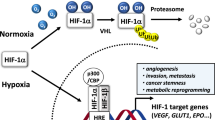
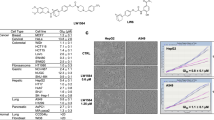
Hypoxia inducible factors (HIFs) are heterodimeric transcription factors induced in many cancers where they frequently promote the expression of protumorigenic pathways. Though transcription factors are typically considered 'undruggable', the PAS-B domain of the HIF-2α subunit contains a large cavity within its hydrophobic core that offers a unique foothold for small-molecule regulation. Here we identify artificial ligands that bind within this pocket and characterize the resulting structural and functional changes caused by binding. Notably, these ligands antagonize HIF-2 heterodimerization and DNA-binding activity in vitro and in cultured cells, reducing HIF-2 target gene expression. Despite the high sequence identity between HIF-2α and HIF-1α, these ligands are highly selective and do not affect HIF-1 function. These chemical tools establish the molecular basis for selective regulation of HIF-2, providing potential therapeutic opportunities to intervene in HIF-2–driven tumors, such as renal cell carcinomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Semenza, G.L. Hypoxia-inducible factors: mediators of cancer progression and targets for cancer therapy. Trends Pharmacol. Sci. 33, 207–214 (2012).
Xia, X. et al. Integrative analysis of HIF binding and transactivation reveals its role in maintaining histone methylation homeostasis. Proc. Natl. Acad. Sci. USA 106, 4260–4265 (2009).
Schödel, J. et al. High-resolution genome-wide mapping of HIF-binding sites by ChIP-seq. Blood 117, e207–e217 (2011).
Majmundar, A.J., Wong, W.J. & Simon, M.C. Hypoxia-inducible factors and the response to hypoxic stress. Mol. Cell 40, 294–309 (2010).
Greer, S.N., Metcalf, J.L., Wang, Y. & Ohh, M. The updated biology of hypoxia-inducible factor. EMBO J. 31, 2448–2460 (2012).
Kondo, K., Kim, W.Y., Lechpammer, M. & Kaelin, W.G. Jr. Inhibition of HIF2α is sufficient to suppress pVHL-defective tumor growth. PLoS Biol. 1, E83 (2003).
Kondo, K., Klco, J., Nakamura, E., Lechpammer, M. & Kaelin, W.G. Jr. Inhibition of HIF is necessary for tumor suppression by the von Hippel-Lindau protein. Cancer Cell 1, 237–246 (2002).
Maranchie, J.K. et al. The contribution of VHL substrate binding and HIF1-α to the phenotype of VHL loss in renal cell carcinoma. Cancer Cell 1, 247–255 (2002).
Erbel, P.J., Card, P.B., Karakuzu, O., Bruick, R.K. & Gardner, K.H. Structural basis for PAS domain heterodimerization in the basic helix-loop-helix–PAS transcription factor hypoxia-inducible factor. Proc. Natl. Acad. Sci. USA 100, 15504–15509 (2003).
Yang, J. et al. Functions of the Per/ARNT/Sim (PAS) domains of the hypoxia inducible factor (HIF). J. Biol. Chem. 280, 36047–36054 (2005).
Partch, C.L., Card, P.B., Amezcua, C.A. & Gardner, K.H. Molecular basis of coiled coil coactivator recruitment by the aryl hydrocarbon receptor nuclear translocator (ARNT). J. Biol. Chem. 284, 15184–15192 (2009).
Partch, C.L. & Gardner, K.H. Coactivators necessary for transcriptional output of the hypoxia inducible factor, HIF, are directly recruited by ARNT PAS-B. Proc. Natl. Acad. Sci. USA 108, 7739–7744 (2011).
Henry, J.T. & Crosson, S. Ligand-binding PAS domains in a genomic, cellular, and structural context. Annu. Rev. Microbiol. 65, 261–286 (2011).
Harper, S.M., Neil, L.C. & Gardner, K.H. Structural basis of a phototropin light switch. Science 301, 1541–1544 (2003).
Wells, J.A. & McClendon, C.L. Reaching for high-hanging fruit in drug discovery at protein-protein interfaces. Nature 450, 1001–1009 (2007).
Koehler, A.N. A complex task? Direct modulation of transcription factors with small molecules. Curr. Opin. Chem. Biol. 14, 331–340 (2010).
Key, J., Scheuermann, T.H., Anderson, P.C., Daggett, V. & Gardner, K.H. Principles of ligand binding within a completely buried cavity in HIF2α PAS-B. J. Am. Chem. Soc. 131, 17647–17654 (2009).
Scheuermann, T.H. et al. Artificial ligand binding within the HIF2α PAS-B domain of the HIF2 transcription factor. Proc. Natl. Acad. Sci. USA 106, 450–455 (2009).
Qing, G. & Simon, M.C. Hypoxia inducible factor-2α: a critical mediator of aggressive tumor phenotypes. Curr. Opin. Genet. Dev. 19, 60–66 (2009).
Morris, M.R. et al. Mutation analysis of hypoxia-inducible factors HIF1α and HIF2α in renal cell carcinoma. Anticancer Res. 29, 4337–4343 (2009).
Kaelin, W.G. Jr. Treatment of kidney cancer: insights provided by the VHL tumor-suppressor protein. Cancer 115, 2262–2272 (2009).
Shen, C. et al. Genetic and functional studies implicate HIF1α as a 14q kidney cancer suppressor gene. Cancer Discov. 1, 222–235 (2011).
Amezcua, C.A., Harper, S.M., Rutter, J. & Gardner, K.H. Structure and interactions of PAS kinase N-terminal PAS domain: model for intramolecular kinase regulation. Structure 10, 1349–1361 (2002).
Rogers, J.L. et al. Development of inhibitors of the PAS-B domain of the HIF-2α transcription factor. J. Med. Chem. 10.1021/jm301847z (30 January 2013).
Krieg, M. et al. Up-regulation of hypoxia-inducible factors HIF-1α and HIF-2α under normoxic conditions in renal carcinoma cells by von Hippel-Lindau tumor suppressor gene loss of function. Oncogene 19, 5435–5443 (2000).
Dioum, E.M. et al. Regulation of hypoxia-inducible factor 2α signaling by the stress-responsive deacetylase sirtuin 1. Science 324, 1289–1293 (2009).
Scortegagna, M. et al. HIF-2α regulates murine hematopoietic development in an erythropoietin-dependent manner. Blood 105, 3133–3140 (2005).
Semenza, G.L. Targeting HIF-1 for cancer therapy. Nat. Rev. Cancer 3, 721–732 (2003).
Keith, B., Johnson, R.S. & Simon, M.C. HIF1α and HIF2α: sibling rivalry in hypoxic tumour growth and progression. Nat. Rev. Cancer 12, 9–22 (2012).
Franovic, A., Holterman, C.E., Payette, J. & Lee, S. Human cancers converge at the HIF-2α oncogenic axis. Proc. Natl. Acad. Sci. USA 106, 21306–21311 (2009).
Holmquist-Mengelbier, L. et al. Recruitment of HIF-1α and HIF-2α to common target genes is differentially regulated in neuroblastoma: HIF-2α promotes an aggressive phenotype. Cancer Cell 10, 413–423 (2006).
Pietras, A. et al. HIF-2α maintains an undifferentiated state in neural crest-like human neuroblastoma tumor-initiating cells. Proc. Natl. Acad. Sci. USA 106, 16805–16810 (2009).
Mazumdar, J. et al. HIF-2α deletion promotes Kras-driven lung tumor development. Proc. Natl. Acad. Sci. USA 107, 14182–14187 (2010).
Ivan, M. et al. HIFα targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. Science 292, 464–468 (2001).
Jaakkola, P. et al. Targeting of HIF-α to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 292, 468–472 (2001).
Lando, D. et al. FIH-1 is an asparaginyl hydroxylase enzyme that regulates the transcriptional activity of hypoxia-inducible factor. Genes Dev. 16, 1466–1471 (2002).
Lando, D., Peet, D.J., Whelan, D.A., Gorman, J.J. & Whitelaw, M.L. Asparagine hydroxylation of the HIF transactivation domain: a hypoxic switch. Science 295, 858–861 (2002).
Kaelin, W.G. Von Hippel-Lindau disease. Annu. Rev. Pathol. 2, 145–173 (2007).
Halavaty, A.S. & Moffat, K. N- and C-terminal flanking regions modulate light-induced signal transduction in the LOV2 domain of the blue light sensor phototropin 1 from Avena sativa. Biochemistry 46, 14001–14009 (2007).
Park, E.J. et al. Targeting the PAS-A domain of HIF-1α for development of small molecule inhibitors of HIF-1. Cell Cycle 5, 1847–1853 (2006).
Lee, K. et al. Acriflavine inhibits HIF-1 dimerization, tumor growth, and vascularization. Proc. Natl. Acad. Sci. USA 106, 17910–17915 (2009).
Semenza, G.L. HIF-1: upstream and downstream of cancer metabolism. Curr. Opin. Genet. Dev. 20, 51–56 (2010).
Johnson, B.A. & Blevins, R.A. NMRView: a computer program for the visualization and analysis of NMR data. J. Biomol. NMR 4, 603–614 (1994).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Schüttelkopf, A.W. & van Aalten, D.M. PRODRG: a tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr. D Biol. Crystallogr. 60, 1355–1363 (2004).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Winn, M.D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D Biol. Crystallogr. 67, 235–242 (2011).
Martí-Renom, M.A. et al. Comparative protein structure modeling of genes and genomes. Annu. Rev. Biophys. Biomol. Struct. 29, 291–325 (2000).
Bookout, A.L. & Mangelsdorf, D.J. Quantitative real-time PCR protocol for analysis of nuclear receptor signaling pathways. Nucl. Recept. Signal. 1, e012 (2003).
Scheuermann, T.H., Yang, J., Zhang, L., Gardner, K.H. & Bruick, R.K. Hypoxia-inducible factors Per/ARNT/Sim domains: structure and function. Methods Enzymol. 435, 3–24 (2007).
Chen, R., Dioum, E.M., Hogg, R.T., Gerard, R.D. & Garcia, J.A. Hypoxia increases sirtuin 1 expression in a hypoxia-inducible factor-dependent manner. J. Biol. Chem. 286, 13869–13878 (2011).
McNaney, C.A. et al. An automated liquid chromatography-mass spectrometry process to determine metabolic stability half-life and intrinsic clearance of drug candidates by substrate depletion. Assay Drug Dev. Technol. 6, 121–129 (2008).
Drexler, D.M. et al. An automated high throughput liquid chromatography-mass spectrometry process to assess the metabolic stability of drug candidates. Assay Drug Dev. Technol. 5, 247–264 (2007).
Acknowledgements
The authors thank S. Wang and members of the University of Texas Southwestern High-Throughput Screening Core Facility, D. Tomchick, C. Brautigam, J. MacMillan, N. Williams and members of our laboratories for their help. This work was funded by grants from the US National Institutes of Health (P01 CA095471, P30 CA142543) and the Cancer Prevention Research Institute of Texas (RP-100846). R.K.B. is the Michael L. Rosenberg Scholar in Medical Research and was supported by a Career Award in the Biomedical Sciences from the Burroughs Wellcome Fund; K.H.G. is the Virginia Lazenby O'Hara Chair in Biochemistry and W.W. Caruth Scholar in Biomedical Research; and U.K.T. is a W.W. Caruth Jr. Scholar in Biomedical Research. J.A.G. was supported by funds provided by the Department of Veterans Affairs. Results shown in this report are derived from work performed at Argonne National Laboratory, Structural Biology Center at the Advanced Photon Source. Argonne National Laboratory is operated by UChicago Argonne, LLC, for the US Department of Energy, Office of Biological and Environmental Research under contract DE-AC02-06CH11357. This investigation was conducted in a facility constructed with support from the Research Facilities Improvement Program (grant no. C06 RR 15437-01) from the National Center for Research Resources, US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
R.K.B. and K.H.G. conceived and designed the experiments. T.H.S., Q.L., H.-W.M., L.Z., R.C., J.N. and J.L. performed the experiments. R.K.B., K.H.G., T.H.S., J.K., J.A.G., D.E.F. and U.K.T. analyzed the data. R.K.B., K.H.G. and T.H.S. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
Declaration: R.K.B., D.E.F., K.H.G., T.H.S. and U.K.T. have filed US Patent Application no. 61/583,662 covering the method described in the paper. R.K.B., K.H.G., T.H.S., J.K. and U.K.T. have received stock options and other financial compensation from Peloton Therapeutics, Inc.
Supplementary information
Supplementary Text and Figures
Supplementary Note, Supplementary Results (PDF 3816 kb)
Rights and permissions
About this article
Cite this article
Scheuermann, T., Li, Q., Ma, HW. et al. Allosteric inhibition of hypoxia inducible factor-2 with small molecules. Nat Chem Biol 9, 271–276 (2013). https://doi.org/10.1038/nchembio.1185
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1185
This article is cited by
-
Targeting hypoxia-inducible factors: therapeutic opportunities and challenges
Nature Reviews Drug Discovery (2023)
-
Discovery of novel direct small-molecule inhibitors targeting HIF-2α using structure-based virtual screening, molecular dynamics simulation, and MM-GBSA calculations
Molecular Diversity (2023)
-
Monoterpenoid aryl hydrocarbon receptor allosteric antagonists protect against ultraviolet skin damage in female mice
Nature Communications (2023)
-
The hypoxia-driven crosstalk between tumor and tumor-associated macrophages: mechanisms and clinical treatment strategies
Molecular Cancer (2022)
-
Identification of oleoylethanolamide as an endogenous ligand for HIF-3α
Nature Communications (2022)