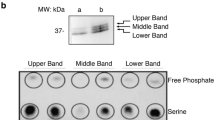
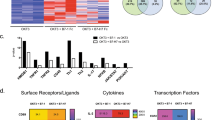
Abstract
To drive lymphocyte proliferation and differentiation, common γ-chain (γc) cytokine receptors require hours to days of sustained stimulation. JAK1 and JAK3 kinases are found together in all γc-receptor complexes, but how their respective catalytic activities contribute to signaling over time is not known. Here we dissect the temporal requirements for JAK3 kinase activity with a selective covalent inhibitor (JAK3i). By monitoring phosphorylation of the transcription factor STAT5 over 20 h in CD4+ T cells stimulated with interleukin 2 (IL-2), we document a second wave of signaling that is much more sensitive to JAK3i than the first wave. Selective inhibition of this second wave is sufficient to block cyclin expression and entry to S phase. An inhibitor-resistant JAK3 mutant (C905S) rescued all effects of JAK3i in isolated T cells and in mice. Our chemical genetic toolkit elucidates a biphasic requirement for JAK3 kinase activity in IL-2–driven T cell proliferation and will find broad utility in studies of γc-receptor signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rochman, Y., Spolski, R. & Leonard, W.J. New insights into the regulation of T cells by γ(c) family cytokines. Nat. Rev. Immunol. 9, 480–490 (2009).
Miyazaki, T. et al. Functional activation of Jak1 and Jak3 by selective association with IL-2 receptor subunits. Science 266, 1045–1047 (1994).
Ghoreschi, K., Laurence, A. & O'Shea, J.J. Janus kinases in immune cell signaling. Immunol. Rev. 228, 273–287 (2009).
Liao, W., Lin, J.-X. & Leonard, W.J. Interleukin-2 at the crossroads of effector responses, tolerance, and immunotherapy. Immunity 38, 13–25 (2013).
Lin, J.-X. et al. Critical Role of STAT5 transcription factor tetramerization for cytokine responses and normal immune function. Immunity 36, 586–599 (2012).
Laurence, A. et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 26, 371–381 (2007).
Lockyer, H.M., Tran, E. & Nelson, B.H. STAT5 is essential for Akt/p70S6 kinase activity during IL-2-induced lymphocyte proliferation. J. Immunol. 179, 5301–5308 (2007).
Yao, Z. et al. Stat5a/b are essential for normal lymphoid development and differentiation. Proc. Natl. Acad. Sci. USA 103, 1000–1005 (2006).
Thomis, D.C., Gurniak, C.B., Tivol, E., Sharpe, A.H. & Berg, L.J. Defects in B lymphocyte maturation and T lymphocyte activation in mice lacking Jak3. Science 270, 794–797 (1995).
Thomis, D.C. & Berg, L.J. Peripheral expression of Jak3 is required to maintain T lymphocyte function. J. Exp. Med. 185, 197–206 (1997).
Witthuhn, B.A., Williams, M.D., Kerawalla, H. & Uckun, F.M. Differential substrate recognition capabilities of Janus family protein tyrosine kinases within the interleukin 2 receptor (IL2R) system: Jak3 as a potential molecular target for treatment of leukemias with a hyperactive Jak-Stat signaling machinery. Leuk. Lymphoma 32, 289–297 (1999).
Haan, C. et al. Jak1 has a dominant role over Jak3 in signal transduction through γc-containing cytokine receptors. Chem. Biol. 18, 314–323 (2011).
Perona-Wright, G., Mohrs, K. & Mohrs, M. Sustained signaling by canonical helper T cell cytokines throughout the reactive lymph node. Nat. Immunol. 11, 520–526 (2010).
Swainson, L. et al. IL-7-induced proliferation of recent thymic emigrants requires activation of the PI3K pathway. Blood 109, 1034–1042 (2007).
Liao, W. et al. Opposing actions of IL-2 and IL-21 on Th9 differentiation correlate with their differential regulation of BCL6 expression. Proc. Natl. Acad. Sci. USA 111, 3508–3513 (2014).
Arneja, A., Johnson, H., Gabrovsek, L., Lauffenburger, D.A. & White, F.M. Qualitatively different T cell phenotypic responses to IL-2 versus IL-15 are unified by identical dependences on receptor signal strength and duration. J. Immunol. 192, 123–135 (2014).
Cantrell, D.A. & Smith, K.A. The interleukin-2 T-cell system: a new cell growth model. Science 224, 1312–1316 (1984).
London, N. et al. Covalent docking of large libraries for the discovery of chemical probes. Nat. Chem. Biol. 10, 1066–1072 (2014).
Goedken, E.R. et al. Tricyclic covalent inhibitors selectively target Jak3 through an active site thiol. J. Biol. Chem. 290, 4573–4589 (2015).
Tan, L. et al. Development of selective covalent Janus kinase 3 inhibitors. J. Med. Chem. 58, 6589–6606 (2015).
Clark, J.D., Flanagan, M.E. & Telliez, J.-B. Discovery and development of Janus kinase (JAK) inhibitors for inflammatory diseases. J. Med. Chem. 57, 5023–5038 (2014).
Readinger, J.A., Mueller, K.L., Venegas, A.M., Horai, R. & Schwartzberg, P.L. Tec kinases regulate T-lymphocyte development and function: new insights into the roles of Itk and Rlk/Txk. Immunol. Rev. 228, 93–114 (2009).
Ahearn, S.P. et al. Pyrrolopyrimidines as Janus kinase inhibitors. WIPO patent WO085802A1 (2013).
Brennan, P. et al. Phosphatidylinositol 3-kinase couples the interleukin-2 receptor to the cell cycle regulator E2F. Immunity 7, 679–689 (1997).
Cornish, G.H., Sinclair, L.V. & Cantrell, D.A. Differential regulation of T-cell growth by IL-2 and IL-15. Blood 108, 600–608 (2006).
Schaeffer, E.M. et al. Requirement for Tec kinases Rlk and Itk in T cell receptor signaling and immunity. Science 284, 638–641 (1999).
Rowell, E.A. & Wells, A.D. The role of cyclin-dependent kinases in T-cell development, proliferation, and function. Crit. Rev. Immunol. 26, 189–212 (2006).
Wormald, S. et al. The comparative roles of suppressor of cytokine signaling-1 and -3 in the inhibition and desensitization of cytokine signaling. J. Biol. Chem. 281, 11135–11143 (2006).
Cornish, A.L. et al. Suppressor of cytokine signaling-1 regulates signaling in response to interleukin-2 and other γ c-dependent cytokines in peripheral T cells. J. Biol. Chem. 278, 22755–22761 (2003).
Yang, X.O. et al. The signaling suppressor CIS controls proallergic T cell development and allergic airway inflammation. Nat. Immunol. 14, 732–740 (2013).
Babon, J.J. et al. Suppression of cytokine signaling by SOCS3: characterization of the mode of inhibition and the basis of its specificity. Immunity 36, 239–250 (2012).
Dowty, M.E. et al. Preclinical to clinical translation of tofacitinib, a Janus kinase inhibitor, in rheumatoid arthritis. J. Pharmacol. Exp. Ther. 348, 165–173 (2014).
Xing, L. et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat. Med. 20, 1043–1049 (2014).
Ghoreschi, K. et al. Modulation of innate and adaptive immune responses by tofacitinib (CP-690,550). J. Immunol. 186, 4234–4243 (2011).
Krutzik, P.O. & Nolan, G.P. Fluorescent cell barcoding in flow cytometry allows high-throughput drug screening and signaling profiling. Nat. Methods 3, 361–368 (2006).
Acknowledgements
We thank B. Au-Yeung, J. Carelli, H. Wang and K. Shokat for helpful discussion and suggestions and A. Roque for animal husbandry. This work was funded by the Rosalind Russell and Ephraim P. Engleman Rheumatology Research Center (A.W.) and the Howard Hughes Medical Institute (A.W. and J.T.). G.A.S. was supported by the University of California, San Francisco Medical Scientist Training Program and the US National Institute of Allergy and Infectious Diseases (F30AI120517-01).
Author information
Authors and Affiliations
Contributions
G.A.S. designed experiments, performed chemical synthesis and biological experiments and wrote the manuscript. K.U. performed chemical synthesis. A.W. designed experiments and wrote the manuscript. J.T. designed experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1–6 and Supplementary Figures 1–12. (PDF 3570 kb)
Supplementary Note
Synthetic Procedures (PDF 151 kb)
Rights and permissions
About this article
Cite this article
Smith, G., Uchida, K., Weiss, A. et al. Essential biphasic role for JAK3 catalytic activity in IL-2 receptor signaling. Nat Chem Biol 12, 373–379 (2016). https://doi.org/10.1038/nchembio.2056
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2056
This article is cited by
-
Breakthrough of solid tumor treatment: CAR-NK immunotherapy
Cell Death Discovery (2024)
-
Therapeutic modulation of JAK-STAT, mTOR, and PPAR-γ signaling in neurological dysfunctions
Journal of Molecular Medicine (2023)
-
Generation of a chemical genetic model for JAK3
Scientific Reports (2021)
-
Role of intracellular signaling pathways and their inhibitors in the treatment of inflammation
Inflammopharmacology (2021)
-
Comparative Efficacy and Safety of Peficitinib 25, 50, 100, and 150 mg in Patients with Active Rheumatoid Arthritis: A Bayesian Network Meta-Analysis of Randomized Controlled Trials
Clinical Drug Investigation (2020)