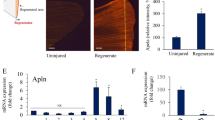
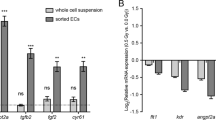
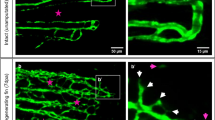
Abstract
We examined the role of angiogenesis and the need for receptor signaling using chemical inhibition of the vascular endothelial growth factor receptor in the adult zebrafish tail fin. Using a small-molecule inhibitor, we were able to exert precise control over blood vessel regeneration. An angiogenic limit to tissue regeneration was determined, as avascular tissue containing skin, pigment, neuronal axons and bone precursors could regenerate up to about 1 mm. This indicates that tissues can regenerate without direct interaction with endothelial cells and at a distance from blood supply. We also investigated whether the effects of chemical inhibition could be enhanced in zebrafish vascular mutants. We found that adult zebrafish, heterozygous for a mutation in the critical receptor effector phospholipase Cγ1, show a greater sensitivity to chemical inhibition. This study illustrates the utility of the adult zebrafish as a new model system for receptor signaling and chemical biology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chanda, S.K. Fulfilling the promise: drug discovery in the post-genomic era. Drug Discov. Today 8, 168–174 (2003).
Drews, J. Drug discovery: a historical perspective. Science 287, 1960–1964 (2000).
Lenz, G.R. Chemical ligands, genomics, and drug discovery. Drug Discov. Today 5, 145–156 (2000).
Peterson, R.T., Link, B.A., Dowling, J.E. & Schreiber, S.L. Small molecule developmental screens reveal the logic and timing of vertebrate development. Proc. Natl. Acad. Sci. USA 97, 12965–12969 (2000).
Zon, L.I. & Peterson, R.T. In vivo drug discovery in the zebrafish. Nat. Rev. Drug Discov. 4, 35–44 (2005).
Poss, K.D., Keating, M.T. & Nechiporuk, A. Tales of regeneration in zebrafish. Dev. Dyn. 226, 202–210 (2003).
Akimenko, M.A., Mari-Beffa, M., Becerra, J. & Geraudie, J. Old questions, new tools, and some answers to the mystery of fin regeneration. Dev. Dyn. 226, 190–201 (2003).
Reginelli, A.D., Wang, Y.Q., Sassoon, D. & Muneoka, K. Digit tip regeneration correlates with regions of Msx1 (Hox 7) expression in fetal and newborn mice. Development 121, 1065–1076 (1995).
Han, M., Yang, X., Farrington, J.E. & Muneoka, K. Digit regeneration is regulated by Msx1 and BMP4 in fetal mice. Development 130, 5123–5132 (2003).
Akimenko, M.A., Johnson, S.L., Westerfield, M. & Ekker, M. Differential induction of four msx homeobox genes during fin development and regeneration in zebrafish. Development 121, 347–357 (1995).
Laforest, L. et al. Involvement of the sonic hedgehog, patched 1 and bmp2 genes in patterning of the zebrafish dermal fin rays. Development 125, 4175–4184 (1998).
Nechiporuk, A. & Keating, M.T. A proliferation gradient between proximal and msxb-expressing distal blastema directs zebrafish fin regeneration. Development 129, 2607–2617 (2002).
Keating, M.T. Genetic approaches to disease and regeneration. Phil. Trans. R. Soc. Lond. B 359, 795–798 (2004).
Ferrara, N., Gerber, H.P. & LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 9, 669–676 (2003).
Bold, G. et al. New anilinophthalazines as potent and orally well absorbed inhibitors of the VEGF receptor tyrosine kinases useful as antagonists of tumor-driven angiogenesis. J. Med. Chem. 43, 2310–2323 (2000).
Wood, J.M. et al. PTK787/ZK 222584, a novel and potent inhibitor of vascular endothelial growth factor receptor tyrosine kinases, impairs vascular endothelial growth factor-induced responses and tumor growth after oral administration. Cancer Res. 60, 2178–2189 (2000).
Poss, K.D., Wilson, L.G. & Keating, M.T. Heart regeneration in zebrafish. Science 298, 2188–2190 (2002).
Huang, C.C., Lawson, N.D., Weinstein, B.M. & Johnson, S.L. reg6 is required for branching morphogenesis during blood vessel regeneration in zebrafish caudal fins. Dev. Biol. 264, 263–274 (2003).
Lawson, N. & Weinstein, B. In vivo imaging of embryonic vascular development using transgenic zebrafish. Dev. Biol. 248, 307–318 (2002).
Gerhardt, H. et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell Biol. 161, 1163–1177 (2003).
Manley, P.W. et al. Advances in the structural biology, design and clinical development of VEGF-R kinase inhibitors for the treatment of angiogenesis. Biochim. Biophys. Acta 1697, 17–27 (2004).
Chan, J., Bayliss, P.E., Wood, J.M. & Roberts, T.M. Dissection of angiogenic signaling in zebrafish using a chemical genetic approach. Cancer Cell 1, 257–267 (2002).
Parichy, D.M., Rawls, J.F., Pratt, S.J., Whitfield, T.T. & Johnson, S.L. Zebrafish sparse corresponds to an orthologue of c-kit and is required for the morphogenesis of a subpopulation of melanocytes, but is not essential for hematopoiesis or primordial germ cell development. Development 126, 3425–3436 (1999).
McKim, J., Schmieder, P. & Veith, G. Absorption dynamics of organic chemical transport across trout gills as related to octanol-water partition coefficient. Toxicol. Appl. Pharmacol. 77, 1–10 (1985).
Poss, K.D. et al. Roles for Fgf signaling during zebrafish fin regeneration. Dev. Biol. 222, 347–358 (2000).
Quint, E. et al. Bone patterning is altered in the regenerating zebrafish caudal fin after ectopic expression of sonic hedgehog and bmp2b or exposure to cyclopamine. Proc. Natl. Acad. Sci. USA 99, 8713–8718 (2002).
Metcalfe, W.K., Myers, P.Z., Trevarrow, B., Bass, M.B. & Kimmel, C.B. Primary neurons that express the L2/HNK-1 carbohydrate during early development in the zebrafish. Development 110, 491–504 (1990).
Johnson, S.L. & Weston, J.A. Temperature-sensitive mutations that cause stage-specific defects in Zebrafish fin regeneration. Genetics 141, 1583–1595 (1995).
LeCouter, J. et al. Angiogenesis-independent endothelial protection of liver: role of VEGFR-1. Science 299, 890–893 (2003).
Gerhardt, H. & Betsholtz, C. Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res. 314, 15–23 (2003).
Cleaver, O. & Melton, D.A. Endothelial signaling during development. Nat. Med. 9, 661–668 (2003).
Cross, L.M., Cook, M.A., Lin, S., Chen, J.N. & Rubinstein, A.L. Rapid analysis of angiogenesis drugs in a live fluorescent zebrafish assay. Arterioscler. Thromb. Vasc. Biol. 23, 911–912 (2003).
Liao, A.T. et al. Inhibition of constitutively active forms of mutant kit by multitargeted indolinone tyrosine kinase inhibitors. Blood 100, 585–593 (2002).
Fabian, M.A. et al. A small molecule-kinase interaction map for clinical kinase inhibitors. Nat. Biotechnol. 23, 329–336 (2005).
Arap, W. et al. Steps toward mapping the human vasculature by phage display. Nat. Med. 8, 121–127 (2002).
Chi, J.T. et al. Endothelial cell diversity revealed by global expression profiling. Proc. Natl. Acad. Sci. USA 100, 10623–10628 (2003).
Lawson, N.D., Mugford, J.W., Diamond, B.A. & Weinstein, B.M. Phospholipase Cγ-1 is required downstream of vascular endothelial growth factor during arterial development. Genes Dev. 17, 1346–1351 (2003).
Weinstein, B.M., Stemple, D.L., Driever, W. & Fishman, M.C. Gridlock, a localized heritable vascular patterning defect in the zebrafish. Nat. Med. 1, 1143–1147 (1995).
Zhong, T.P. gridlock, an HLH gene required for assembly of the aorta in zebrafish. Science 287, 1820–1824 (2000).
Stainier, D.Y., Weinstein, B.M., Detrich, H.W., III, Zon, L.I. & Fishman, M.C. Cloche, an early acting zebrafish gene, is required by both the endothelial and hematopoietic lineages. Development 121, 3141–3150 (1995).
Peterson, R.T. et al. Chemical suppression of a genetic mutation in a zebrafish model of aortic coarctation. Nat. Biotechnol. 22, 595–599 (2004).
Zhong, T.P. Gridlock signalling pathway fashions the first embryonic artery. Nature 414, 216–220 (2001).
Takahashi, T., Yamaguchi, S., Chida, K. & Shibuya, M. A single autophosphorylation site on KDR/Flk-1 is essential for VEGF-A-dependent activation of PLC-γ and DNA synthesis in vascular endothelial cells. EMBO J. 20, 2768–2778 (2001).
Sakurai, Y., Ohgimoto, K., Kataoka, Y., Yoshida, N. & Shibuya, M. Essential role of Flk-1 (VEGF receptor 2) tyrosine residue 1173 in vasculogenesis in mice. Proc. Natl. Acad. Sci. USA 102, 1076–1081 (2005).
Liao, W. et al. The zebrafish gene cloche acts upstream of a flk-1 homologue to regulate endothelial cell differentiation. Development 124, 381–389 (1997).
Westerfield, M. The Zebrafish Book: Guide for the Laboratory Use of Zebrafish (Danio rerio) (Univ. of Oregon Press, Eugene, Oregon, USA, 1995).
Morikawa, S. et al. Abnormalities in pericytes on blood vessels and endothelial sprouts in tumors. Am. J. Pathol. 160, 985–1000 (2002).
Chan, J. et al. Morphogenesis of prechordal plate and notochord requires intact Eph/ephrin B signaling. Dev. Biol. 234, 470–482 (2001).
Acknowledgements
We thank J. Folkman, N. Lawson, R. Peterson, J. Mably, F. Serluca, G. Naumov and C. Barnes for helpful discussions and/or critical reading of the manuscript. We acknowledge M. Lin and R. Bolcome for technical assistance with zebrafish care and maintenance and K. Johnson for help with graphics. We thank L. Trakimas for electron microscopy work. This work was supported in part by sponsored research agreements from Novartis to T.M.R. and J.C. and by an award from the Sidney Kimmel Foundation for Cancer Research to J.C. J.C is a Kimmel Cancer Scholar.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J.C. and T.M.R. receive support for research programs from Novartis. T.M.R. has consulting relationships with Upstate Biotechnology Inc. and Novartis Pharmaceuticals Inc. J.W. and T.O. are employees of Novartis while engaged in this research project. K.B. and M.K. are current employees of Novartis whose employment began after the completion of their involvement in this project. In addition, J.W., T.O and M.K. have personal financial interests in Novartis Pharmaceuticals Inc.
Supplementary information
Supplementary Fig. 1
Nascent vessels are more susceptible to VEGFR inhibition. (PDF 6147 kb)
Supplementary Fig. 2
Real-time PCR analysis of the expression of selected genes during fin regeneration and baseline levels. (PDF 1026 kb)
Supplementary Table 1
Targeted genes and primers used for real time PCR analysis. (PDF 50 kb)
Supplementary Table 2
Summary of statistical evaluation of the effect of genetic background and PTK787 treatment on zebrafish tail fin regeneration. (PDF 43 kb)
Supplementary Video 1
Zebrafish movie showing caudal fin circulation. (MOV 8844 kb)
Rights and permissions
About this article
Cite this article
Bayliss, P., Bellavance, K., Whitehead, G. et al. Chemical modulation of receptor signaling inhibits regenerative angiogenesis in adult zebrafish. Nat Chem Biol 2, 265–273 (2006). https://doi.org/10.1038/nchembio778
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio778
This article is cited by
-
Apela promotes blood vessel regeneration and remodeling in zebrafish
Scientific Reports (2024)
-
Understanding the process of angiogenesis in regenerating earthworm
In Vitro Cellular & Developmental Biology - Animal (2023)
-
Low doses of ionizing radiation enhance angiogenesis and consequently accelerate post-embryonic development but not regeneration in zebrafish
Scientific Reports (2020)
-
A Novel In Vivo Model to Study Impaired Tissue Regeneration Mediated by Cigarette Smoke
Scientific Reports (2018)
-
Comparative regenerative mechanisms across different mammalian tissues
npj Regenerative Medicine (2018)