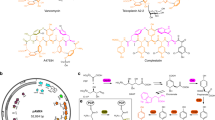
Abstract
Nonribosomal peptides (NRPs) are a class of microbial secondary metabolites that have a wide variety of medicinally important biological activities, such as antibiotic (vancomycin), immunosuppressive (cyclosporin A), antiviral (luzopeptin A) and antitumor (echinomycin and triostin A) activities1,2. However, many microbes are not amenable to cultivation and require time-consuming empirical optimization of incubation conditions for mass production of desired secondary metabolites for clinical and commercial use3. Therefore, a fast, simple system for heterologous production of natural products is much desired. Here we show the first example of the de novo total biosynthesis of biologically active forms of heterologous NRPs in Escherichia coli. Our system can serve not only as an effective and flexible platform for large-scale preparation of natural products from simple carbon and nitrogen sources, but also as a general tool for detailed characterizations and rapid engineering of biosynthetic pathways for microbial syntheses of novel compounds and their analogs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schwarzer, D., Finking, R. & Marahiel, M.A. Nonribosomal peptides: from genes to products. Nat. Prod. Rep. 20, 275–287 (2003).
Boger, D.L., Ichikawa, S., Tse, W.C., Hedrick, M.P. & Jin, Q. Total syntheses of thiocoraline and BE-22179 and assessment of their DNA binding and biological properties. J. Am. Chem. Soc. 123, 561–568 (2001).
Shuler, M.L. Bioreactor engineering as an enabling technology to tap biodiversity. The case of taxol. Ann. NY Acad. Sci. 745, 455–461 (1994).
Steinerova, N. et al. Production of quinomycin A in Streptomyces lasaliensis. Folia Microbiol. (Praha) 32, 1–5 (1987).
Reid, D.G., Doddrell, D.M., Williams, D.H. & Fox, K.R. A 15N nuclear magnetic resonance study of the biosynthesis of quinoxaline antibiotics. Biochim. Biophys. Acta 798, 111–114 (1984).
Chen, H., Hubbard, B.K., O'Connor, S.E. & Walsh, C.T. Formation of beta-hydroxy histidine in the biosynthesis of nikkomycin antibiotics. Chem. Biol. 9, 103–112 (2002).
Kurnasov, O. et al. Aerobic tryptophan degradation pathway in bacteria: novel kynurenine formamidase. FEMS Microbiol. Lett. 227, 219–227 (2003).
Schmoock, G. et al. Functional cross-talk between fatty acid synthesis and nonribosomal peptide synthesis in quinoxaline antibiotic-producing streptomycetes. J. Biol. Chem. 280, 4339–4349 (2005).
Trauger, J.W., Kohli, R.M., Mootz, H.D., Marahiel, M.A. & Walsh, C.T. Peptide cyclization catalysed by the thioesterase domain of tyrocidine synthetase. Nature 407, 215–218 (2000).
Cornish, A., Waring, M.J. & Nolan, R.D. Conversion of triostins to quinomycins by protoplasts of Streptomyces echinatus. J. Antibiot. (Tokyo) 36, 1664–1670 (1983).
Takahashi, K., Koshino, H., Esumi, Y., Tsuda, E. & Kurosawa, K. SW-163C and E novel antitumor depsipeptides produced by Streptomyces sp.II. Structure elucidation. J. Antibiot. (Tokyo) 54, 622–627 (2001).
Rance, M.J. et al. UK-63,052 complex, new quinomycin antibiotics from Streptomyces braegensis subsp. japonicus; taxonomy, fermentation, isolation, characterisation and antimicrobial activity. J. Antibiot. (Tokyo) 42, 206–217 (1989).
Barrett, A.G., Hamprecht, D., White, A.J. & Williams, D.J. Iterative cyclopropanation: a concise strategy for the total synthesis of the hexacyclopropane cholesteryl ester transfer protein inhibitor U-106305. J. Am. Chem. Soc. 119, 8608–8615 (1997).
Van Lanen, S.G. & Iwata-Reuyl, D. Kinetic mechanism of the tRNA-modifying enzyme S-adenosylmethionine:tRNA ribosyltransferase-isomerase (QueA). Biochemistry 42, 5312–5320 (2003).
Ciardelli, T.L., Chakravarty, P.K. & Olsen, R.K. Des-N-tetramethyltriostin A and bis-L-seryldes-N-tetramethyltriostin A, synthetic analogues of the quinoxaline antibiotics. J. Am. Chem. Soc. 100, 7684–7690 (1978).
Lambalot, R.H. et al. A new enzyme superfamily - the phosphopantetheinyl transferases. Chem. Biol. 3, 923–936 (1996).
Sørensen, H.P. & Mortensen, K.K. Advanced genetic strategies for recombinant protein expression in Escherichia coli. J. Biotechnol. 115, 113–128 (2005).
Lau, J., Tran, C., Licari, P. & Galazzo, J. Development of a high cell-density fed-batch bioprocess for the heterologous production of 6-deoxyerythronolide B in Escherichia coli. J. Biotechnol. 110, 95–103 (2004).
Furuya, K. & Hutchinson, C.R. The DrrC protein of Streptomyces peucetius, a UvrA-like protein, is a DNA-binding protein whose gene is induced by daunorubicin. FEMS Microbiol. Lett. 168, 243–249 (1998).
Neidle, S., Pearl, L.H. & Skelly, J.V. DNA structure and perturbation by drug binding. Biochem. J. 243, 1–13 (1987).
Royo, J.L., Moreno-Ruiz, E., Cebolla, A. & Santero, E. Stable long-term indigo production by overexpression of dioxygenase genes using a chromosomal integrated cascade expression circuit. J. Biotechnol. 116, 113–124 (2005).
Watanabe, K., Rude, M.A., Walsh, C.T. & Khosla, C. Engineered biosynthesis of an ansamycin polyketide precursor in Escherichia coli. Proc. Natl. Acad. Sci. USA 100, 9774–9778 (2003).
Peiru, S., Menzella, H.G., Rodriguez, E., Carney, J. & Gramajo, H. Production of the potent antibacterial polyketide erythromycin C in Escherichia coli. Appl. Environ. Microbiol. 71, 2539–2547 (2005).
Pfeifer, B.A., Wang, C.C., Walsh, C.T. & Khosla, C. Biosynthesis of yersiniabactin, a complex polyketide-nonribosomal peptide, using Escherichia coli as a heterologous host. Appl. Environ. Microbiol. 69, 6698–6702 (2003).
Watanabe, K., Wang, C.C., Boddy, C.N., Cane, D.E. & Khosla, C. Understanding substrate specificity of polyketide synthase modules by generating hybrid multimodular synthases. J. Biol. Chem. 278, 42020–42026 (2003).
Gruenewald, S., Mootz, H.D., Stehmeier, P. & Stachelhaus, T. In vivo production of artificial nonribosomal peptide products in the heterologous host Escherichia coli. Appl. Environ. Microbiol. 70, 3282–3291 (2004).
Cheung, H.T. et al. The conformation of echinomycin in solution. J. Am. Chem. Soc. 100, 46–54 (1978).
Blake, T.J., Kalman, J.R. & Williams, D.H. Two symmetrical conformations of the triostin antibiotics in solution. Tetrahedr. Lett. 18, 2621–2624 (1977).
McGinnis, S. & Madden, T.L. BLAST: at the core of a powerful and diverse set of sequence analysis tools. Nucleic Acids Res. 32, W20–W25 (2004).
Dell, A. et al. Structure revision of the antibiotic echinomycin. J. Am. Chem. Soc. 97, 2497–2502 (1975).
Acknowledgements
We thank M.J. Waring and M. Searcey for kindly providing the authentic reference samples of triostin A and TANDEM, respectively, and H. Kinashi (Hiroshima University) for advice in handling the streptomycete linear plasmid. This work was financially supported by Grant-in-Aids for Scientific Research from the Japan Society for the Promotion of Science (JSPS) (A) 17208010 (H.O.), by Uehara Memorial Foundation PK220920 (H.O.), by National Institute of General Medical Sciences grant GM 075857-01 (C.C.C.W.) and by American Cancer Society grant RSG-06-010-01-CDD (C.C.C.W.). K.W. is a recipient of fellowships from the Japan Antibiotics Research Association, Pfizer Infectious Disease Foundation and the Agricultural Chemical Research Foundation. A.M. is a recipient of JSPS predoctoral fellowship 177009104.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
SDS-PAGE analysis of the purified E. coli-produced echinomycin biosynthetic proteins. (PDF 2970 kb)
Supplementary Fig. 2
Maps of the plasmid-borne echinomycin biosynthetic gene cluster. (PDF 84 kb)
Supplementary Fig. 3
Assays for checking the maintenance and stability of the plasmids carrying the echinomycin biosynthetic cluster in E. coli DH5α. (PDF 2878 kb)
Supplementary Fig. 4
Chemical characteristic spectra of the authentic reference for echinomycin. (PDF 161 kb)
Supplementary Fig. 5
Chemical characteristic spectra of the authentic reference for triostin A. (PDF 167 kb)
Supplementary Methods
Methods used for echinomycin biosynthetic cluster isolation and a table of the sequences of the primers used in this manuscript. (PDF 147 kb)
Rights and permissions
About this article
Cite this article
Watanabe, K., Hotta, K., Praseuth, A. et al. Total biosynthesis of antitumor nonribosomal peptides in Escherichia coli. Nat Chem Biol 2, 423–428 (2006). https://doi.org/10.1038/nchembio803
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio803
This article is cited by
-
Structural and functional analyses of the echinomycin resistance conferring protein Ecm16 from Streptomyces lasalocidi
Scientific Reports (2023)
-
Amino acid (acyl carrier protein) ligase-associated biosynthetic gene clusters reveal unexplored biosynthetic potential
Molecular Genetics and Genomics (2023)
-
Structural basis for backbone N-methylation by an interrupted adenylation domain
Nature Chemical Biology (2018)
-
Improved production of 1-deoxynojirymicin in Escherichia coli through metabolic engineering
World Journal of Microbiology and Biotechnology (2018)
-
Scale-up bioprocess development for production of the antibiotic valinomycin in Escherichia coli based on consistent fed-batch cultivations
Microbial Cell Factories (2015)