Abstract
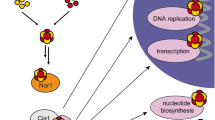
Biogenesis of iron-sulfur ([Fe-S]) proteins in eukaryotes requires the function of complex proteinaceous machineries in both mitochondria and cytosol. In contrast to the mitochondrial pathway, little is known about [Fe-S] protein assembly in the cytosol. So far, four highly conserved proteins (Cfd1, Nbp35, Nar1 and Cia1) have been identified as members of the cytosolic [Fe-S] protein assembly machinery, but their molecular function is unresolved. Using in vivo and in vitro approaches, we found that the soluble P-loop NTPases Cfd1 and Nbp35 form a complex and bind up to three [4Fe-4S] clusters, one at the N terminus of Nbp35 and one each at a new C-terminal cysteine-rich motif present in both proteins. These labile [Fe-S] clusters can be rapidly transferred and incorporated into target [Fe-S] apoproteins in a Nar1- and Cia1-dependent fashion. Our data suggest that the Cfd1–Nbp35 complex functions as a novel scaffold for [Fe-S] cluster assembly in the eukaryotic cytosol.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Beinert, H., Holm, R.H. & Münck, E. Iron-sulfur clusters: nature's modular, multipurpose structures. Science 277, 653–659 (1997).
Beinert, H., Meyer, J. & Lill, R. in Encyclopedia of Biological Chemistry Vol. 2 (eds. Lennarz, W.J. & Lane, M.D.) 482–489 (Academic, Amsterdam, 2004).
Barras, F., Loiseau, L. & Py, B. How Escherichia coli and Saccharomyces cerevisiae build Fe/S proteins. Adv. Microb. Physiol. 50, 41–101 (2005).
Johnson, D.C., Dean, D.R., Smith, A.D. & Johnson, M.K. Structure, function and formation of biological iron-sulfur clusters. Annu. Rev. Biochem. 74, 247–281 (2005).
Fontecave, M., Choudens, S.O., Py, B. & Barras, F. Mechanisms of iron-sulfur cluster assembly: the SUF machinery. J. Biol. Inorg. Chem. 10, 713–721 (2005).
Balk, J. & Lobreaux, S. Biogenesis of iron-sulfur proteins in plants. Trends Plant Sci. 10, 324–331 (2005).
Rubio, L.M. & Ludden, P.W. Maturation of nitrogenase: a biochemical puzzle. J. Bacteriol. 187, 405–414 (2005).
Lill, R. & Mühlenhoff, U. Iron-sulfur protein biogenesis in eukaryotes. Trends Biochem. Sci. 30, 133–141 (2005).
Lill, R. & Mühlenhoff, U. Iron-sulfur protein biogenesis in eukaryotes: components and mechanisms. Annu. Rev. Cell Dev. Biol. 22, 457–486 (2006).
Rouault, T.A. & Tong, W.H. Iron-sulphur cluster biogenesis and mitochondrial iron homeostasis. Nat. Rev. Mol. Cell Biol. 6, 345–351 (2005).
Roy, A., Solodovnikova, N., Nicholson, T., Antholine, W. & Walden, W.E. A novel eukaryotic factor for cytosolic Fe-S cluster assembly. EMBO J. 22, 4826–4835 (2003).
Balk, J., Pierik, A.J., Aguilar Netz, D., Mühlenhoff, U. & Lill, R. The hydrogenase-like Nar1p is essential for maturation of cytosolic and nuclear iron-sulphur proteins. EMBO J. 23, 2105–2115 (2004).
Hausmann, A. et al. The eukaryotic P-loop NTPase Nbp35: an essential component of the cytosolic and nuclear iron-sulfur protein assembly machinery. Proc. Natl. Acad. Sci. USA 102, 3266–3271 (2005).
Balk, J., Aguilar Netz, D.J., Tepper, K., Pierik, A.J. & Lill, R. The essential WD40 protein Cia1 is involved in a late step of cytosolic and nuclear iron-sulfur protein assembly. Mol. Cell. Biol. 25, 10833–10841 (2005).
Kispal, G., Csere, P., Prohl, C. & Lill, R. The mitochondrial proteins Atm1p and Nfs1p are required for biogenesis of cytosolic Fe/S proteins. EMBO J. 18, 3981–3989 (1999).
Lange, H., Kispal, G., Kaut, A. & Lill, R. A mitochondrial ferredoxin is essential for biogenesis of intra- and extra-mitochondrial Fe/S proteins. Proc. Natl. Acad. Sci. USA 97, 1050–1055 (2000).
Li, J., Saxena, S., Pain, D. & Dancis, A. Adrenodoxin reductase homolog (Arh1p) of yeast mitochondria required for iron homeostasis. J. Biol. Chem. 276, 1503–1509 (2001).
Gerber, J., Neumann, K., Prohl, C., Mühlenhoff, U. & Lill, R. The yeast scaffold proteins Isu1p and Isu2p are required inside mitochondria for maturation of cytosolic Fe/S proteins. Mol. Cell. Biol. 24, 4848–4857 (2004).
Pondarre, C. et al. The mitochondrial ATP-binding cassette transporter Abcb7 is essential in mice and participates in cytosolic iron-sulphur cluster biogenesis. Hum. Mol. Genet. 15, 953–964 (2006).
Mühlenhoff, U. et al. Functional characterization of the eukaryotic cysteine desulfurase Nfs1p from Saccharomyces cerevisiae. J. Biol. Chem. 279, 36906–36915 (2004).
Biederbick, A. et al. Role of human mitochondrial Nfs1 in cytosolic iron-sulfur protein biogenesis and iron regulation. Mol. Cell. Biol. 26, 5675–5687 (2006).
Fosset, C. et al. RNA silencing of mitochondrial m-Nfs1 reduces Fe-S enzyme activity both in mitochondria and cytosol of mammalian cells. J. Biol. Chem. 281, 25398–25406 (2006).
Ghaemmaghami, S. et al. Global analysis of protein expression in yeast. Nature 425, 737–741 (2003).
Puig, O. et al. The tandem affinity purification (TAP) method: a general procedure of protein complex purification. Methods 24, 218–229 (2001).
Skerra, A. & Schmidt, T.G. Use of the Strep-Tag and streptavidin for detection and purification of recombinant proteins. Methods Enzymol. 326, 271–304 (2000).
Orme-Johnson, W.H. & Orme-Johnson, N.R. Overview of iron-sulfur proteins. Methods Enzymol. 53, 259–268 (1978).
Agar, J.N. et al. IscU as a scaffold for iron-sulfur cluster biosynthesis: sequential assembly of [2Fe-2S] and [4Fe-4S] clusters in IscU. Biochemistry 39, 7856–7862 (2000).
Yuvaniyama, P., Agar, J.N., Cash, V.L., Johnson, M.K. & Dean, D.R. NifS-directed assembly of a transient [2Fe-2S] cluster within the NifU protein. Proc. Natl. Acad. Sci. USA 97, 599–604 (2000).
Mühlenhoff, U., Gerber, J., Richhardt, N. & Lill, R. Components involved in assembly and dislocation of iron-sulfur clusters on the scaffold protein Isu1p. EMBO J. 22, 4815–4825 (2003).
Bonomi, F., Iametti, S., Ta, D. & Vickery, L.E. Multiple turnover transfer of [2Fe2S] clusters by the iron-sulfur cluster assembly scaffold proteins IscU and IscA. J. Biol. Chem. 280, 29513–29518 (2005).
Smith, A.D. et al. NifS-mediated assembly of [4Fe-4S] clusters in the N- and C-terminal domains of the NifU scaffold protein. Biochemistry 44, 12955–12969 (2005).
Dutkiewicz, R. et al. The Hsp70 chaperone Ssq1p is dispensable for iron-sulfur cluster formation on the scaffold protein Isu1p. J. Biol. Chem. 281, 7801–7808 (2006).
Chandramouli, K. & Johnson, M.K. HscA and HscB stimulate [2Fe-2S] cluster transfer from IscU to apoferredoxin in an ATP-dependent reaction. Biochemistry 45, 11087–11095 (2006).
Chen, O.S. et al. Transcription of the yeast iron regulon responds not directly to iron but rather to iron-sulfur cluster biosynthesis. J. Biol. Chem. 279, 29513–29518 (2004).
Leipe, D.D., Wolf, Y.I., Koonin, E.V. & Aravind, L. Classification and evolution of P-loop GTPases and related ATPases. J. Mol. Biol. 317, 41–72 (2002).
Skovran, E. & Downs, D.M. Lack of the ApbC or ApbE protein results in a defect in Fe-S cluster metabolism in Salmonella enterica serovar Typhimurium. J. Bacteriol. 185, 98–106 (2003).
Lezhneva, L., Amann, K. & Meurer, J. The universally conserved HCF101 protein is involved in assembly of [4Fe-4S]-cluster-containing complexes in Arabidopsis thaliana chloroplasts. Plant J. 37, 174–185 (2004).
Stockel, J. & Oelmuller, R. A novel protein for photosystem I biogenesis. J. Biol. Chem. 279, 10243–10251 (2004).
Lill, R. & Kispal, G. Maturation of cellular Fe/S proteins: the essential function of mitochondria. Trends Biochem. Sci. 25, 352–356 (2000).
Gerber, J., Mühlenhoff, U. & Lill, R. An interaction between frataxin and Isu1/Nfs1 that is crucial for Fe/S cluster synthesis on Isu1. EMBO Rep. 4, 906–911 (2003).
Yoon, T. & Cowan, J.A. Frataxin-mediated iron delivery to ferrochelatase in the final step of heme biosynthesis. J. Biol. Chem. 279, 25943–25946 (2004).
Bencze, K.Z. et al. The structure and function of frataxin. Crit. Rev. Biochem. Mol. Biol. 41, 269–291 (2006).
Puccio, H. & Koenig, M. Friedreich ataxia: a paradigm for mitochondrial diseases. Curr. Opin. Genet. Dev. 12, 272–277 (2002).
Bekri, S. et al. Human ABC7 transporter: gene structure and mutation causing X-linked sideroblastic anemia with ataxia (XLSA/A) with disruption of cytosolic iron-sulfur protein maturation. Blood 96, 3256–3264 (2000).
Rudolf, J., Makrantoni, V., Ingledew, W.J., Stark, M.J. & White, M.F. The DNA repair helicases XPD and FancJ have essential iron-sulfur domains. Mol. Cell 23, 801–808 (2006).
Mühlenhoff, U., Richhardt, N., Ristow, M., Kispal, G. & Lill, R. The yeast frataxin homologue Yfh1p plays a specific role in the maturation of cellular Fe/S proteins. Hum. Mol. Genet. 11, 2025–2036 (2002).
Kerner, M.J. et al. Proteome-wide analysis of chaperonin-dependent protein folding in Escherichia coli. Cell 122, 209–220 (2005).
Takahashi, Y. & Nakamura, M. Functional assignment of the ORF2-iscS-iscU-iscA-hscB-hscA-fdx-ORF3 gene cluster involved in the assembly of Fe-S clusters in Escherichia coli. J. Biochem. 126, 917–926 (1999).
Floss, B., Igloi, G.L., Cassier-Chauvat, C. & Mühlenhoff, U. Molecular characterization and overexpression of the petF gene from Synechococcus elongatus: evidence for a second site of electrostatic interaction between ferredoxin and the PS I-D subunit. Photosynth. Res. 54, 63–71 (1997).
Jungermann, K., Thauer, R.K., Rupprecht, E., Ohrloff, C. & Decker, K. Ferredoxin mediated hydrogen formation from NADPH in a cell-free system of Clostridium kluyveri. FEBS Lett. 3, 144–146 (1969).
Mumberg, D., Müller, R. & Funk, M. Regulatable promoters of Saccharomyces cerevisiae: comparison of transcriptional activity and their use for heterologous expression. Nucleic Acids Res. 22, 5767–5768 (1994).
Acknowledgements
We thank R.K. Thauer (Max-Planck-Institute, Marburg) for access to the EPR spectrometer and providing purified ferredoxin, and J. Balk and W. Walden for discussion. This work was supported by grants from Deutsche Forschungsgemeinschaft (Sonderforschungsbereich 593, Graduiertenkolleg 1216, Gottfried-Wilhelm Leibniz program), the European Commission (MitEURO) and Fonds der chemischen Industrie.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Stable complex formation between Cfd1 and Nbp35. (PDF 123 kb)
Supplementary Fig. 2
Spectroscopic properties of Nbp35 and its C253A mutant. (PDF 73 kb)
Supplementary Fig. 3
The [Fe-S] clusters bound to Cfd1, Nbp35 or the Cfd1–Nbp35 complex are labile in the presence of oxygen. (PDF 103 kb)
Supplementary Table 1
Constructs used in this work. (PDF 21 kb)
Rights and permissions
About this article
Cite this article
Netz, D., Pierik, A., Stümpfig, M. et al. The Cfd1–Nbp35 complex acts as a scaffold for iron-sulfur protein assembly in the yeast cytosol. Nat Chem Biol 3, 278–286 (2007). https://doi.org/10.1038/nchembio872
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio872
This article is cited by
-
A two-hybrid system reveals previously uncharacterized protein–protein interactions within the Helicobacter pylori NIF iron–sulfur maturation system
Scientific Reports (2021)
-
The bacterial MrpORP is a novel Mrp/NBP35 protein involved in iron-sulfur biogenesis
Scientific Reports (2019)
-
Investigating the role(s) of SufT and the domain of unknown function 59 (DUF59) in the maturation of iron–sulfur proteins
Current Genetics (2018)
-
The NMR contribution to protein–protein networking in Fe–S protein maturation
JBIC Journal of Biological Inorganic Chemistry (2018)
-
Fe–S cluster assembly in the supergroup Excavata
JBIC Journal of Biological Inorganic Chemistry (2018)