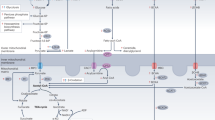
Abstract
The advent of myocardial metabolic imaging more than 30 years ago ushered in a paradigm shift in the clinical management of patients with ischemic and nonischemic heart disease. A classic example is the so-called metabolic memory of altered glucose and fatty acid metabolism in regions of myocardial ischemia and reperfusion. At the cellular level, metabolic memory is driven by changes in the activities and expression of a host of metabolic enzymes, including reactivation of the fetal gene program. The future of metabolic imaging will require a more-refined understanding of the pathways of metabolic adaptation and maladaptation of the heart. Recent evidence suggests that metabolic signals alter metabolic fluxes and give rise to specific metabolic patterns that, in turn, lead to changes in translational and/or transcriptional activities in the cardiac myocyte. In other words, metabolism provides a link between environmental stimuli and a host of intracellular signaling pathways. This concept has not yet been fully explored in vivo, although metabolic adaptation represents the earliest response to myocardial ischemia and left ventricular remodeling.
Key Points
-
Imaging of energy-providing pathways during and after an episode of myocardial ischemia might provide important insights into the pathophysiology of coronary artery disease
-
Designing metabolic imaging modalities that take into account the complexities of the pathways of energy transfer remains a challenge
-
While the normal heart readily mobilizes its metabolic reserve, metabolism is dysregulated in the failing heart and the myocardium is less responsive to inotropic stimulation
-
Nuclear imaging techniques for cardiac metabolism must be viewed in context with the principles of metabolic regulation
-
Metabolic remodeling precedes functional and structural remodeling in heart failure
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kawai Y et al. (2001) Significance of reduced uptake of iodinated fatty acid analogue for the evaluation of patients with acute chest pain. J Am Coll Cardiol 38: 1888–1894
Dilsizian V et al. (2005). Metabolic imaging with β-methyl-p-[123I]-iodophenyl-pentadecanoic acid (BMIPP) identifies ischemic memory following demand ischemia. Circulation 112: 2169–2174
Tillisch JH et al. (1986) Reversibility of cardiac wall-motion abnormalities predicted by positron tomography. N Engl J Med 314: 884–888
Taegtmeyer H (2004) Cardiac metabolism as a target for the treatment of heart failure. Circulation 110: 894–896
Taegtmeyer H (1994) Energy metabolism of the heart: from basic concepts to clinical applications. Curr Probl Cardiol 19: 59–113
Taegtmeyer H et al. (1980) Utilization of energy-providing substrates in the isolated working rat heart. Biochem J 186: 701–711
Newsholme EA and Crabtree B (1973) Metabolic aspects of enzyme activity regulation. Symp Soc Exp Biol 27: 429–460
Taegtmeyer H (2000) Genetics of energetics: transcriptional responses in cardiac metabolism. Ann Biomed Eng 28: 871–876
Taegtmeyer H et al. (2004) Linking gene expression to function: metabolic flexibility in the normal and diseased heart. Ann NY Acad Sci 1015: 202–213
Finck BN and Kelly DP (2007) Peroxisome proliferator-activated receptor γ coactivator-1 (PGC-1) regulatory cascade in cardiac physiology and disease. Circulation 115: 2540–2548
Kelly DP and Strauss AW (1994) Inherited cardiomyopathies. N Engl J Med 330: 913–919
Strauss AW et al. (1995) Molecular basis of human mitochondrial very-long-chain acyl-CoA dehydrogenase deficiency causing cardiomyopathy and sudden death in childhood. Proc Natl Acad Sci USA 92: 10496–10500
Kurtz DM et al. (1998) Targeted disruption of mouse long-chain acyl-CoA dehydrogenase gene reveals crucial roles for fatty acid oxidation. Proc Natl Acad Sci USA 95: 15592–15597
Graham BH et al. (1997) A mouse model for mitochondrial myopathy and cardiomyopathy resulting from a deficiency in the heart/muscle isoform of the adenine nucleotide translocator. Nat Genet 16: 226–234
Goodwin GW et al. (1998) Regulation of energy metabolism of the heart during acute increase in heart work. J Biol Chem 273: 29530–29539
Young ME et al. (2002) Adaptation and maladaptation of the heart in diabetes. Part II: potential mechanisms. Circulation 105: 1861–1870
Sharma S et al. (2004) Intramyocardial lipid accumulation in the failing human heart resembles the lipotoxic rat heart. FASEB J 18: 1692–1700
Rajabi M et al. (2007) Return to the fetal gene program protects the stressed heart: a strong hypothesis. Heart Fail Rev 12: 331–343
Dewald O et al. (2005) Downregulation of peroxisome proliferator-activated receptor-α gene expression in a mouse model of ischemic cardiomyopathy is dependent on reactive oxygen species and prevents lipotoxicity. Circulation 112: 407–415
Young ME et al. (2001) Reactivation of peroxisome proliferator-activated receptor α is associated with contractile dysfunction in hypertrophied rat heart. J Biol Chem 276: 44390–44395
Zhou YT et al. (2000) Lipotoxic heart disease in obese rats: implications for human obesity. Proc Natl Acad Sci USA 97: 1784–1789
Schaffer JE (2003) Lipotoxicity: when tissues overeat. Curr Opin Lipidol 14: 281–287
Fallavollita JA et al. (2003) Hibernating myocardiam retains metabolic and contractile reserve despite regional reductions in flow, function, and oxygen consumption at rest. Circ Res 92: 48–55
Mäki M et al. (1996) Glucose uptake in the chronically dysfunctional but viable myocardium. Circulation 93: 1658–1666
Chen TM et al. (1997) Effects of insulin on glucose uptake by rat hearts during and after coronary flow reduction. Am J Physiol 273: H2170–H2177
Herrero P et al. (2007) PET measurements of myocardial glucose metabolism with 1-11C-glucose and kinetic modeling. J Nucl Med 48: 955–964
Messina SA et al. (2007) Delayed recovery of fatty acid metabolism after transient myocardial ischemia: a potential imaging target for “ischemic memory”. Curr Cardiol Rep 9: 159–165
Depre C and Taegtmeyer H (2000) Metabolic aspects of programmed cell survival and cell death in the heart. Cardiovasc Res 45: 538–548
Bertomeu-Gonzalez V et al. (2006) Role of trimetazidine in management of ischemic cardiomyopathy. Am J Cardiol 98 (Suppl 1): 19J–124J
Bennett SK et al. (2002) Effect of metoprolol on absolute myocardial blood flow in patients with heart failure secondary to ischemic or non-ischemic cardiomyopathy. Am J Cardiol 89: 1431–1434
Wallhaus TR et al. (2001) Myocardial free fatty acid and glucose use after carvedilol treatment in patients with congestive heart failure. Circulation 103: 2441–2446
McVeigh JJ and Lopaschuk GD (1990) Dichloroacetate stimulation of glucose oxidation improves recovery of ischemic rat hearts. Am J Physiol 259: H1079–H1085
Broderick TL et al. (1992) Carnitine stimulation of glucose oxidation in the fatty acid perfused isolated working rat heart. J Biol Chem 267: 3758–3763
McCormack JG et al. (1996) Ranolazine stimulates glucose oxidation in normoxic, ischemic, and reperfused ischemic rat hearts. Circulation 93: 135–142
Thomas L (1974) The technology of medicine. In The Lives of a Cell; Notes of a Biology Watcher, 35–42. New York: Viking Press
Acknowledgements
We thank Roxy A Tate for editorial assistance. H Taegtmeyer's laboratory is supported by grants from the National Heart, Lung, and Blood Institute of the US Public Health Service.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Taegtmeyer, H., Dilsizian, V. Imaging myocardial metabolism and ischemic memory. Nat Rev Cardiol 5 (Suppl 2), S42–S48 (2008). https://doi.org/10.1038/ncpcardio1186
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpcardio1186
This article is cited by
-
LVEF reserve: State of the heart is a matter of time, jeopardy and ischemic memory
Journal of Nuclear Cardiology (2022)
-
Strategies for Imaging Metabolic Remodeling of the Heart in Obesity and Heart Failure
Current Cardiology Reports (2022)
-
The Cardiorenal Axis: Myocardial Perfusion, Metabolism, and Innervation
Current Cardiology Reports (2019)
-
Cardiac molecular imaging to track left ventricular remodeling in heart failure
Journal of Nuclear Cardiology (2017)
-
Exploring the Pathophysiology of Takotsubo Cardiomyopathy
Current Cardiology Reports (2017)