Abstract
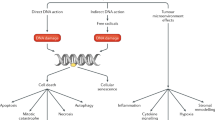
Taxanes have emerged as a potent class of chemotherapeutic agents in many malignancies, with two taxanes now in clinical use. Their mechanism of action against tumor cells is by alteration of microtubule dynamics, which causes cell-cycle arrest during mitosis. Docetaxel binds to the microtubules with a higher affinity than paclitaxel, and over a broader range of cell-cycle activities. It has also been shown to promote apoptosis via BCL2 phosphorylation. In hormone-refractory prostate cancer, docetaxel has been studied as both a single agent and in combination with estramustine, and in different treatment schedules, with demonstrated efficacy. Two phase III trials have confirmed a survival benefit, making docetaxel the first chemotherapy agent with proven efficacy against prostate cancer. In urothelial cancer, docetaxel has demonstrated activity and has been investigated as a single agent and in combination regimens. A phase III trial comparing docetaxel and cisplatin to methotrexate, vinblastine, doxorubicin, and cisplatin was inferior when evaluating response rates and overall survival. More recent phase II trials combining docetaxel with two additional agents have shown promise, but confirmatory trials are needed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Mollinedo F and Gajate C (2003) Microtubules, microtubule-interfering agents and apoptosis. Apoptosis 8: 413–450
Valiron O et al. (2001) Microtubule dynamics. Cell Mol Life Sci 58: 2069–2084
Checchi P et al. (2003) Microtubule-interacting drugs for cancer treatment. Trends Pharmacol Sci 24: 361–365
Mitchison T and Kirschner M (1984) Dynamic instability of microtubule growth. Nature 312: 237–242
Jordan MA and Wilson L (2004) Microtubules as a target for anticancer drugs. Nat Rev Cancer 4: 253–265
Margolis RL and Wilson L (1978) Opposite end assembly and disassembly of microtubules at steady state in vitro. Cell 13: 1–8
Downing KH (2000) Structural basis for the interaction of tubulin with proteins and drugs that affect microtubule dynamics. Annu Rev Cell Dev Biol 16: 89–111
Stein CA (1999) Mechanisms of action of taxanes in prostate cancer. Semin Oncol 26 (Suppl 17): 3–7
Haldar S et al. (1997) Bcl2 is the guardian of microtubule integrity. Cancer Res 57: 229–233
Tsujimoto Y and Croce CM (1986) Analysis of the structure, transcripts, and protein products of bcl-2, the gene involved in human follicular lymphoma. Proc Natl Acad Sci U S A 83: 5214–5218
Krajewski S et al. (1995) Reduced expression of proapoptotic gene BAX is associated with poor response rates to combination chemotherapy and shorter survival in women with metastatic breast adenocarcinoma. Cancer Res 55: 4471–4478
Apakama I et al. (1996) bcl-2 overexpression combined with p53 protein accumulation correlates with hormone-refractory prostate cancer. Br J Cancer 74: 1258–1262
Haldar S et al. (1996) Taxol induces bcl-2 phosphorylation and death of prostate cancer cells. Cancer Res 56: 1253–1255
Bruno R and Sanderink GJ (1993) Pharmacokinetics and metabolism of Taxotere (docetaxel). Cancer Surv 17: 305–313
Extra JM et al. (1993) Phase I and pharmacokinetic study of Taxotere (RP 56976; NSC 628503) given as a short intravenous infusion. Cancer Res 53: 1037–1042
US Food and Drug Administration (online May 2004) Taxotere® (docetaxel) prescribing information. [View PDF] (accessed January 2005)
Picus J and Schultz M (1999) Docetaxel (Taxotere) as monotherapy in the treatment of hormone-refractory prostate cancer: preliminary results. Semin Oncol 26 (Suppl 17): 14–18
Friedland D et al. (1999) A phase II trial of docetaxel (Taxotere) in hormone-refractory prostate cancer: correlation of antitumor effect to phosphorylation of Bcl-2. Semin Oncol 26 (Suppl 17): 19–23
Berry W et al. (2001) Phase II trial of single-agent weekly docetaxel in hormone-refractory, symptomatic, metastatic carcinoma of the prostate. Semin Oncol 28 (Suppl 15): 8–15
Beer TM et al. (2001) Phase II study of weekly docetaxel in symptomatic androgen-independent prostate cancer. Ann Oncol 12: 1273–1279
Gravis G et al. (2003) Weekly administration of docetaxel for symptomatic metastatic hormone-refractory prostate carcinoma. Cancer 98: 1627–1634
Kreis W et al. (1997) Unique synergism or antagonism of combinations of chemotherapeutic and hormonal agents in human prostate cancer cell lines. Br J Urol 79: 196–202
Petrylak DP et al. (2000) Response and preliminary survival results of a phase II study of docetaxel (D) estramustine (E) in patients with androgen-independent prostate cancer (AIPCA). Proc Am Soc Clin Oncol 19: (abstract 334)
Sinibaldi VJ et al. (2002) Phase II evaluation of docetaxel plus one-day oral estramustine phosphate in the treatment of patients with androgen independent prostate carcinoma. Cancer 94: 1457–1465
Savarese DM et al. (2001) Phase II study of docetaxel, estramustine, and low-dose hydrocortisone in men with hormone-refractory prostate cancer: a final report of CALGB 9780. Cancer and Leukemia Group B. J Clin Oncol 19: 2509–2516
Petrylak DP et al. (2004) Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med 351: 1513–1520
Tannock IF et al. (2004) Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 351: 1502–1512
McCaffrey JA et al. (1997) Phase II trial of docetaxel in patients with advanced or metastatic transitional-cell carcinoma. J Clin Oncol 15: 1853–1857
de Wit R et al. (1998) Docetaxel (Taxotere): an active agent in metastatic urothelial cancer; results of a phase II study in non-chemotherapy-pretreated patients. Br J Cancer 78: 1342–1345
Dimopoulos MA et al. (1998) Treatment of patients with metastatic urothelial carcinoma and impaired renal function with single-agent docetaxel. Urology 52: 56–60
Garcia del Muro X et al. (2002) Phase II multicentre study of docetaxel plus cisplatin in patients with advanced urothelial cancer. Br J Cancer 86: 326–330
Gitlitz BJ et al. (2003) A phase II study of gemcitabine and docetaxel therapy in patients with advanced urothelial carcinoma. Cancer 98: 1863–1869
Dreicer R et al. (2003) Phase II trial of gemcitabine and docetaxel in patients with advanced carcinoma of the urothelium: a trial of the Eastern Cooperative Oncology Group. Cancer 97: 2743–2747
Bamias A et al. (2004) Docetaxel and cisplatin with granulocyte colony-stimulating factor (G-CSF) versus MVAC with G-CSF in advanced urothelial carcinoma: a multicenter, randomized, phase III study from the Hellenic Cooperative Oncology Group. J Clin Oncol 22: 220–228
Sengelov L et al. (1998) Docetaxel and cisplatin in metastatic urothelial cancer: a phase II study. J Clin Oncol 16: 3392–3397
von der Maase H et al. (2000) Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol 17: 3068–3077
Pectasides D et al. (2000) Chemotherapy with cisplatin, epirubicin and docetaxel in transitional cell urothelial cancer. Phase II trial. Eur J Cancer 36: 74–79
Pectasides D et al. (2002) Weekly chemotherapy with docetaxel, gemcitabine and cisplatin in advanced transitional cell urothelial cancer: a phase II trial. Ann Oncol 13: 243–250
National Cancer Institute (online 12 December 2003) Common Terminology Criteria for Adverse Events v3.0. [http://ctep.cancer.gov/reporting/ctc.html] (accessed January 2005)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mackler, N., Pienta, K. Drug Insight: use of docetaxel in prostate and urothelial cancers. Nat Rev Urol 2, 92–100 (2005). https://doi.org/10.1038/ncpuro0099
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpuro0099
This article is cited by
-
A Reflection on the Mechanism of the Role of Nanoparticles in Increasing the Efficacy of Anti-tumour Properties of Docetaxel
Current Pathobiology Reports (2021)
-
Optimizing pharmacokinetics of intravesical chemotherapy for bladder cancer
Nature Reviews Urology (2019)
-
Additive naftopidil treatment synergizes docetaxel-induced apoptosis in human prostate cancer cells
Journal of Cancer Research and Clinical Oncology (2018)
-
Dual Drug Loaded Biodegradable Nanofibrous Microsphere for Improving Anti-Colon Cancer Activity
Scientific Reports (2016)
-
Do patients with metastatic urothelial carcinoma benefit from docetaxel as second-line chemotherapy?
Clinical and Translational Oncology (2014)