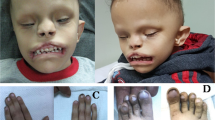
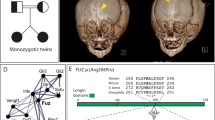
Abstract
KIF7, the human ortholog of Drosophila Costal2, is a key component of the Hedgehog signaling pathway. Here we report mutations in KIF7 in individuals with hydrolethalus and acrocallosal syndromes, two multiple malformation disorders with overlapping features that include polydactyly, brain abnormalities and cleft palate. Consistent with a role of KIF7 in Hedgehog signaling, we show deregulation of most GLI transcription factor targets and impaired GLI3 processing in tissues from individuals with KIF7 mutations. KIF7 is also a likely contributor of alleles across the ciliopathy spectrum, as sequencing of a diverse cohort identified several missense mutations detrimental to protein function. In addition, in vivo genetic interaction studies indicated that knockdown of KIF7 could exacerbate the phenotype induced by knockdown of other ciliopathy transcripts. Our data show the role of KIF7 in human primary cilia, especially in the Hedgehog pathway through the regulation of GLI targets, and expand the clinical spectrum of ciliopathies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Baker, K. & Beales, P.L. Making sense of cilia in disease: the human ciliopathies. Am. J. Med. Genet. C. Semin. Med. Genet. 151C, 281–295 (2009).
Salonen, R., Herva, R. & Norio, R. The hydrolethalus syndrome: delineation of a “new,” lethal malformation syndrome based on 28 patients. Clin. Genet. 19, 321–330 (1981).
Mee, L. et al. Hydrolethalus syndrome is caused by a missense mutation in a novel gene HYLS1. Hum. Mol. Genet. 14, 1475–1488 (2005).
Dammermann, A. et al. The hydrolethalus syndrome protein HYLS-1 links core centriole structure to cilia formation. Genes Dev. 23, 2046–2059 (2009).
Schinzel, A. & Schmid, W. Hallux duplication, postaxial polydactyly, absence of the corpus callosum, severe mental retardation, and additional anomalies in two unrelated patients: a new syndrome. Am. J. Med. Genet. 6, 241–249 (1980).
Schinzel, A. & Kaufmann, U. The acrocallosal syndrome in sisters. Clin. Genet. 30, 399–405 (1986).
Robbins, D.J. et al. Hedgehog elicits signal transduction by means of a large complex containing the kinesin-related protein costal2. Cell 90, 225–234 (1997).
Kalderon, D. Hedgehog signaling: Costal-2 bridges the transduction gap. Curr. Biol. 14, R67–R69 (2004).
Cheung, H.O. et al. The kinesin protein Kif7 is a critical regulator of Gli transcription factors in mammalian hedgehog signaling. Sci. Signal. 2, ra29 (2009).
Endoh-Yamagami, S. et al. The mammalian Cos2 homolog Kif7 plays an essential role in modulating Hh signal transduction during development. Curr. Biol. 19, 1320–1326 (2009).
Liem, K.F. Jr., He, M., Ocbina, P.J. & Anderson, K.V. Mouse Kif7/Costal2 is a cilia-associated protein that regulates Sonic hedgehog signaling. Proc. Natl. Acad. Sci. USA 106, 13377–13382 (2009).
Khanna, H. et al. A common allele in RPGRIP1L is a modifier of retinal degeneration in ciliopathies. Nat. Genet. 41, 739–745 (2009).
Leitch, C.C. et al. Hypomorphic mutations in syndromic encephalocele genes are associated with Bardet-Biedl syndrome. Nat. Genet. 40, 443–448 (2008).
Zaghloul, N.A. et al. Functional analyses of variants reveal a significant role for dominant negative and common alleles in oligogenic Bardet-Biedl syndrome. Proc. Natl. Acad. Sci. USA 107, 10602–10607 (2010).
Tay, S.Y., Ingham, P.W. & Roy, S. A homologue of the Drosophila kinesin-like protein Costal2 regulates Hedgehog signal transduction in the vertebrate embryo. Development 132, 625–634 (2005).
Nasevicius, A. & Ekker, S.C. Effective targeted gene 'knockdown' in zebrafish. Nat. Genet. 26, 216–220 (2000).
Wilson, C.W. et al. Fused has evolved divergent roles in vertebrate Hedgehog signalling and motile ciliogenesis. Nature 459, 98–102 (2009).
Wolff, C., Roy, S. & Ingham, P.W. Multiple muscle cell identities induced by distinct levels and timing of hedgehog activity in the zebrafish embryo. Curr. Biol. 13, 1169–1181 (2003).
Badano, J.L. et al. Dissection of epistasis in oligogenic Bardet-Biedl syndrome. Nature 439, 326–330 (2006).
Davis, E.E. et al. TTC21B contributes both causal and modifying alleles across the ciliopathy spectrum. Nat. Genet. 43, 189–196 (2011).
Patzke, S. et al. CSPP is a ciliary protein interacting with Nephrocystin 8 and required for cilia formation. Mol. Biol. Cell 21, 2555–2567 (2010).
Delous, M. et al. The ciliary gene RPGRIP1L is mutated in cerebello-oculo-renal syndrome (Joubert syndrome type B) and Meckel syndrome. Nat. Genet. 39, 875–881 (2007).
Valente, E.M. et al. Mutations in TMEM216 perturb ciliogenesis and cause Joubert, Meckel and related syndromes. Nat. Genet. 42, 619–625 (2010).
Goetz, S.C. & Anderson, K.V. The primary cilium: a signalling centre during vertebrate development. Nat. Rev. Genet. 11, 331–344 (2010).
Kim, J. et al. Functional genomic screen for modulators of ciliogenesis and cilium length. Nature 464, 1048–1051 (2010).
Monnier, V., Ho, K.S., Sanial, M., Scott, M.P. & Plessis, A. Hedgehog signal transduction proteins: contacts of the Fused kinase and Ci transcription factor with the kinesin-related protein Costal2. BMC Dev. Biol. 2, 4 (2002).
Martinelli, D.C. & Fan, C.M. Gas1 extends the range of Hedgehog action by facilitating its signaling. Genes Dev. 21, 1231–1243 (2007).
Acknowledgements
We are grateful to families and to the French Society of Fetal Pathology (SOFFOET) for participating in the study, to C. Dubourg, P. Wieacker, B. Leroy, N. Laurent, V. Fermeaux, S. Odent for fetuses' referral and to A. Schinzel and A. David for ACLS samples. We thank M. Zarhrate, A. Aguilar, N. Spasky and L. Besse for technical help. We thank N. Boddaert for helpful discussion. This work was supported by grants from ANR (FETALCILIOPATHIES number 07-MRAR-010-02 and FOETOCILPATH number BLAN-1122-01), E-RARE (Cranirare number 07-ERare-001-01) the Scientific and Technological Research Council of Turkey (TUBITAK, grant number 108S420 to N.A.A.), the US National Institutes of Health grant R01HD04260 from the National Institute of Child Health and Development (N.K.), R01DK072301 from the National Institute of Diabetes, Digestive and Kidney Disorders (N.K.) and the European Union (EU-SYSCILIA; E.E.D., N.K. and P.L.B.). A.P. was granted a fellowship from the Académie de Médecine. S.T. is supported by NIH 'Hereditary Basis of Neural Tube Defects' N° NS039818-07 to M. Speer. K.L.M.C. is supported by the Huygens Scholarship Programme and the Netherlands Organization for Scientific Research (NWO Toptalent-021.001.014). M.W. was a fellow of the Guggenheim Foundation and was supported by the March of Dimes Foundation (1-FY07-422). P.L.B. is a Wellcome Trust Senior Research Fellow. N.K. is a Distinguished George W. Brumley Professor.
Author information
Authors and Affiliations
Contributions
S. Patrier, M.-H.S.F., F.E.-R. and J.-P.S. performed the fetal examinations. N.J. did the fetal karyotyping. L.R. and M.G. referred the HLS family. D.B. did the ultrasound diagnosis. G.O., Y.A., G.E.U., K.B., R.H., P.L.B., C.T., V.C.-D. and E.C. referred subjects. C.L.B., M.W., K.C., N.A.A., C.B.-F., E.U. and P.N. performed the homozygosity mapping and LOD score calculation. C.G., N.E., L.F., K.L.M.C., M.W. and C.B.-F. performed linkage and sequence analyses. S. Pruvost and E.U. did the chip experiments. S.T. and N.C. performed the expression analysis. A.S. performed the electron microscopy. N.G. and A.P. performed the confocal microscopy and measurement of cilia length. E.E.D. and N.K. did the zebrafish experiments. A.M., S.L. and M.V. advised the work. T.A.-B. designed the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2 and Supplementary Tables 1–4. (PDF 335 kb)
Supplementary Table 5
List of genes deregulated in fetuses with KIF7 mutations (XLS 3780 kb)
Rights and permissions
About this article
Cite this article
Putoux, A., Thomas, S., Coene, K. et al. KIF7 mutations cause fetal hydrolethalus and acrocallosal syndromes. Nat Genet 43, 601–606 (2011). https://doi.org/10.1038/ng.826
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.826
This article is cited by
-
Regulation of ciliary homeostasis by intraflagellar transport-independent kinesins
Cell Death & Disease (2024)
-
Genetic and protein interaction studies between the ciliary dyslexia candidate genes DYX1C1 and DCDC2
BMC Molecular and Cell Biology (2023)
-
Molecular investigation in individuals with orofacial clefts and microphthalmia-anophthalmia-coloboma spectrum
European Journal of Human Genetics (2023)
-
Molecular diagnosis and novel genes and phenotypes in a pediatric thoracic insufficiency cohort
Scientific Reports (2023)
-
A new enrichment approach for candidate gene detection in unexplained recurrent pregnancy loss and implantation failure
Molecular Genetics and Genomics (2023)