Abstract
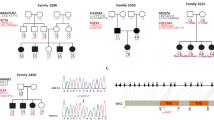
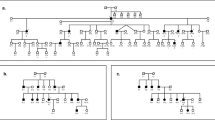
Two percent of the residents of Bengkala, Bali, have profound, congenital, neurosensory, nonsyndromal deafness due to an autosomal recessive mutation at the DFNB3 locus. We have employed a direct genorne–wide disequilibrium search strategy, allele–frequency–dependent homozygosity mapping (AHM), and an analysis of historical recombinants to map DFNB3 and position the locus relative to flanking markers. DFNB3 maps to chromosome 17, closest to D17S261, pRM7–GT and D17S805. In individuals homozygous for DFNB3, historical recombinant genotypes for the flanking markers, D17S122 and D17S783, place DFNB3 in a 5.3 cM interval of the pericentromeric region of chromosome 17 on a refined linkage map of 17p–17q12. Based on conserved synteny, the murine sh2 gene may be the homologue of DFNB3.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Morton, N.E. Genetic epidemiology of hearing impairment. Genetics of Hearing Impairment (eds Ruben, R.J., Van DeWater, T.R. & Steel, K.P.) 630, 16–31 (New York Academy of Sciences, New York, 1991).
Nance, W.E. & McConnell, F.E. Status and prospects in hereditary deafness. In Advances in Human Genetics, (eds Harris, H. & Hirschhom, K.) 4, 173–237 (Plenum Press, New York, 1973).
Nance, W.E. & Sweeney, A. Genetic factors in deafness of early life. Otolaryngologic Clinic of North America 8, 19–48 (1975).
Marres, H.A.M. & Cramers, W.R.J. Autosomal recessive nonsyndromal profound childhood deafness in a large pedigree. Arch. Otolaryngol. Head Neck Surg. 115, 591–595 (1989).
Steele, M.W. Genetics of congenital deafness. Ped. Clin. North Am. 28, 973–980 (1981).
Stevenson, A.C. & Cheeseman, E.A. Hereditary deaf mutism, with particular reference to Northern Ireland. Ann. hum. Genet. 20, 177–234 (1956).
Brownstein, Z., Friedlander, Y., Peritz, E. & Cohen, T. Estimated number of loci for autosomal recessive severe nerve deafness within the Israeli Jewish population, with implications for genetic counseling. Am. J. med. Genet. 41, 306–312 (1991).
Chung, C.S. & Brown, K.S. Family studies of early childhood deafness ascertained through the Clarke school for the deaf. Am. J. hum. Genet. 22, 630–640 (1970).
Winata, S. et al. Congenital nonsyndromal autosomal recessive deafness in Bengkala, an isolated Balinese village. J. med Genet. (in the press).
Weber, J.L. & Wong, C. Mutation of short tandem repeats. Hum. molec. Genet. 2, 1123–1128 (1993).
Ott, J. Strategies for characterizing highly polymorphic markers in human gene mapping. Am. J. hum. Genet. 51, 283–290 (1992).
Lander, E.S. & Botstein, D. Homozygosity mapping: A way to map human recessive traits with DNA of inbred children. Science 236, 1567–1570 (1987).
Mueller, R.F. & Bishop, D.T. Autozygosity mapping, complex consanguinity and autosomal recessive hearing impairment gene. Hered. Deaf. Newsletter 9, 14–15 (1992).
Sirugo, G. et al. Friedreich Ataxia in Louisiana Acadians: demonstration of a founder effect by analysis of microsatellite-generated extended haplotypes. Am. J. hum. Genet. 50, 559–566 (1992).
Hastbacka, J., Kaitila, I., Sistonen, P. & de la Chapelle, A. Linkage disequilibrium mapping in isolated founder populations: diastrophic dysplasia in Finland. Nature Genet. 2, 204–211 (1992).
Ott, J. Analysis of human genetic linkage (Johns Hopkins Press, Baltimore, 1991).
Weeks, D.E., Ott, J. & Lathrop, G.M. SLINK: a general simulation program for linkage analysis. Am J. hum. Genet. 47, A204 (1990).
Weber, J.L. et al. Evidence for human melotic recombination interference obtained through construction of a short tandem repeat-polymorphism linkage map of chromosome 19. Am. J. hum. Genet. 53, 1079–1095 (1993).
Pentao, L., Wise, C.A., Chinault, C., Patel, P.I. & Lupski, J.R. Charcot-Marie-Tooth type 1A duplication appears to arise from recombination at repeat sequences flanking the 1.5 Mb monomer unit. Nature Genet. 2, 292–300 (1993).
Chevillard, C. et al. Relationship between Charcot-Marie-Tooth 1A and Smith-Magenis regions. snU3 may be a candidate gene for the Smith-Magenis syndrome. Hum. molec. Genet. 2, 1235–1243 (1993).
Chance, P.F. et al. DNA deletion associated with hereditary neuropathy with liability to pressure palsies. Cell 72, 143–151 (1993).
Roa, B.B., Dyck, P.J., Marks, H.G., Chance, P.F. & Lupski, J.R. Dejerine-Sottas syndrome associated with point mutation In the peripheral myelin protein 22 (PMP22) gene. Nature Genet. 5, 269–273 (1993).
Roa, B.B. et al. Charcot-Marie-Tooth disease type 1A associated with a spontaneous point mutation In the PMP22 gene. New Engl. J. Med. 329, 96–101 (1993).
Valentijn, L.J. et al. The peripheral myelin gene PMP-22/GAS-3 is duplicated in Charcot-Marie-Tooth disease type 1A. Nature Genet. 1, 166–170 (1992).
Morell et al. Analysis of short tandem repeat (STR) allele frequency distributions in a Balinese population. Hum. molec. Genet., (in the press).
Greenberg, F. et al. Molecular analysis of the Smith-Magenis syndrome: A possible contiguous-gene syndrome associated with del (17) (p11.2). Am. J. hum. Genet. 49, 1207–1218 (1991).
Guilford, P. et al. A non-syndromic form of neurosensory, recessive deafness maps to the pericentromeric region of chromosome 13q. Nature Genet. 6, 24–28 (1994).
Guilford, P. et al. A human gene responsible for neurosensory, nonsyndromic recessive deafness is a candidate homologue of the mouse sh-1 gene. Hum. molec. Genet. 3, 989–993 (1994).
Ross, D.A., Kennerson, M.L. & Nicholson, G.A. Identification of fourteen potential sequences (HTF islands) in the CMT1A duplication/HNPP deletion area. Am. J. hum. Genet. 53, 1351 (1993).
Steel, K.P. Similarities between mice and humans with hereditary deafness. In Genetics of Hearing Impairment, (eds Ruben, R.J., Van DeWater, T.R. & Steel, K.P.) 630, 68–79 (New York Academy of Sciences, New York, 1991).
Lyon, M.F. & Kirby, M.C. Mouse chromosome atlas. Mouse Genome 91, 61 (1993).
Welcher, A.A., Suter, U., De Leon, M., Snipes, G.J. & Shooter, E.M. A myelin protein is encoded by the homologue of a growth arrest-specific gene. Proc. natn. Acad. Sci. U.S.A. 88, 7195–7199 (1991).
Suter, U. et al. Trembler mouse carries a point mutation in a myelin gene. Nature 356, 241–244 (1992).
Buetow, K.H. et al. Integrated human genome-wide maps constructed using CEPH reference panel. Nature Genet. 6, 391–393 (1994).
Anderson, L.A. et al. High-density genetic map of the BRCA1 region of chromosome 17q12–q21. Genomics 17, 618–623 (1993).
Abel, K.J. et al. A radiation hybrid map of the BRCA1 region of chromosome 17q12–q21. Genomics 17, 632–641 (1993).
Flejter, W.L. et al. Multicolor FISH mapping with Alu-PCR-Amplified YAC clone DNA determines the order of markers in the BRCA1 region on chromosomes 17q12–q21. Genomics 17, 624–631 (1993).
Weber, J.L. Know thy genome. Nature Genet. 7, 343–344 (1994).
Reed, P.W. et al. Chromosome-specific microsatellite sets for fluorescence-based, semi-automated genome mapping. Nature Genet. 7, 390–395 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Friedman, T., Liang, Y., Weber, J. et al. A gene for congenital, recessive deafness DFNB3 maps to the pericentromeric region of chromosome 17. Nat Genet 9, 86–91 (1995). https://doi.org/10.1038/ng0195-86
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0195-86
This article is cited by
-
Novel compound heterozygous MYO15A splicing variants in autosomal recessive non-syndromic hearing loss
BMC Medical Genomics (2024)
-
Loss-of-function mutations in MYO15A and OTOF cause non-syndromic hearing loss in two Yemeni families
Human Genomics (2023)
-
Analysis of the genotype–phenotype correlation of MYO15A variants in Chinese non-syndromic hearing loss patients
BMC Medical Genomics (2022)
-
Identification of autosomal recessive nonsyndromic hearing impairment genes through the study of consanguineous and non-consanguineous families: past, present, and future
Human Genetics (2022)
-
A synonymous variant in MYO15A enriched in the Ashkenazi Jewish population causes autosomal recessive hearing loss due to abnormal splicing
European Journal of Human Genetics (2021)