Abstract
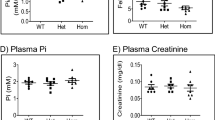
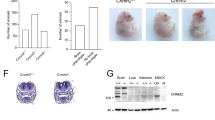
Iron is essential for many cellular functions; consequently, disturbances of iron homeostasis, leading to either iron deficiency or iron overload, can have significant clinical consequences. Despite the clinical prevalence of these disorders, the mechanism by which dietary iron is absorbed into the body is poorly understood. We have identified a key component in intestinal iron transport by study of the sex–linked anaemia (sla) mouse, which has a block in intestinal iron transport1. Mice carrying the sla mutation develop moderate to severe microcytic hypochromic anaemia1. Although these mice take up iron from the intestinal lumen into mature epithelial cells normally2, the subsequent exit of iron into the circulation is diminished3. As a result, iron accumulates in enterocytes and is lost during turnover of the intestinal epithelium4. Biochemical studies have failed to identify the underlying difference between sla and normal mice, therefore, we used a genetic approach to identify the gene mutant in sla mice. We describe here a novel gene, Heph, encoding a transmembrane–bound ceruloplasmin homologue that is mutant in the sla mouse and highly expressed in intestine. We suggest that the hephaestin protein is a multi–copper ferroxidase necessary for iron egress from intestinal enterocytes into the circulation and that it is an important link between copper and iron metabolism in mammals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Grewal, M.S. A sex–linked anemia in the mouse. Genet. Res. 3, 238–247 (1962).
Manis, J. Active transport of iron by intestine: selective genetic defect in the mouse. Nature 227, 385–386 (1970).
Bannerman, R.M. Genetic defects of iron transport. Fed. Proc. 35, 2281–2285 (1976).
Edwards, J.A., Hoke, J.E., Mattioli, M. & Reichlin, M. Ferritin distribution and synthesis in sex–linked anemia. J. Lab. Clin. Med. 90, 68–76 (1977).
Falconer, D.S. & Isaacson, J.H. The genetics of sex–linked anaemia in the mouse. Genet. Res. 3, 248 –250 (1962).
Anderson, G.J. et al. Mapping the gene for sex–linked anemia: an inherited defect of intestinal iron absorption in the mouse. Genomics 48, 34–39 (1998).
Brockdorff, N., Kay, G., Cattanach, B.M. & Rastan, S. Molecular genetic analysis of the Ta25H deletion: evidence for additional deleted loci. Mamm. Genome 1, 152–157 (1991).
Klomp, L.W., Farhangrazi, Z.S., Dugan, L.L. & Gitlin, J.D. Ceruloplasmin gene expression in the murine central nervous system [see comments]. J. Clin. Invest. 98, 207– 215 (1996).
Skikne, B.S. & Baynes, R.D. in Iron Metabolism in Health and Disease (eds Brock, J.H., Halliday, J.W., Pippard, M.J. & Powell, L.W.) 151–187 (W.B. Saunders, London, 1994).
Askwith, C.C., de Silva, D. & Kaplan, J. Molecular biology of iron acquisition in Saccharomyces cerevisiae. Mol. Microbiol. 20, 27– 34 (1996).
Askwith, C. et al. The FET3 gene of S. cerevisiae encodes a multicopper oxidase required for ferrous iron uptake. Cell 76, 403–410 (1994).
De Silva, D.M., Askwith, C.C., Eide, D. & Kaplan, J. The FET3 gene product required for high affinity iron transport in yeast is a cell surface ferroxidase. J. Biol. Chem. 270, 1098– 1101 (1995).
Stearman, R., Yuan, D.S., Yamaguchi–Iwai, Y., Klausner, R.D. & Dancis, A. A permease–oxidase complex involved in high–affinity iron uptake in yeast. Science 271, 1552–1557 (1996).
Lee, G.R., Nacht, S., Lukens, J.N. & Cartwright, G.E. Iron metabolism in copper–deficient swine. J. Clin. Invest. 47 , 2058–2069 (1968).
Harris, Z.L. et al. Aceruloplasminemia: molecular characterization of this disorder of iron metabolism. Proc. Natl Acad. Sci. USA 92, 2539–2543 (1995).
Logan, J.I. Hereditary deficiency of ferroxidase (aka caeruloplasmin) [editorial]. J. Neurol. Neurosurg. Psychiatry 61, 431– 432 (1996).
Danks, D.M. in The Metabolic Basis of Inherited Disease (eds Scriver, C., Beaudet, A., Sly, W. & Valle, D.) 1411–1432 (McGraw–Hill, New York, New York, 1989).
Bothwell, T.H., Charlton, R.W., Cook, J.D. & Finch, C.A. in Iron Metabolism in Man 256–283 (Blackwell Scientific Publications, Oxford, 1979).
Lombard, M., Chua, E. & O'Toole, P. Regulation of intestinal non–haem iron absorption. Gut 40, 435–439 (1997).
Halliday, J.W. & Powell, L.W. in Iron and Human Disease (eds Lauffer, R.B. et al.) 131– 160 (CRC Press, Boca Raton, 1992).
Bothwell, T.H. & MacPhail, A.P. Hereditary hemochromatosis: etiologic, pathologic, and clinical aspects. Semin. Hematol. 35, 55–71 (1998).
Davis, L.G., Dibner, M.D. & Battey, J.F. in Basic Methods in Molecular Biology 42–43 (Elsevier, New York, 1986).
Kuo, Y.M., Gitschier, J. & Packman, S. Developmental expression of the mouse mottled and toxic milk genes suggests distinct functions for the Menkes and Wilson disease copper transporters. Hum. Mol. Genet. 6, 1043– 1049 (1997).
Acknowledgements
We thank N. Brockdorff and E. Tuddenham for assaying the sla animals; M. Gunthorpe and J. DeYoung for DNA synthesis and sequencing; M. Schueler and H. Willard for communicating unpublished results; and M. Fleming and N. Andrews for unpublished sla mapping data. J.G. is an associate investigator with the Howard Hughes Medical Institute. This work was supported in part by a grant from the National Health and Medical Research Council of Australia to G.J.A. and by a grant from the National Institutes of Health to J.G.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vulpe, C., Kuo, YM., Murphy, T. et al. Hephaestin, a ceruloplasmin homologue implicated in intestinal iron transport, is defective in the sla mouse. Nat Genet 21, 195–199 (1999). https://doi.org/10.1038/5979
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5979
This article is cited by
-
Association between iron metabolism and SARS-COV-2 infection, determined by ferritin, hephaestin and hypoxia-induced factor-1 alpha levels in COVID-19 patients
Molecular Biology Reports (2023)
-
Lactoferrin: from the structure to the functional orchestration of iron homeostasis
BioMetals (2023)
-
Can iron chelators ameliorate viral infections?
BioMetals (2023)
-
Serum ferritin levels are associated with advanced liver fibrosis in treatment-naive autoimmune hepatitis
BMC Gastroenterology (2022)
-
Internalization and Decrease of Duodenal DMT1 Involved in Transient Suppression of Iron Uptake in Short-Acting Mucosal Block
Biological Trace Element Research (2022)